Research - (2021) Volume 9, Issue 6
A Study of Hemoglobin and Iron Status of Term Neonates Born to Anaemic Mothers
Shiji R, S Jagadeeshwari and S Sundari*
*Correspondence: S Sundari, Department of Paediatrics, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, India, Email:
Abstract
The present study includes the correlation between cord blood hemoglobin, serum iron and serum ferritin levels of term neonates with maternal hemoglobin levels and serum iron and serum ferritin levels of mothers, and to study the correlation between anthropometry and maternal anemia and iron status of neonates. This was a Cross-Sectional Study done on 100 singleton term neonates and their mothers fulfilling the inclusion and exclusion criteria. Maternal venous samples and Cord blood samples were collected for Hemoglobin, Serum Iron and Serum Ferritin estimation. Thorough physical examination of the neonate including anthropometry (Weight, Length, Head Circumference and Chest Circumference) was done. The data collected was analyzed using suitable statistically tests. The Present study focuses on the correlation between cord blood hemoglobin, serum iron and serum ferritin levels of term neonates with maternal hemoglobin levels. The Present study focuses on the correlation between maternal anemia and anthropometry of neonates and study the correlation between the iron status of neonates and anthropometry.
Keywords
Hemoglobin, Serum iron, Schistosomiasis, Placenta, Anthropometry, IREs
Introduction
Iron deficiency anemia is a common problem, especially in developing countries. Anaemia affects nearly half of all the pregnant women in the world, 52% in developing countries compared with 23% in the developed world [1]. The most common cause of anaemia is poor nutrition, deficiencies of iron and other micronutrients, malaria, hookworm disease, and schistosomiasis; HIV infection and hemoglobinopathies are additional factors [2]. Maternal anemia has several deleterious effects on the health of the mother and the foetus. Mounting evidence indicates that maternal iron deficiency in pregnancy reduces fetal iron stores, perhaps well into the first year of life [3]. Iron sufficiency is of paramount importance in the neonatal period. Controversy exists whether the transfer of iron to the foetus from the mother is determined by fetal demands or by maternal iron stores [4].
Iron is transferred to the foetus through the placenta against a concentration gradient, particularly during the later stages of pregnancy, with the amount of iron transported across the placenta increasing as pregnancy progresses. Several factors influence the concentration of Hb at birth, including gestational age, gender, state of maternal iron reserves, and other factors affecting the maternal-fetal iron exchange. In term pregnancies, 70 – 80% of fetal iron is present in the red cells as Hb [5]. Anaemia in new-borns is established with a cord blood Hb concentration <13.0 g/dL. The cut-off cord blood serum ferritin (CSF) indicating an iron deficiency in the Newborn is set at <35.0 mg/L at which point the iron available to meet normal brain requirements is restricted. CSF is, in addition to the direct examination of stainable iron in bone-marrow smears, the most specific indicator of body iron reserves, Iron homeostasis and iron status indicators [6].
Body iron balance are primarily determined by regulation of absorption of iron where the efficiency of absorption is inversely related to the body iron status. This efficiency may vary from as low as 1% to 2% to nearly 50%to 60% depending on the characteristics of the food source of iron and the iron status of the individual [7]. Regulation is exerted at the level of movement both into the cell through the receptor-mediated process as well as moving across the basolateral membrane into the plasma pool. This former process likely involves both the transferrin receptor and the divalent metal transporter proteins while the latter involves the cellular iron exporter, ferroprotein [8]. The amount of ferroprotein expressed on the basolateral membrane is influenced by a hepatocyte secreted signal peptide named hepcidin. As liver iron stores increase, more hepcidin is released into the plasma pool to decrease the ferroprotein mediated movement of gastrointestinal cell iron across the basolateral membrane.
Iron in the plasma pool is transported by transferrin, which has 2 identical binding sites for ferric ions [9]. This iron will be delivered to any cell that expresses the transferrin receptor; the developing red cell mass takes about 80% of this plasma iron turnover daily.
