Research Article - (2021) Volume 9, Issue 12
Awareness about Diabetic Microangiopathy among Dental Practitioners
Dhanraj ganapathy1*, Ramya Ramadoss2 and Martina Catherine3
*Correspondence: Dhanraj ganapathy, Department of Prosthodontics, Saveetha Institute of Medical and Technical Sciences, Chennai, India, Email:
Abstract
Introduction: Diabetic microangiopathy is distinguished by abnormal development and permeability of small blood vessels, culminating in local edema and impaired function of dependent tissues. Microcirculation dysfunction in diabetes is caused by a variety of mechanisms that are still completely unknown.
Aim: The purpose of the survey was for assessing the awareness about diabetic microangiopathy among dental practitioners.
Materials and Method: A cross-sectional survey was performed with a self-designed questionnaire with ten questions circulated amongst 100 dental practitioners. The questionnaire assessed the awareness about diabetic microangiopathy, the molecular mechanism of action, pathological pathways of diabetic microangiopathy, advanced glycation end products, vascular pericytes, the role of hyperosmolar stress, and management of diabetic microangiopathy. The responses were recorded, tabulated, and analyzed.
Results: 87% of the respondents were aware of diabetic microangiopathy, 5% were aware of the pathological mechanism of diabetic microangiopathy, 5% were aware of the advanced glycation endproducts, 4% were aware of the role of pericytes in diabetic microangiopathy, 3% were aware of the role of hyperosmolar stress in .diabetic microangiopathy and 97% were aware of effects of diabetes on oral diseases. The awareness levels about diabetic microangiopathy were similar among the general dental practitioners and specialists with no statistically significant differences (p>0.05; Pearson's chi-square test)
Conclusion: Awareness about diabetic microangiopathy and the effect of diabetes on oral diseases was high among dental practitioners. However, the knowledge and awareness about the mechanisms and management strategies for diabetic microangiopathy were inadequate. Hence more intense awareness and continuing education programs should be initiated rigorously to enhance better understanding of diabetic microangiopathy amongst dental practitioners.
Keywords
Awareness, diabetes, microangiopathy, dental practitioners, oral diseasesIntroduction
Diabetic microangiopathy is distinguished by abnormal development and permeability of small blood vessels, culminating in local edema and impaired function of dependent tissues. Microcirculation dysfunction in diabetes is caused by a variety of mechanisms that are still completely unknown. Dysregulated vascular regeneration, on the other hand, appears to play a major role. Furthermore, oxidative and hyperosmolar stress, along with induction of inflammatory pathways mediated by advanced glycation end-products, have been identified as important underlying processes. Hyperglycemia, a common symptom of diabetes, is understood to worsen the macrovascular disease, but it's also thought to be the main trigger for the initiation of microvascular diseases, which are now the renowned reason of blindness (retinopathy), end-stage renal failure (kidney disease), diabetic cardiomyopathy, and peripheral neuropathy. Oxidative stress generated by excessive glucose is one mechanism, possibly an important final common pathway, via which hyperglycemia induces or aggravates micro and macrovascular damage. It causes endothelial injury by activating the protein kinase C (PKC)-diacylglycerol (DAG) and hexosamine pathways, as well as prompting the polyol pathway, the formation of advanced glycation end products (AGEs), and the initiation of the protein kinase C (PKC)-diacylglycerol (DAG) and hexosamine mechanisms. Diabetic microvascular disease is marked by aberrant microcirculatory vessel development and permeability. The microvascular bed's smallest functional units include arterioles, capillaries, and venules. Microvessels, unlike macro vessels, which have a specific purpose in delivering oxygen and micronutrients to the microcirculation in all organs and tissues, have specialized responsibilities in oxygen and micronutrients distribution. Main micro-circulatory aspects include permeability to small molecules, modulation of physical dimensions, and functional qualities of the basement membrane, which vary considerably in microvascular beds such as the glomeruli, retina, myocardium, skin, and muscle [1]. Podocyte and pericyte apoptosis are distinct alterations in diabetic microvessels that occur in the kidney and retina, respectively. Diabetes causes endothelial cell damage and failure, which contributes to the loss of microvascular repair mechanisms. Various cell populations dedicated to endothelium repair and angiogenesis have been shown to maintain the integrity of the vascular wall over the last decade . These microvascular alterations can have a negative impact on the oral mucosa and periodontal tissues, aggravating a variety of oral diseases. Hence this study was formulated to assess the awareness about diabetic microangiopathy among dental practitioners.
