Research Article - (2021) Volume 9, Issue 12
A Comparison of IV Dexmedetomidine and Midazolam for Sedation in Patients under Neuraxial Anaesthesia
*Correspondence: U. Juvairiya Banu, Department of Anaesthesiology, Pain Medicine and Critical Care, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, India, Email:
Abstract
The Sedation reduces the postoperative recall of intra operative events. In the present study, total of 60 cases were included ; of which 30 were in group D for whom sedation with inj.Dexmedetomidine was given intravenously after neuraxial blockad e, and 30 in group. A significant decrease m pulse rate and mean arterial blood pressure (MAP) were observed .when compared with baseline m both groups throughout the surgery but the fall in pulse rate and MAP was greater with Dexmedetomidine infusion up to 45 minutes after spinal anaesthesia when compared with Midazolam Dexmedetomidine in Group D showed a better grade of Ramsay's Sedation score compared to inj. Midazolam in Group M. Dexmedetomidine for sedation was safe and feasible compared to Midazolam..Keywords
midazolam, anesthesia, pulse rateIntroduction
Neuraxial Anaesthesia 1s a unique technique to provide sensory and motor blockade in the large part of the body. Usually, without any additive, one can achieve 60-90 minutes of anaesthesia with a spinal block. Nowadays, anaesthetists are fortunate enough to have agents that can be used either intrathecally or intravenously to enhance the efficacy and duration of the block. They are named Adjuvants. Some of the Adjuvants that were previously used are Epinephrine, Magnesium Sulphate, Fentanyl, Midazolam, Clonidine, Dexmedetomidine activates the central nervous system and decreases plasma catecholamine level with stimulation of alpha 2 adrenoceptors in post-synaptic site, resulting in decrease in heart rate and blood pressure, in addition to sedation and anxiolysis. It was demonstrated to decrease pam and catecholamine [1].
Neuraxial anaesthesia is a unique technique to provide sensory and motor blockade in the body. Dexmedetomidine activates the central nervous system and decreases plasma catecholamine level with stimulation of alpha2 adrenoreceptors in post-synaptic site, resulting in decrease in heart rate and blood pressure, added to sedation and anxiolysis [2].
Midazolam which is a short-acting water-soluble form of benzodiazepine, is commonly used for premedication m order to perform amnes1a, sedation and to reduce perioperative anxiety. This effect depends on the binding to the gamma ammo butyrate receptors (GABA) at benzodiazepine site. This study is to compare the hemodynamic parameters, respiratory parameters and sedative effect of Intravenous Dexmedetomidine and Intravenous Midazolam [3].
This study is to compare the hemodynamic parameters, respiratory parameters and sedative effect of Intravenous Dexmedetomidine and Intravenous Midazolam.
Methodology
This study was done to compare intravenous Dexmedetomidine and intravenous Midazolam for sedation in patients under Neuraxial Anaesthesia. It was a randomized study. This study was approved by the Institutional Ethical Committee. 60 patients were posted for surgenes at Sree Balaji Medical College and Hospital, Chennai, and were studied during June 2016 - September 2017.
Inclusion Criteria: The ASA I, II & III patients. Age between 25-65 yrs. weighing 40-90 kg of both Genders. Patients undergoing electivesurgeries with Neuraxial. Anaesthesia with duration of 1 to 2 hrs.
Exclusion Criteria: ASA IV Patients. Morbidly obese patients. Chronic alcoholic patients. Pregnant women. Patients with hepatic diseases.
Patients were randomly divided into two groups as per the drug chosen for intraoperative sedation (Group D &M). The patients of group D were give n. Dexmedetomidine (1mcg/kg). Group M were injected with 0.01to 0.1 mg /kg, after which all the parameters were recorded.
Primary Stability of Miniscrew Implants
Some factors influencing stability are:
• Device features: length, diameter, thread shape, thread pitch, thread design, thread type, cutting flute, construction material
• Operator/surgical technique-related factors
• Patient characteristics: cortical bone thickness.
Design and Structural Characteristics
Length
The threaded portion of the mini implant is referred to as the length, which can vary from 4 to 15 mm. An increase in maximum insertion torque, removal torque and pullout resistance can be achieved by an increase in mini implant length. Mini implant lengths of 5 mm and more do not affect primary stability, because these greater lengths of mini implants will be entering medullary bone.
The thickness of soft tissue at site of insertion must also be taken into account, along with a bone support of 5-6mm, during the selection of mini implant length. The ideal length of a mini implant is 9 mm, as this is thought to cause less stress on the surrounding bone than shorter mini implants and has less risk of damage to neighbouring anatomical structures than longer mini implants. From a clinical standpoint, a mini implant of 4-6 mm can be inserted into the majority of intraoral sites.
Diameter
The diameter of the mini implant is directly proportional to the primary stability of the implant. The larger surface area allows for the force to be distributed, thereby reducing the pressure on the bone at insertion site.