The remaining 20% is distributed to other body iron pools and the excess iron put into storage in the multi - unit protein, ferritin [10]. The amount of transferrin receptor (TfR) located on the cell surface is regulated by cellular iron status through translational regulation involving Iron Response Elements (IREs) on the noncoding regions of the mRNA and rates of recycling of the TfR on and off the membrane. As a cell becomes iron deficient, there is an upregulation of the translation of TfR-mRNA and more receptor is produced; the inverse is seen when the cells are replete with iron [11].
Materials and Methods
Study design
Cross-sectional study.
Study population
We studied the neonates born at Sree Balaji Medical College and Hospital and their mothers.
Sample size
100 singleton term neonates and their mothers.
Study period
One year from April 2017 to March 2018.
Study place
The study was done in the Department of Paediatrics and Department of Obstetrics and Gynecology at Sree Balaji Medical College and Hospital, Chennai.
Inclusion criteria
Singleton term neonates and their mothers.
Exclusion criteria
- Multiple gestations.
- Preterm neonates.
- Premature rupture of membranes (PROM>18 hrs).
- Maternal Fever.
- Foul-smelling liquor.
- Antepartum haemorrhage (APH).
- Pregnancy Induced Hypertension (PIH).
- Eclampsia.
- Gestational Diabetes-mellitus or overt Diabetes complicating pregnancy.
- Mothers with liver disorders, kidney disorders, and other systemic illness.
- Women who have received blood transfusion.
Study method
After obtaining informed consent from the parents, detailed maternal history was recorded in a predesigned proforma. Venous blood sample was collected from the pregnant women during labour. And cord blood sample was collected from the umbilical cord immediately after clamping without milking the cord. The maternal venous and baby’s cord blood samples were collected in Vacutainer blood collection tubes with EDTAK3 as an anticoagulant for estimation of Hemoglobin levels, haematocrit and RBC indices. And in plain vacutainer tubes for estimation of Serum iron and Serum Ferritin levels. A through physical examination of the neonate including anthropometry (Weight, Length, Head Circumference, and Chest Circumference) was done.
Laboratory analysis
Hemoglobin level, haematocrit and RBC indices were calculated using an automated analyser. Serum Iron using calorimetry method and Serum Ferritin using immunohistochemistry. The data collected has been analysed using suitable statistically tests.
Study definition
Mothers with haemoglobin <11gm/dl were defined as anaemic as per WHO cut off. The gestational age of pregnancy was calculated from the last menstrual period and was confirmed by new Ballard score. Term gestation was defined as gestational age between 37 weeks 0 days to 41 weeks 6 days.
BG Prasad scale: The socio-economic classification was measured based on the BG Prasad scale. This scale was introduced in 1961 was later modified in 1982, 2001, 2013 and in 2016. The scale is updated periodically as inflation is an ongoing process. There are tools and calculations available which enables real time update of the classification to maintain the validity of the published data. The revised classification for 2016 is as follows (Table 1).
Table 1: BG Prasad scale.
| Social Class | Amount (Rs. /month) |
|---|---|
| I (upper class) | 6261 and above |
| II (upper middle class) | 3099-6260 |
| III (middle class) | 1835-3098 |
| IV (lower middle class) | 949-1834 |
| V (lower class) | <948 |
Statistical analysis
The statistical analysis was done using the Chi-Square test to find the association between the parity, educational status of the father and mother, occupational status with the severity of anaemia. Differences in mean parameters between the pregnant women and their newborns in the four groups were determined using the analysis of variance. Pearson’s correlation coefficient was used for the correlation analysis. A p-value of <0.05 was significant for all the tests.
Results
100 mothers and their new-borns were enrolled in the study to determine the relationship between them with regard to their iron status. These 100 mothers were divided into 4 groups based on their hemoglobin levels done by automated analyser.
Group 1: Hemoglobin level≤6.9 gm/dl.
Group 2: Hemoglobin level 7g/dl -9.9g/dl.
Group 3: Hemoglobin level 10g/dl -10.9g/dl.
Group 4: Hemoglobin level ≥ 11g/dl.
No of pregnant women in each group
- Group 1 contained 9 mothers and they constituted 09% of the sample size.
- Group 2 contained 61 mothers and they constituted 61% of the sample size.
- Group 3 contained 10 mothers and they constituted 10% of the sample size.
- Group 4 contained 20 mothers and they constituted 20% of the sample size.