Materials and Method
A cross-sectional survey was performed with a selfdesigned questionnaire with ten questions circulated amongst 100 dental practitioners selected in Chennai city through a simple random sampling method. Among the enrolled participants 68 were general dental practitioners and 32 were specialists. The validity and reliability of the questionnaire were ensured. The after obtaining informed consent. The questionnaire assessed the awareness about diabetic microangiopathy, the molecular mechanism of action, pathological pathways of diabetic microangiopathy .advanced glycation end products endothelial progenitor cells, vascular pericytes role of hyperosmolar stress, and management of diabetic microangiopathy. The responses were recorded, tabulated, and analyzed.Throughout the study, the confidentiality of the study participants' information was preserved by making the participants' information anonymous and requesting them to offer honest answers. Prior to participation, each subject gave their informed consent. The research was carried out in compliance with the Helsinki Declaration, and the Ethics Committee and Institutional Review Board of the institution gave their approval [2]. Descriptive statistics were used and results were expressed as percentages
Results
Among the respondents who participated in this study, 87% were aware of diabetic microangiopathy, 5% were aware of the pathological mechanism of diabetic microangiopathy, 5% were aware of the advanced glycation endproducts, 4% were aware of the role of pericytes in diabetic microangiopathy, 3% were aware of the role of hyperosmolar stress in .diabetic microangiopathy and 97% were aware of effects of diabetes on oral diseases.
The awareness levels about diabetic microangiopathy were similar among the general dental practitioners and specialists with no statistically significant differences (p>0.05; Pearson's chi-square test) (Figures 1-6)(Table 1)
Table 1: Responses of the participants on awareness about Diabetic microangiopathyGDP-General Dental Practitioner SP-Specialists.
| Awareness levels of participants(N=100,GDP=68,SP=32) | Affirmative responses | Negative responses | P value (Pearson's Chi square test for GDP and SP) |
|---|---|---|---|
| Awareness about Diabetic microangiopathy | 0.87 | 0.13 | >0.05 |
| Awareness about pathological mechanisms of Diabetic microangiopathy | 0.05 | 0.95 | >0.05 |
| Awareness about advanced glycation endproducts | 0.05 | 0.95 | >0.05 |
| Awareness about role of pericytes | 0.03 | 0.97 | >0.05 |
| Awareness about role of hyper osmolar stress in diabetic microangiopathy | 0.03 | 0.97 | >0.05 |
| Awareness about the effects of diabetes on oral diseases . | 0.97 | 0.03 | >0.05 |
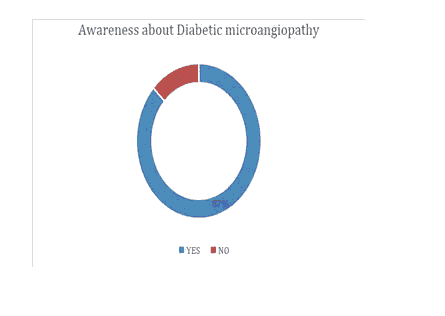
Figure 1: Awareness about Diabetic microangiopathy.
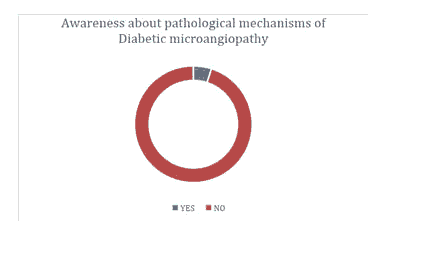
Figure 2:Awareness about pathological mechanisms of Diabetic microangiopathy.
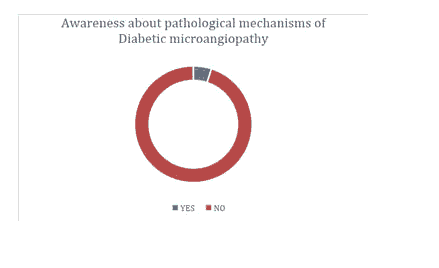
Figure 3:Awareness about advanced glycation end products.
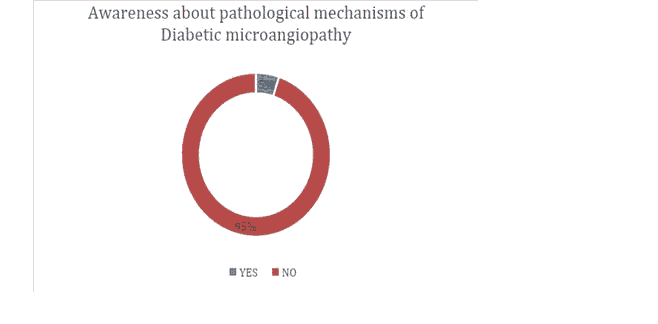
Figure 4: Awareness about role of pericytes.
