Research - (2020) Volume 8, Issue 6
Adult Type Granulosa Cell Tumour in a Peri-Menopausal Woman: A Rare Ovarian Malignancy
Mansi Kumar, Bhavika Kalthe, AS Dhillon and Gagandeep Kaur*
*Correspondence: Gagandeep Kaur, Department of Obstetrics and Gynaecology, Maharishi Markandeshwar Deemed to be University, India, Email:
Abstract
Granulosa cell tumors of the ovary are exceedingly rare neoplasms that originate from sex-cord stromal cells. The long natural history of granulosa cell tumors and their tendency to recur years after the initial diagnosis are the most prominent of their characteristics. The secretion of estradiol is the reason for signs at presentation such as vaginal bleeding and precocious puberty. Abdominal pain and hemoperitoneum, which occasionally can occur, are attributable to tumor rupture. The most common finding in pelvic examination is a tumor mass, which is subsequently confirmed with imaging techniques. Surgery is the mainstay of initial management for histological diagnosis, appropriate staging, and debulking. Chemotherapy is the treatment of choice for patients with advanced, recurrent, or metastatic disease, and BEP is the preferred regimen. Although the overall rate of response to treatment is high, the impact of treatment on disease-free or overall survival is unknown. Prolonged surveillance is mandatory because tumors tend to recur years after the initial diagnosis. Here we present a rare case of Granulosa cell tumour in a perimenopausal woman. Massive ascites as the first sign of ovarian granulosa cell tumor (GCT) in a peri-menopausal female is an extremely rare presentation and its clinical features and treatment methods have not been well described yet.
Keywords
GCT (Granulosa Cell Tumour), BEP (bleomycin, etoposide, and cisplatin).
Introduction
Granulosa cell tumour (GCT) of the ovary is an uncommon neoplasm that represents 2% to 5% of all ovarian cancers. As the name implies, GCT is derived from the granulosa cell, a hormonally active component of the ovarian stroma that is responsible for estradiol production. GCT is, therefore, referred to as an ovarian sex cordstromal tumour and it accounts for 70% of the tumours in this category. This neoplasm is divided into adult and juvenile types based on different clinical and histopathologic features. Juvenile GCT represents only 5% of this tumour type and usually occurs in pre-pubertal girls and women younger than 30 years [1-3]. These patients may present with iso-sexual precocious, pseudo puberty or abdominal and pelvic pain owing to a large pelvic mass. Patients with juvenile GCT typically present at an early stage and have a favorable prognosis, although those with more advanced-stage disease may experience an aggressive clinical course [1,3,4]. Although patients with the more common adult form of GCT may also present with early-stage disease, relapses tend to occur in a more indolent fashion, often many years after the original diagnosis. GCT can occur at any age but most commonly presents during the peri-menopausal or early postmenopausal period, with a median age of diagnosis between 50 and 54 years in most series [5-14]. The estimated incidence of GCT in the United States is 0.99/100,000, whereas the reported incidence in other developed countries ranges from 0.4 to 1.7 per 100,000 [7,8,10,11]. Several putative risk factors have been associated with the development of GCT, although most are not reproducible and have unknown clinical or biologic significance. For instance, menopausal status and parity do not seem to influence risk [5,9,11-14]. Other factors such as use of fertility drugs or oral contraceptives are not consistently associated with a higher risk [15-17]. Unlike epithelial ovarian cancer, there seems to be no known inherited predisposition for the development of this tumor. Inherited germline mutations of BRCA1 or BRCA2 have not been associated thus far with a higher risk of developing GCT.
Clinical Report
A 40-year-old woman with amenorrhoea for half a year was referred to our tertiary care center with abdominal distention and heaviness along with breathlessness for a few months. The previous medical history was composed by bronchial asthma and irregular menstrual cycles. On examination, abdomen was distended with a mass approximately 15*15 centimeters in size of firm consistency, mobile with welldefined upper margin arising from the pelvis. Vaginal examination suggested of a bulky mass of 24 weeks of uterine size, mobile, non-tender with fullness of bilateral fornixes. No rectal mucosa involvement was found clinically. The laboratory test including complete blood counts, renal and liver function, CA19-9, CEA, β-HCG, AF, LDH suggested isolated raise of CA-125 (205 IU/L) and serum inhibin (120 pg/ml) levels. Transabdominal ultrasonography pictured a large well- defined complex heterogeneous soft tissue mass with solid and cystic components in the right adnexa measuring approximately 18*12*9 centimeters with solid component showing vascularity without the Right ovary being separately visualized. Minimal free fluid along omental thickening at places. CXR showed right sided pleural effusion. Further CECT- abdomen and pelvis (Figure 1) revealed large lobulated heterogeneously enhancing mass in the lower abdomen likely arising from right adnexa as described suggestive of malignant ovarian lesion with minimal ascites and moderate right sided hydro- pneumothorax. Pleural tap was performed which showed high glucose (97mg/dl) and leukocyte count of 120 cells/cumm but no malignant cells. USG guided FNAC was performed which showed cytological features suggestive of granulosa cell tumour. Therefore, staging laparotomy was performed along with total abdominal hysterectomy, bilateral Salpingo-oophorectomy and infra-colic omentectomy. Intraoperatively, a huge, globular, irregular mass measuring 18*13*5 cm with intact capsule was removed and about 400 ml of ascitic fluid was drained and sent for cytological and biochemical examination. Post-operatively patient did well and was discharged on the 8th postoperative day. Histopathology of the mass confirmed Adult type granulosa cell tumour of stage. Patient was finally given 6 cycles of Chemotherapy with bleomycin, etoposide, and cisplatin regimen (Figures 2-4).