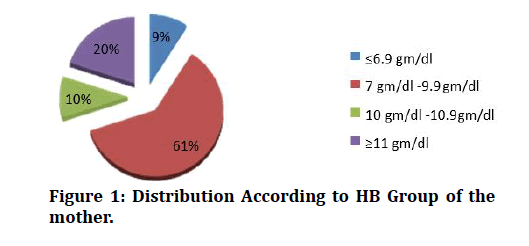
The above table depicts Hemoglobin values of the mothers. Among 100 mothers 9(9%) mothers were in severe anaemia, 61(61%) group were in mild anaemia, 10 (10 %) were in moderate anaemia and 20(20%) mothers were normal. This data shows that 80% of the pregnant mothers were anaemic (Table 2 and Figure 1).
Figure 1: Distribution According to HB Group of the mother.
Table 2: Classification of pregnant women into 4 groups according to the Hemoglobin level.
| Group | Frequency | Percent |
|---|---|---|
| ≤6.9 gm/dl | 9 | 9 |
| 7 gm/dl -9.9 gm/dl | 61 | 61 |
| 10 gm/dl -10.9 gm/dl | 10 | 10 |
| ≥11 gm/dl | 20 | 20 |
| Total | 100 | 100 |
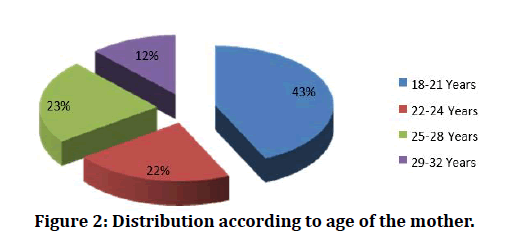
The above table 3 depicts age of the mothers. Among 100 mothers 43(43%) mothers were in 18 -21 age group, 22(22%) were in 22 -24 years, 23 (23 %) were in 25-28 years and only 12(12%) mothers age were 29 -32 years. This data shows that 88% of the pregnant mothers age were 18-28 years (Figure 2).
Figure 2: Distribution according to age of the mother.
Table 3: Age.
| Age group | Frequency | Percent |
|---|---|---|
| 18-21 Years | 43 | 43 |
| 22-24 Years | 22 | 22 |
| 25-28 Years | 23 | 23 |
| 29-32 Years | 12 | 12 |
| Total | 100 | 100 |
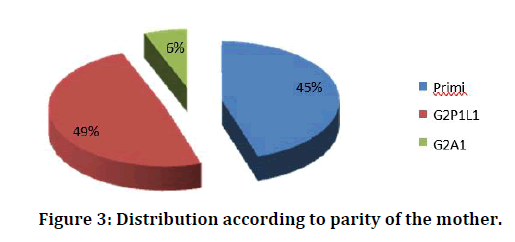
Only primi and second gravida in the age group of 18 to 32 were selected to make the different study groups comparable with respect to age and parity. In the present study group, 45% were primi and they were the major group. Rest of the women were non primi and they constitute 55%. In the group of second gravida women with previous live births were 49% and the women with previous abortions were 6% (Table 4 and Figure 3).
Figure 3: Distribution according to parity of the mother.
Table 4: Parity.
| Parity | Frequency | Percent |
| Primi | 45 | 45 |
| G2P1L1 | 49 | 49 |
| G2A1 | 6 | 6 |
| Total | 100 | 100 |
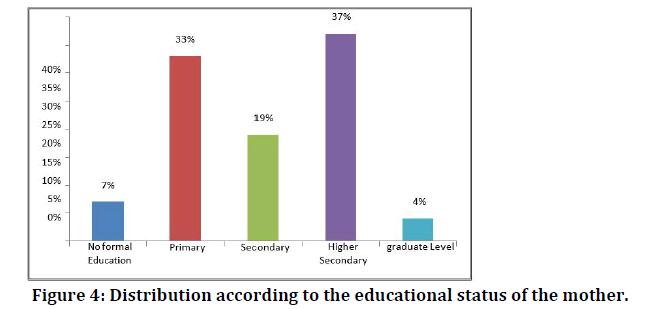
Of the 100 mothers, 7 of them were not able to read and write and 4 of them were degree holders. 33 had education up to the primary school level, 19 up to the secondary level, and 37 mothers were having higher secondary level education (Table 5 and Figure 4).