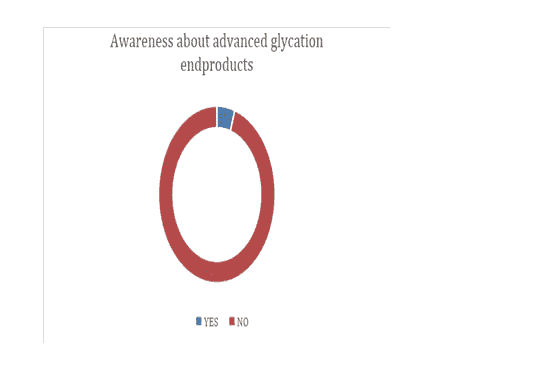
Figure 5:Awareness about role of hyper osmolar stress in diabetic microangiopathy.
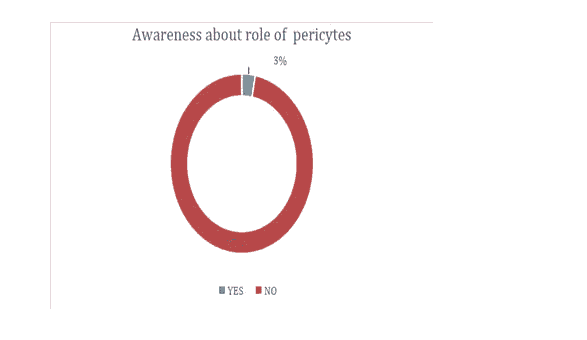
Figure 6:Awareness about the effects of diabetes on oral diseases.
Discussion
Various studies have revealed that excessive glucose exposure causes a range of biochemical, morphological, and functional alterations in mature vascular endothelial cells [3]. The biochemical alterations aggregation of advanced glycation end-products (AGEs); the increased output of the procoagulant protein von Willebrand Factor (VWF); enhanced apoptosis induced by elevated oxidative stress boosted intracellular Ca2+; mitochondrial dysfunction; changes in intracellular lipid metabolism; amplification of the mitogen-activated protein kinase (MAPK) signaling pathway; initiation of the mitogen-activated protein kinase.The structural changes are increased production of extracellular matrix proteins, collagen, and fibronectin, as well as associated enzymes (matrix metalloproteinases, MMPs.The functional alterations are decreased cell proliferation and migration; impairment of endothelium-dependent vasodilation decreased production of vasodilators, increased production of vasoconstrictors; promotion of ischemia, and neo-angiogenesis. Investigators have recently focused on vascular pericytes as essential regulators of vascular development, stability, and repair, making them prospective therapeutic targets. Diabetes causes a significant change in pericyte abundance, function, and recruitment, which leads to microvascular injury in the retina, kidney, and heart. The selective degradation of pericytes in capillary vessels is an early histopathologic characteristic of diabetic microangiopathy. Pericytes in diabetes patients' retinas retreat as a result of faster apoptosis, resulting in increased vascular permeability and retinal edema [4]. When pericytes are lost, a disorderly capillarization is activated, resulting in acellular capillaries and microaneurysms, which cause areas of under perfusion in the dependent tissues. The production of AGEs and the tumor necrosis factor are two mechanisms underlying pericyte death. One of the most prominent mechanisms involved in the pathophysiology of micro-and macrovascular complications of diabetes is the increased formation of AGEs via non-enzymatic glycation and glycoxidation pathways. The intracellular accumulation of AGEs, as well as their interaction with related receptors, RAGEs, alters cytoplasmic and transcription nuclear factors, notably proteins involved in gene transcription control. AGEs also induce strong cross-links to form with collagen, resulting in chemical and biophysical alterations in collagen structure and functional changes such as thickening of the basement membrane and high resistance to proteolytic degradation. Furthermore, regardless of the level of hyperglycemia, AGE-related intracellular glycation of mitochondrial respiratory chain proteins produces more abundant ROS, which increases the development of AGE. Excessive AGE generation causes microvessel thickening, hypertension, endothelial dysfunction, pericyte loss, platelet survival reduction, and platelet aggregation. All of these anomalies could lead to a procoagulant state, which might lead to ischemia and the production of growth factors, which might lead to angiogenesis and neovascularization. Hyperglycemia's hyperosmolar component is a key biophysical modulator of diabetic vascular disease. Hyperglycemia induces an increase in plasma osmolarity, which induces osmotic efflux of water, that decreases the intracellular volume, and encourages adaptive mechanisms such as the activation of hyperosmolarity-responsive genes implicated in glucotoxicity and vascular injury. However, in people, the most effective therapy for preventing microvascular consequences of diabetes is still intense hyperglycemia treatment or improved glycol metabolic management. While the effects of glycemic management on macrovascular consequences are still unknown, its involvement in reducing the risk of retinopathy is clear. At this time, the most important goal is to maintain stricter glycemic control in diabetes patients. This step should be implemented as soon as possible in the disease's natural history, as it is presently the sole way to prevent and delay the course of diabetic microangiopathy. Several new kinds of antihyperglycemic medicines have become available in the recent two decades. Incretins (glucagon-like peptide-1 receptor agonists), DPP4 inhibitors, sodium-glucose cotransporter-2 (SGLT-2) inhibitors, thiazolidinediones, and alpha-glucosidase inhibitors are among them. These drugs can be used alone or in combination with more well-known drugs and insulin. Diabetes research has been going on for years, examining the many clinical consequences of this common