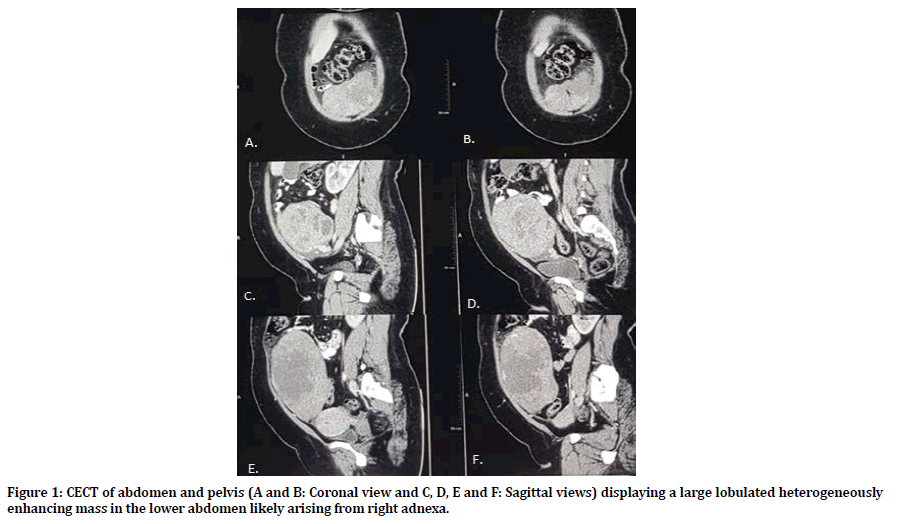
Figure 1. CECT of abdomen and pelvis (A and B: Coronal view and C, D, E and F: Sagittal views) displaying a large lobulated heterogeneously enhancing mass in the lower abdomen likely arising from right adnexa.
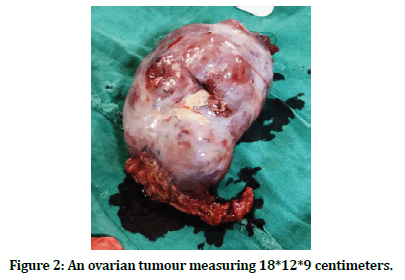
Figure 2. An ovarian tumour measuring 18*12*9 centimeters.
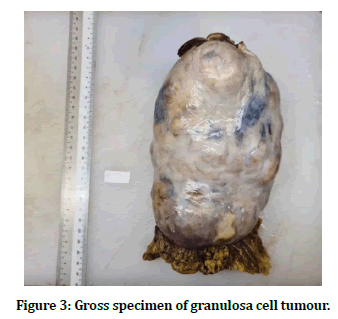
Figure 3. Gross specimen of granulosa cell tumour.
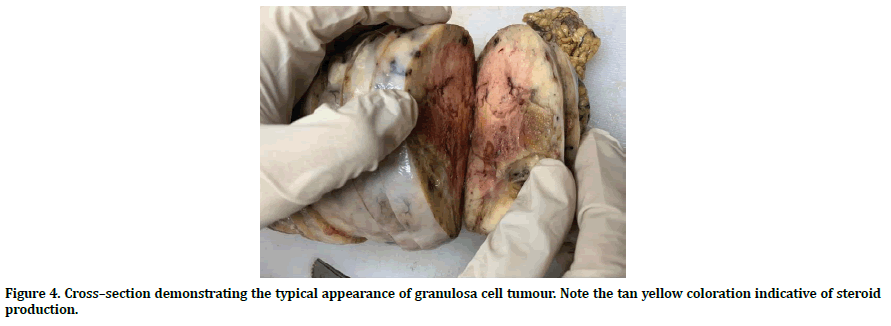
Figure 4. Cross–section demonstrating the typical appearance of granulosa cell tumour. Note the tan yellow coloration indicative of steroid production.