Figure 4: Distribution according to the educational status of the mother.
Table 5: Classification of mothers according to their educational status.
| Educational status of mother | Frequency | Percent |
|---|---|---|
| No formal Education | 7 | 7 |
| Primary | 33 | 33 |
| Secondary | 19 | 19 |
| Higher Secondary | 37 | 37 |
| graduate Level | 4 | 4 |
| Total | 100 | 100 |
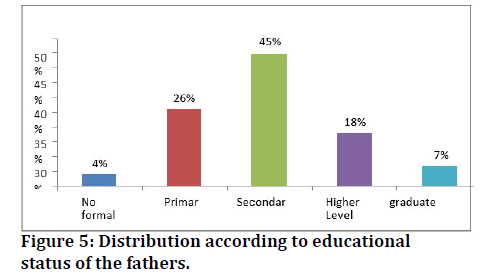
The table 6 depicts educational status of the fathers, among 100 study participants, 4 of the fathers were not able to read and write and 7 of them were degree holders. 26 had education up to the primary school level, 45 up to the secondary level, and 18 fathers were having higher secondary level education (Figure 5).
Figure 5: Distribution according to educational status of the fathers.
Table 6: Classification of fathers according to the educational status.
| Educational status of Father | Frequency | Percent |
|---|---|---|
| No formal Education | 4 | 4 |
| Primary | 26 | 26 |
| Secondary | 45 | 45 |
| Higher Secondary | 18 | 18 |
| graduate Level | 7 | 7 |
| Total | 100 | 100 |
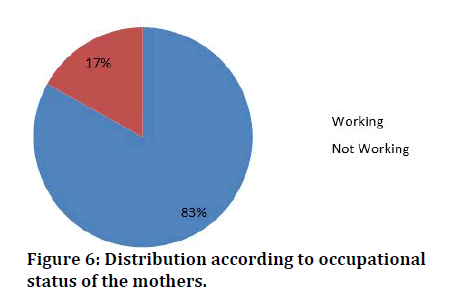
The above table depicts occupation status of the mothers, among 100 study participants, 83 of them were working and remaining 17 % were not working (Table 7 and Figure 6).
Figure 6: Distribution according to occupational status of the mothers.
Table 7: The occupational status.
| Occupation | Frequency | Percent |
|---|---|---|
| Working | 83 | 83 |
| Not Working | 17 | 17 |
| Total | 100 | 100 |
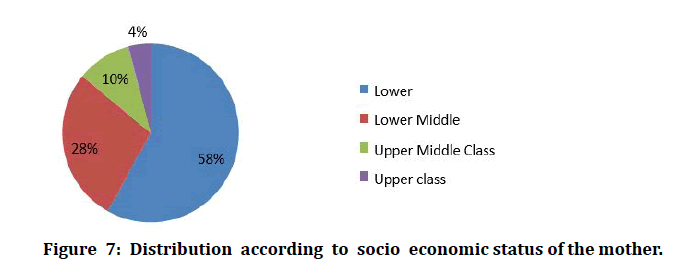
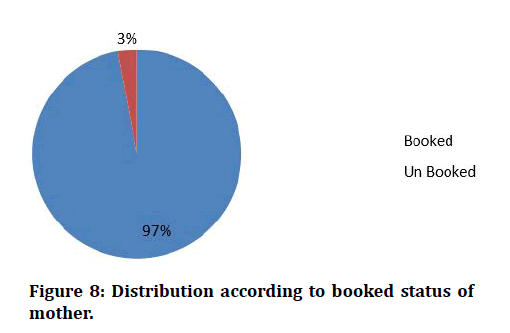
The above table depicts socio economic status of the study participants classified by B.G.Prasad scale, among 100 , majority 58% of them belonged to lower status, 28% were belonged to lower middle class status and remaining 10% ,4% belonged to upper middle class and upper class respectively (Table 8 and Figure 7). Out of the 100 mothers studied, 97 had at least 3 antenatal visits and they are booked cases. Only three women were unbooked (Table 9 and Figure 8).
Figure 7: Distribution according to socio economic status of the mother.
Figure 8: Distribution according to booked status of mother.