Biomechanical yield, implantation success and fracture resistance are most influenced by diameter rather than length of implant. Stable results have been observed in implants of diameter 1.5-2.3 mm in maxilla and mandible & a significant loss of anchorage has been observed when mini implants with diameters of less than 1.2 mm were used. The ideal diameter is probably between 1.3 and 1.5 mm.
there is a risk of unwanted root contact when mini implants of diameters greater than 2 mm are used, as the mean inter- radicular space is generally between 2.5 mm and 3.5 mm.17The mini implants with a 1.3 mm diameter have shown a success rate of 88.6%, which is not particularly high but needs to be weighed against the lesser risk of iatrogenic damage [4].
Results
Both the Groups D and M were statistically comparable with regard to the mean heart rate where it was statistically insignificant (P > 0.05) during the baseline and after 5 minutes. Group M showed a higher heart rate during the significance. Both the group patients showed higher systolic and diastolic blood pressure. The oxygen saturation was statistically insignificant.
Table1: Mean heart rate of group D &M.
| Variables | Group d | Group m | p-value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Range | Mean± sd | Range | Mean± sd | ||||||
| Baseline | 60 - 112 | 82.9 ± | 12.23 | 63 -110 | 86.2 | ± | 12.7 | 0.309 | |
| After 5 min | 55 - 90 | 74.17 | ± | 12.79 | 60 -118 | 80.23 | ± | 14.02 | 0.067 |
| After 10 min | 54 - 104 | 76.17 | ± | 10.27 | 70 -118 | 89.87 | ± | 3.31 | 0.003 |
| After 15 min | 55 - 102 | 79.03 | ± | 12.62 | 68 -124 | 89.43 | ± | 12.44 | 0.004 |
| After 20 min | 55-105 | 77.8 | ± | 11.4 | 60 -124 | 85.97 | ± | 12.25 | 0.012 |
| After 35 min | 55 - 110 | 77.5 | ± | 11.9 | 65 -127 | 86.43 | ± | 12.41 | 0 |
| After 50 min | 60 - 95 | 79.27 | ± | 8.87 | 62 -106 | 84.27 | ± | 10.02 | 0.021 |
| After 65 min | 60 - 95 | 79.43 | ± | 9.23 | 65 -123 | 85.14 | ± | 10.15 | 0.004 |
| After 80 min | 60 - 95 | 79.31 | ± | 9.1 | 65 -120 | 84.32 | ± | 9.23 | 0.001 |
Table2: Systolic blood pressure comparison.
| Variables | Group d | Group m | P- Value |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range | Mean | ± | Sd | Range | Mean | ± | Sd | |||
| Baseline | 104 - | 160 | 127.07 | ± | 14.5 | 101 - 148 | 123.43 | ± | 10.62 | 0.273 |
| After 5 min | 75- | 171 | 121.6 | ± | 22.3 | 86 - 135 | 108.47 | ± | 12.46 | 0.005 |
| After 10 min | 90 - | 169 | 125.37 | ± | 18 | 100 - 180 | 139.87 | ± | 18.33 | 0.006 |
| After 15 min | 89 - | 151 | 118.43 | ± | 15.5 | 98 - 170 | 131.2 | ± | 19.06 | 0.003 |
| After 20 min | 83 - | 160 | 114.33 | ± | 15.1 | 102-174 | 126.21 | ± | 18.55 | 0.006 |
| After 35 min | 87 - | 162 | 115.57 | ± | 15.7 | 102-176 | 127.23 | ± | 18.52 | 0.01 |
| After 50 min | 92 - | 164 | 124.73 | ± | 15.6 | 104 - 179 | 127.2 | ± | 13.52 | 0.012 |
| After 65 min | 92 - | 165 | 126.22 | ± | 15.4 | 106 - 170 | 129.44 | ± | 13.44 | 0.041 |
| After 80 min | 92 - | 164 | 126.11 | ± | 15.3 | 106 - 172 | 128.51 | ± | 12.57 | 0.024 |
Both the Groups D and M were statistically comparable with regard to the systolic blood pressure where it was statistically insignificant (P > 0.05) during the baseline. However, it was statistically significant (P < 0.05) in all the other timeframes.
Table3: Diastolic blood pressure comparison.
| Variables | Group d | Group m | P- | ||
|---|---|---|---|---|---|
| Range | Mean± sd | Range | Mean± sd | Value | |
| Baseline | 62 - 100 | 80.3 ±9.24 | 53 - 107 | 85.27 ± 12.16 | 0.0802 |
| After 5 min | 54 - 96 | 77.2 ±10.63 | 50 - 88 | 71.37 ± 8.88 | 0.02 |
| After 10 min | 57 - 109 | 83.53 ± 12.35 | 60 - 116 | 89.73 ± 12.51 | 0.0192 |
| After 15 min | 61 - 102 | 76.53 ± 9.95 | 56 - 106 | 82.23 ± 12.12 | 0.058 |
| After 20 min | 60 - 100 | 76.88 ± 9.44 | 57 - 100 | 81.14 ± 12.55 | 0.051 |
| After 35 min | 58 - 105 | 77.76 ± 11.84 | 56 - 98 | 79.44 ± 12.41 | 0.009 |
| After 50 min | 51 - 100 | 79.70 ± 11.65 | 60 - 97 | 78.87 ± 10.92 | 0.0051 |
| After 65 min | 53 - 104 | 79.94 ± 10.98 | 62 - 100 | 79.02 ± 10.52 | 0.0414 |
| After 80 min | 54 - 105 | 78.65 ± 9.87 | 63 - 102 | 79.12 ± 10.98 | 0.0212 |
Group M showed a higher systolic blood pressure during the significance. Both the Groups D and M were statistically comparable with regard to the diastolic blood pressure where it was statistically insignificant (P > 0.05) during the baseline. However, it was statistically significant (P < 0.05) in all the other time frames. Group M showed a higher diastolic blood pressure during the significance.