Discussion
Granulosa cell neoplasms were first described by Rokitansky in 1855. It is a rare neoplasm, forming about 2–3% of all ovarian malignancies of which 95% are of the adult type. Amenorrhoea was the most frequent presentation in the reproductive age group, and menorrhagia associated with postmenopausal bleeding was frequent in the older age group. GCTs are rare sex-cord stromal ovarian tumours constituting 5% to 8% of ovarian neoplasms. The patient in our report had typical symptoms of abdominal distention, breathlessness and amenorrhoea with moderate ascites and right sided moderate pleural effusion. Furthermore, her ascites and pleural effusion gradually resolved after the surgery. There is a similar reported case of fibroma with ascites and pleural effusion, which resolve after removal of the tumo [18].
In general, there is no specific tumour marker of GCT. Previous studies have shown that the measurement of serum β-hCG and AFP level was important to exclude secreting germ cell tumours [19], which were normal in the present case. In addition, our case revealed a high level of serum and ascites CA125 that resolved dramatically after removal of tumour suggesting that CA125 level may be a tumour marker to assess for residual or recurrent for malignant epithelial ovarian tumour in a woman with massive ascites [20].
Generally, most of patients with GCTs are diagnosed in stage I level, with a tumour limited to the ovary, and usually have a benign clinical course after simple excision [21]. Our case revealed a well-circumscribed, solid ovarian mass, and ascites by the diagnostic modalities for GCTs, which include CT and ultrasonography scans. Although the morphological features that can be helpful in determining prognosis of GCT were revealed, the golden standard method in GCT diagnosis was histopathological evaluation. In our case, histopathology revealed granulosa cells with nuclear atypia and common mitotic activities as well as foci of Call–Exner bodies, which are diagnostic marker for GCT [22]. Further immune-histochemical evaluation showed positive for Inhibin-A which are most helpful parameters in the diagnosis for GCT [23,24]. CD99 is used to be a marker preferred in differentiation of JGCT from low differentiated carcinomas of ovary, as it reacts with normal sertoli and granulosa cells [25]. Moreover, SMA and vimentin are also immune-histochemical markers confirming the diagnosis of GCT [26,27]. According to the FIGO, the primary management of GCT is unilateral oophorectomy or salpingo-ophorectomy to preserve fertility in stage IA, IB, or IC of the child-bearing women. In treatment of advanced-stages GCT (Stage II-IV), tumour and metastases should be extracted by cytoreductive surgery. In advanced stages of GCT, prognosis is worse, and after surgery, adjuvant chemotherapy is recommended for younger women with resected stage IC to IV GCT and appears to contribute to long-lasting remissions [28,29]. The patients are currently treated with platinum-based chemotherapy and reports on their effectiveness suggest an overall response rate of 63% to 80% [30,31]. In addition, related trials on platinum and taxane combination chemotherapy are also reported [31]. In our case, bilateral salphingo-oophorectomy with total abdominal hysterectomy was performed as our patient had a completed family.
References
- Young RH, Dickersin GR, Scully RE. Juvenile granulosa cell tumors of the ovary: A clinicopathologic anaysis of 125 cases. Am J Surg Pathol 1984; 8:575-596.
- Scully RE. Juvenile granulosa cell tumor. Pediatr Pathol 1988; 8:423-427.
- Calaminus G, Wessalowski R, Harms K, et al. Juvenile granulosa cell tumor of the ovary in children and adolescents: Results from 33 patients registered in a prospective cooperative study. Gynecol Oncol 1997; 65:447-452.
- Powell JL, Johnson NA, Bailey CL, et al. Management of advanced juvenile granulosa cell tumors of the ovary. Gynecol Oncol 1993; 48:119-123.
- Fox H, Agrawal K, Langley FA. A clinicopathologic study of 92 cases of granulosa cell tumor of the ovary with special reference to the factors influencing prognosis. Cancer 1975; 35:231-241.
- Pankratz E, Boyes DA, White GW, et al. Granulosa cell tumors: A clinical review of 61 cases. Obstet Gynecol 1978; 52:718-723.
- Stenwig JT, Hazekamp JT, Beecham JB. Granulosa cell tumors of the ovary: A clinicopathological study of 118 cases with long-term follow up. Gynecol Oncol 1979; 7:136-152.
- Bjorkholm E, Silfversward C Prognostic factors in granulosa cell tumors. Gynecol Oncol 1981; 11:261-274.
- Evans AT, Gaffey TA, Malkasian GD, et al. Clinicopathologic review of 118 granulosa and 82 theca cell tumors. Obstet Gynecol 1980; 55:231-238.
- Ohel G, Kaneti H, Schenker JG. Granulosa cell tumors in Israel: A study of 172 cases. Gynecol Oncol 1983; 15:278-286.
- Malmstrom H, Hogberg T, Risberg B, et al. Granulosa cell tumors of the ovary: Prognostic factors and outcome. Gynecol Oncol 1994; 52:50-55.