Table 8: Classification of pregnant women according to the socio-economic status.
| Socio economic status | Frequency | Percent |
|---|---|---|
| Lower | 58 | 58 |
| Lower Middle | 28 | 28 |
| Upper Middle Class | 10 | 10 |
| Upper class | 4 | 4 |
| Total | 100 | 100 |
Table 9: Classification of pregnant women according to booked or unbooked.
| Booked or not | Frequency | Percent |
|---|---|---|
| Booked | 97 | 97 |
| Un Booked | 3 | 3 |
| Total | 100 | 100 |
Discussion
Iron deficiency anemia is the most common nutritional deficiency disorder affecting the pregnant women in our country with a significant impact on maternal and fetal mortality and morbidity. In the present study, 100 pregnant women were selected based on the inclusion criteria. These 100 mothers were divided into 4 groups:
Group 1: Hemoglobinlevel≤6.9gm/dl.
Group 2: Hemoglobin level 7g/dl -9.9g/dl.
Group 3; Hemoglobin level 10g/dl -10.9g/dl Group 4: Hemoglobin level ≥ 11g/dl.
Number of pregnant women in each group
Group 1 contained 9 mothers and they constituted 09% of the sample size. Group 2 contained 61 mothers and they constituted 61% of the sample size. Group 3 contained 10 mothers and they constituted 10% of the sample size. Group 4 contained mothers and they constituted 20% of the sample size. Among 100 mothers 9(9%) mothers were in severe anemia, 61(61%) were in mild anemia, 10 (10 %) were in moderate anemia and 20(20%) mothers were normal [12-17]. This data shows that 80% of the pregnant mothers were anemic. baby weight of the study participants among 9 severe anemia (<6.9 mg/ dl) mothers 7(78%) had 2.00 -2.5 kilograms baby and remaining 2(22%) were had 2.00 to 2.5 kilograms. Among 61 moderate anemic (<7 -9.9 mg/dl) mothers 10(16%) had 2.00 -2.5 kilograms baby, 30(49%) were had 2.00 to 2.5 kilograms and 21(34%) had above 3.0 kilograms baby. Among 10 mild anemic (<10 -10.9 mg/dl) mothers only 2(20%) had 2.00 -2.5 kilograms baby, 4(40%) were had 2.00 to 2.5 kilograms and 4(40%) had above 3.0 kilograms baby. Among 20 normal (≥11 mg/dl) mothers only 2(10%) had 2.00 -2.5 kilograms baby, 9(45%) were had 2.00 to 2.5 kilograms and 9(45%) had above 3.0 kilograms baby [18-21]. The chi-square value was 20.8 which was statistically highly significant p<0.001 which describe that baby wei ght was higher when mothers Hb was higher [1].
Values of Hemoglobin, serum iron and serum Ferritin in maternal and cord blood
In general, the iron-related parameters are higher in the cord blood, compared to the maternal blood. The present study also corroborates this finding in some of the parameters (22-25). The values of hemoglobin, serum iron and serum ferritin in the cord blood were higher than the maternal blood. Similar results were obtained in the studies conducted by Siddappa A. M et al. The mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration were higher in the cord blood compared to the maternal blood [22-25].
Conclusion
The serum iron and serum ferritin values of the neonates correlate with the maternal hemoglobin levels and serum ferritin is exceptionally low in the neonates of the mothers with severe anemia. Compared with the babies of the mothers with normal hemoglobin levels, the babies of the mothers with severe anemia show a low birth weight. The neonates of both anemic and non-anemic women may be born with a hemoglobin in the normal range but the iron stores of the neonate depend on the maternal iron status. The lower iron and ferritin values of the neonate were not reflected by the neonatal RBC indices; hence they should not be used as an indicator for assessing the iron status. The low iron stores of the neonates may be depleted easily when the demands are high in early infancy. Iron deficiency in early life may have long term adverse effects on the cognitive development and may also impair cellular immunity.
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgements
The encouragement and support from Bharath University, Chennai is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- Gambling L, Danzeisen R, Lea RG. Effect of iron deficiency on placental transfer of iron and expression of iron transport proteins in vivo and in vitro. Biochem J 2010; 883–889.