Ramsay's Sedation score m patients who received Dexmedetomidine was of higher grade than those who received Midazolam. Among the cases , 67 % did not have major complicatio ns , 13 % had dizzines s, 10 %had vomit and another 10 % had nausea in the Group D. In the Group M, 70% had no complaints and remaining had nausea (fig.1).
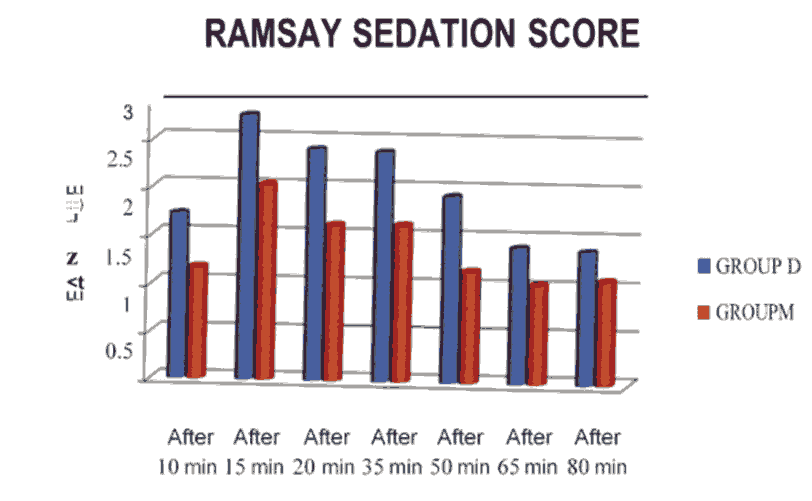
Figure 1: Ramsay sedation score.
Discussion
Haemodynamic effects- heart rate, systolic blood pressure, diastolic blood pressure, mean blood pressure, oxygen saturation and Ramsay's sedation score were monitored.
A significant decrease m pulse rate and mean arterialblood pressure (MAP) were observed when compared with baseline m both groups throughout the surgery but the fall in pulse rate and MAP was greater with Dexmedetomidine infusion upto 45 minutes after spinal anaesthesia when compared with Midazolam. With the data obtained, there was a statistically significant rate of decrease in pulse rate and mean blood pressure in Group D compared to Group M ,similar results were seen in kohrs et al[4]. There was also significant increase in the grade of Ramsay's Sedation score in group D compared to M. Dexmedetomidine had a better satisfactory sedation compared to Midazolam.
conclusion
Dexmedetomidine had a better satisfactory sedation compared to Midazolam. Midazolam provided a lesser haemodynamic fluctuation compared to Dexmedetomidine throughout the intraoperative period. In addition to th is, Midazolam has an additional advantage of cost effectiveness. In both groups, oxygen saturation was maintained throughout the procedure [5].
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee
Conflict of Interest
The authors declare no conflict of interest.
References
- Frizelle, Henry P, Duranteau Jacques, and Samii Kamran. "A comparison of propofol with a propofol-ketamine combination for sedation during spinal anesthesia." Anest Analg 84, (1997):1318-1322.
- Badrinath, Shyamala, Avramov Michail N, Shadrick Melissa, and Witt Thomas R, et al. "The use of a ketamine-propofol combination during monitored anesthesia care." Anest Analg 90, (2000): 858-862.
- Menigaux, Christophe, Fletcher Dominique, Dupont Xavier, and Guignard Bruno, at el. "The benefits of intraoperative small-dose ketamine on postoperative pain after anterior cruciate ligament repair." Anest Analg 90, (2000): 129-135.
- Kohrs, Rainer, and Durieux Marcel E. "Ketamine: teaching an old drug new tricks." Anesthesia & Analgesia 87, (1998): 1186-1193.
- Szumita, Paul M, Baroletti Steven A, Anger Kevin E, and Wechsler Michael E. "Sedation and analgesia in the intensive care unit: evaluating the role of dexmedetomidine." Ame J Healt Syst Pharm 64, (2007): 37-44.
Author Info
Department of Anaesthesiology, Pain Medicine and Critical Care, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: U. Juvairiya Banu A Comparison of IV Dexmedetomidine and Midazolam for Sedation in Patients under Neuraxial Anaesthesia, J Res Med Dent Sci, 2021, 9(11): 1 - 4
Received: 01-Dec-2021 Accepted: 15-Dec-2021 Published: 22-Dec-2021