- Piura B, Nemet D, Yanai-Inbar I, et al. Granulosa cell tumor of the ovary: A study of 18 cases. J Surg Oncol 1994; 55:71-77.
- Miller BE, Barron BA, Wan JY, et al. Prognostic factors in adult granulosa cell tumors of the ovary. Cancer 1997; 79:1951-1955.
- Savage P, Constenla D, Fisher C, et al. Granulosa cell tumors of the ovary: Demographics, survival, and the management of advanced disease. Clin Oncol 1998; 10:242-245.
- Willemsen W, Kruitwagen R, Bastiaans B, et al. Ovarian stimulation, and granulosa-cell tumor. Lancet 1993; 341:986-988.
- Rossing M, Daling JR, Weiss NS, et al. Ovarian tumors in a cohort of infertile women. N Engl J Med 1994; 331:771-776.
- Centers for Disease Control and Prevention. the National Institute of Child Health and Human Development. The reduction in risk of ovarian cancer associated with oral-contraceptive use. The Cancer and Steroid Hormone Study of the Centers for Disease Control and the National Institute of Child Health and Human Development. N Engl J Med 1987; 316:650-655.
- Kaur H, Bagga R, Saha SC, et al. Juvenile granulosa cell tumor of the ovary presenting with pleural effusion and ascites. Int J Clin Oncol 2009; 14:78–81.
- Malati T. Clinical relevance of AFP, its molecular variants and HCG in hepatoblastoma and childhood germ cell tumors. Clin Biochem 2014; 47:734.
- Zhang P, Wang C, Cheng L, et al. Development of a multi-marker model combining HE4, CA125, progesterone, and estradiol for distinguishing benign from malignant pelvic masses in postmenopausal women. Tumour Biol 2016; 37:2183–2191.
- Schneider DT, Calaminus G, Wessalowski R, et al. Therapy of advanced ovarian juvenile granulosa cell tumors. Klin Padiatr 2002; 214:173–178.
- Ahuja A, Iyer VK, Mathur S, et al. Fine needle aspiration cytology diagnosis of metastatic adult granulosa cell tumour showing call-exner bodies. Cytopathology 2013; 24:346–347.
- Haltia UM, Hallamaa M, Tapper J, et al. Roles of human epididymis protein 4, carbohydrate antigen 125, inhibin B and anti-Müllerian hormone in the differential diagnosis and follow-up of ovarian granulosa cell tumors. Gynecol Oncol 2017; 44:83–89.
- Deavers MT, Malpica A, Liu J, et al. Ovarian sex cord-stromal tumors: An immunohistochemical study including a comparison of calretinin and inhibin. Mod Pathol 2003; 16:584–590.
- Jarboe EA, Hirschowitz SL, Geiersbach KB, et al. Juvenile granulosa cell tumors: Immunoreactivity for CD99 and Fli-1 and EWSR1 translocation status: A study of 11 cases. Int J Gynecol Pathol 2014; 33:11–15.
- Kabbani W, Deavers MT, Malpica A, et al. Uterine tumor resembling ovarian sex-cord tumor: Report of a case mimicking cervical adenocarcinoma. Int J Gynecol Pathol 2003; 22:297–302.
- Elbachiri M, Taleb A, Derrabi N, et al. Adult-type granulosa cell tumor of the testis: Report of a case and review of literature. Pan Afr Med J 2017; 26:198.
- Powell JL, Kotwall CA, Shiro BC. Fertility-sparing surgery for advanced juvenile granulosa cell tumor of the ovary. J Pediatr Adolesc Gynecol 2014; 27:89–92.
- Meisel JL, Hyman D, Jotwani A, et al. The role of systemic chemotherapy in the management of granulosa cell tumors. Gynecol Oncol 2015; 136:505–511.
- Park JY, Jin KL, Kim DY, et al. Surgical staging and adjuvant chemotherapy in the management of patients with adult granulosa cell tumors of the ovary. Gynecol Oncol 2012; 125:80–86.
- Graziani SR, Vital CG, Morikawa AT, et al. Phase II study of paclitaxel associated with lipid core nanoparticles (LDE) as third-line treatment of patients with epithelial ovarian carcinoma. Med Oncol 2017; 34:151.
Author Info
Mansi Kumar, Bhavika Kalthe, AS Dhillon and Gagandeep Kaur*
Department of Obstetrics and Gynaecology, Maharishi Markandeshwar Deemed to be University, Mullana, Ambala, Haryana, IndiaCitation: Mansi Kumar, Bhavika Kalthe, A.S. Dhillon, Gagandeep Kaur, Adult Type Granulosa Cell Tumour in a Peri-Menopausal Woman: A Rare Ovarian Malignancy, J Res Med Dent Sci, 2020, 8 (6): 200-204.
Received: 31-Aug-2020 Accepted: 21-Sep-2020