- Kilbride J, Baker TJ, Parapia LA, et al. Anemia during pregnancy as a risk factor for iron-deficiency anemia in infancy: A case-control study in Jordon. Int J Epidemiol 2009; 28:461–468.
- Ackurt F. Biochemical assessment of nutritional status in pre- and post-natal Turkish women and outcome of pregnancy. European J Clini Nutrition 1995; 49:613–22.
- Adebisi O, Strayhorn G. Anemia in pregnancy and race in the United States: Blacks at risk. Fam Med 2009; 37:655-662.
- Agrawal RM, Tripathi AM, Agarwal KN. Cord blood haemoglobin, iron and ferritin status in maternal anaemia. Acta Paediatr Scand 2012; 72:545-548.
- Al-hilli NM. The effect of maternal anaemia on cord blood haemoglobin & newborn birth weigh karbala. J Med 2010; 2.
- Allen L. Pregnancy and iron deficiency: Unresolved issues. Nutr Rev 2007; 55:91-101.
- Allen LH. Anemia and iron deficiency: Effects on pregnancy outcome. Am J Clin Nutr 2008; 71:1280–1284.
- Allen LH. Biological mechanisms that might underlie iron’s effects on fetal growth and preterm birth. J Nutr 2009; 131:581S-589S.
- Bagchi K, Mohanram M, Reddy V. Humoral immune response in children with irondeficiency anaemia. Br Med J 2010; 280:1249-1251.
- Baker RD, Greer FR. Diagnosis and prevention of iron deficiency and iron-deficiency anemia in infants and young children (0 -3 years of age). Pediatrics 2010; 126:1040-1050.
- Beard JL. Iron biology in immune function, muscle metabolism and neuronal functioning. J Nutr 2012; 131:568S-580S.
- Bhaskaram C, Reddy V. Cell mediated immunity in iron- and vitamin deficient children. Br Med J 2008; 3:522-522.
- Birch EE, Khoury JC, Berseth CL. The impact of early nutrition on incidence of allergic manifestations and common respiratory illnesses in children. J Pediatr 2010; 156:902-906.
- Cantwell RJ. Iron deficiency anaemia of infancy. Clin Pediatr 1972; 11:443-449.
- Chandra RK, Saraya AK. Impaired immuno- competence associated with i ron deficiency. J Pediatr 2016; 86:899-902.
- Chandra RK. Reduced bactericidal capacity of polymorphs in iron deficiency. Arch Dis Child 2013; 48:864-866.
- Chaparro CM. Setting the stage for child health and development: prevention of iron deficiency in early infancy. J Nutr 2008; 138:2529–2533.
- Chirstenson RD. Expected hematologic values for term and preterm neonates In: Cristensen, hematologic problems of the neonatel 11th Eedn, Philadelphia, Saunders 2009; 118-122.
- Cogswell ME. Iron supplementation during pregnancy, anemia, and birth weight: A randomized controlled trial. Am J Clin Nutrition 2003; 78:773–781.
- Cogswell ME, Kettle-Khan L, Ramakrishnan U. Iron supplement use among women in the United States: Science, policy, and practice. J Nutr 2013; 133-135.
- Cook JS, Flowers CH, Skikne BS. The quantitative assessment of body iron. Blood 2013; 101:3359- 3364.
- Cutner A, Bead R, Harding J. Failed response to treat anaemia in pregnancy: reasons and evaluation. J Obstet Gynecol 2016; 23-27
- Dallman P. Iron deficiency and the immune response. Am J Clin Nutr 2018; 46:329-334.
- Dallman PR. Nutritional anemias in childhood: Iron, folate, and vitamin B12. In: Suskind RM, Lewinter - Suskind L, eds. Textbook of Pediatric Nutrition. 2nd ed. New York, NY: Raven Press 2013; 91-105.
Author Info
Shiji R, S Jagadeeshwari and S Sundari*
Department of Paediatrics, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Shiji R, S Jagadeeshwari, S Sundari, A Study of Hemoglobin and Iron Status of Term Neonates Born to Anaemic Mothers, J Res Med Dent Sci, 2021, 9(6): 133-139
Received: 25-Mar-2021 Accepted: 15-Jun-2021 Published: 22-Jun-2021