condition. These include, as previously indicated, the necessity for periodontal control, as tissue deterioration may be hastened in diabetics, and early treatment of mouth infection will avoid increasing the metabolic imbalance. It has been discovered that those with uncontrolled diabetes have a higher risk of infection as well as abnormally long healing times, putting their oral health at risk. Periodontal Disease (PD), the sixth consequence of diabetes, is the most common dental issue linked to the disease. In patients with periodontal disease-causing endotoxemia or bacteremia, simple chewing can cause systemic dissemination of periodontal pathogens and their metabolic products, increasing serum levels of inflammatory mediators such as interleukin 6 (IL-6), fibrinogen, and C-reactive protein (CRP). In addition, systemic inflammation might exacerbate insulin resistance and, as a result, diabetes management. As a result, proper periodontal treatment can help improve glycemic management by lowering the level of proinflammatory mediators [5]. Diabetic patients' dental pulp may have reduced dental collateral circulation, a weakened immune response, and a higher risk of infection or pulp necrosis. In terms of molecular pathophysiology, hyperglycemia stimulates bone resorption, inhibits osteoblast development, and reduces bone healing capability. The elimination of periodontal inflammation has been shown to minimize the amount of insulin required for glycemic control in patients. As a result, it's critical to get rid of any tooth pulp infections. Because of the unique characteristics of periapical lesions in diabetic patients, treatment goals and definitions of success should be tailored to these patients. Mucosal diseases are more common in diabetics, probably due to persistent immunosuppression, delayed healing, and/or salivary hypofunction. The following changes have been made: Oral candidiasis, fissured tongue, irritant fibroma, traumatic ulcers, and lichen planus are examples of oral fungal infections. When it comes to the dentist's role in these methods, keep in mind that diabetes is a frequent disease with oral symptoms that can change dental care requirements. Dentists must be well informed with diagnosis and prevention measures in this situation. The patient, the doctor, the dentist, and other healthcare providers must work together to effectively manage diabetes patients. Dentists will be able to anticipate patient needs and engage competently with other healthcare specialists if they have regular check-ups. A thorough examination of the mouth cavity may reveal signs of a systemic illness, allowing for early diagnosis and treatment. As a result, doctors and dentists must be on the lookout for the numerous oral signs of diabetes to make an early diagnosis. To provide the best treatment for diabetic patients, dentists must have a thorough grasp of the pathogenesis, symptoms, and therapy of many forms of diabetes-related orofacial infection.
Conclusion
Awareness about diabetic microangiopathy and the effect of diabetes on oral diseases was high among dental practitioners. However, the knowledge and awareness about the mechanisms and management strategies for diabetic microangiopathy were inadequate. Hence more intense awareness and continuing education programs should be initiated rigorously to enhance a better understanding of diabetic microangiopathy amongst dental practitioners. Also, this survey opens new avenues for exploring newer medications for providing therapeutic benefits.
References
- Madonna R, Balistreri CR, Geng Y-J, et al. Diabetic microangiopathy: Pathogenetic insights and novel therapeutic approaches. Vascul Pharmacol 2017;90:1–7.
- Madonna R, Balistreri CR, Geng Y-J, et al. Diabetic microangiopathy: Pathogenetic insights and novel therapeutic approaches. Vascul Pharmacol 2017;90:1–7.
- Ganapathy D, Shanmugam R, Sekar D, et al. Current Status of Nanoparticles Loaded Medication in the Management of Diabetic Retinopathy . J Evol Med Dent Sci 2020;9 : 1713–1718.
- Usuelli V, La Rocca E. Novel therapeutic approaches for diabetic nephropathy and retinopathy Pharmacol Res. 2015; 98: 39–44.
- Marmor MF. Mechanisms of fluid accumulation in retinal edema. Macular Edema 2000:35-45.
Author Info
Dhanraj ganapathy1*, Ramya Ramadoss2 and Martina Catherine3
1Department of Prosthodontics, Saveetha Institute of Medical and Technical Sciences, Chennai, India2Department of Oral Biology, Saveetha Institute of Medical and Technical Sciences, Chennai, India
3Department of Prosthodontics, Saveetha Institute of Medical and Technical Sciences, Chennai, India
Citation: Dhanraj ganapathy*, Ramya Ramadoss, Martina Catherine, Awareness about Diabetic Microangiopathy among Dental Practitioners, J Res Med Dent Sci, 2021, 9(11): 161-167.
Received: 01-Dec-2021 Accepted: 15-Dec-2021 Published: 22-Dec-2021
