Research - (2021) Volume 9, Issue 4
Clinico Pathological Profiles of Patients with Proximal Versus Distal Gastric Adenocarcinoma
Ashwini Krishnamoorthy and K Kuberan*
*Correspondence: K Kuberan, Department of General Surgery, Sree Balaji Medical College & Hospital, Bharath Institute of Higher Education and Research, India, Email:
Abstract
One of the most striking epidemiologic observations has been the increasing incidence of adenocarcinomas involving the proximal stomach and distal esophagus. This trend is worrisome, because proximal gastric cancers are thought to have a poorer prognosis, stage, compared with distal cancers. Risk factors are Elevated body mass index, caloric intake, Gastroesophageal reflux disease and smoking. Stomach cancer occurs at a higher incidence in men than in women (ratio of approximately 2:1). The incidence increases with age, peaking in the seventh decade. Comparison of clinicopathological profiles of patients with proximal and distal gastric adenocarcinomas.
Keywords
Gastroesophageal, Adenocarcinomas, Distal gastric adenocarcinomas
Introduction
Adenocarcinoma of the stomach has been a leading cause of cancer death worldwide through most of the twentieth century. The incidence of this disease has gradually decreased in many parts of the world, due to changes in diet and other environmental factors. With the exception of just a few countries, the prognosis for this disease in the world remains poor. The explanations for these poor results are multifactorial. The lack of well-defined risk factors and specific symptomatology, and the relatively low incidence have contributed to the late stage of onset seen in our country specifically. In Japan, where gastric cancer is endemic, patients are diagnosed at a very early stage, which is reflected in the excellent 50% 5 year survival rate.
Although the incidence of gastric cancer has decreased dramatically over last century, the decline has been limited to cancers below the gastric cardia. The number of newly diagnosed cases of proximal gastric and gastro-esophageal junction adenocarcinomas has increased markedly since the mid-1980s.The disturbing fact is that these are thought to be biologically more aggressive and more complex to treat than distal tumors [1-35].
Materials and Methods
This is a prospective study of patients with Gastric Adenocarcinomas who were admitted in the Department of General Surgery, Sree Balaji Medical College & hospital, Chennai, during the period between July 2016 and July 2018. The patients’ history of presenting illness and past history of ulcer treatment as well as gastric surgery was noted. Their personal history was noted, which included their socioeconomic status, habit of smoking, alcohol consumption, diet. The weight and height of all patients were noted to calculate the body mass index (weight in kg / height squared in meters). Their blood groups were noted.
All patients included in this study underwent upper gastrointestinal scopy and an imaging study in the form of a contrast enhanced CT scan of the abdomen. During the endoscopy a minimum of seven biopsies were taken from the lesion and if an ulcer was found, biopsies around the ulcer crater were taken. Histopathological examination was then performed on the biopsies. Carcinomas of the gastric cardia were defined as: lesions with the center located within 1 cm proximal and 2 cm distal to the esophagogastricjunction. According to Siewert: type II was “real” gastric cardia carcinoma and type III was carcinoma located more than 2 cm below the esophagogastric junction (up to a maximum of 5 cm).
Proximal gastric adenocarcinomas included type II tumors, type III tumors, and other tumors up to the distal limit of the proximal third of stomach. The tumors located distal to the limit of the proximal third and up to the pylorus were included as distal gastric adenocarcinomas. All tumors were classified based on intra-operative information or pathology report into different types. The TNM categories were defined based on the 1977 classification, and the residual tumor status -R category, was determined intraoperatively and histopathologically. The standard treatment for proximal gastric carcinoma was total gastrectomy with D2 Lymphadenectomy, omentectmoy, with splenectomy. The standard treatment for distal gastric adenoacarcinoma was subtotal gastrectomy with D2 lymphadenectomy and omentectomy.
Demographic and clinical variables that are qualitative are given in frequencies with their percentage. Clinical variables which are quantitative are given in mean and standard deviation. Qualitative variables are analyzed using the Pearson chi-square test and Yates corrected Chi square test. Quantitative variables are analyzed using student independent t-test. P value less than 0. 05 are taken as significant.
Results
Patient demographics and history
In our study the male to female ratio was 1.95:1 overall, whereas for proximal tumours it was 1.5:1 and for distal tumors it was 2.12:1 (Figure 1).
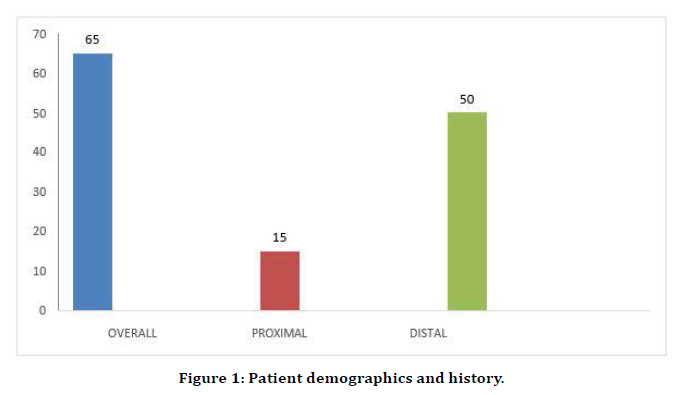
Figure 1. Patient demographics and history.
Male: Female ratio
In our study the average age of the patients was 57. 03 years, with a range of 36 to 72 years. The average age for proximal gastric adenocarcinomas was 53.93 years and for distal gastric adenocarcinomas was 57.98 years; P Value was 0.39, which was not significant (Figure 2).
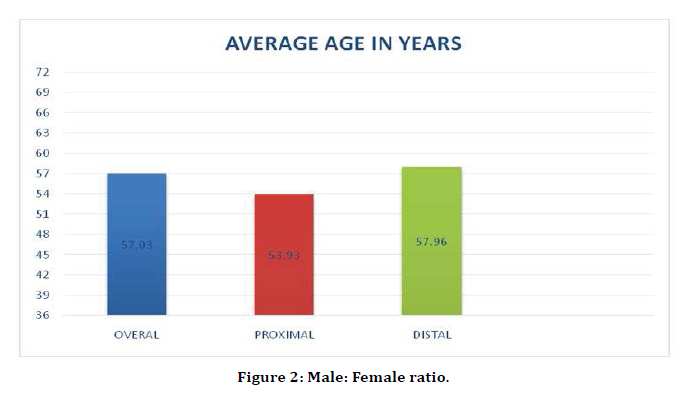
Figure 2. Male: Female ratio.
Body mass index (BMI)
Body mass index was 20.57 overall, whereas it was 20.70 and 20.53 for proximal and distal adenomacarcinoma respectively. The P value was 0.93, which was insignificant. Our findings differed from the findings of other studies where proximal tumors were associate with a higher BMI. This is probably because of the small number of patients in our study (Table 1).
| Overall | Proximal | Distal | Chi-square | P-value | |
|---|---|---|---|---|---|
| BMI | 20.57 ± 6.2 | 20.7 ± 6.8 | 20.53 ± 5.0 | 0.09 | 0.93 |
Table 1: Body mass Index (BMI).
Blood group
In our study we did not find any predilection for a particular blood group. P value was 0.21 which was not significant. But it has been proven, persons with blood group A are more at risk gastric adenocarcinomas (Table 2).
| Blood group | Overall | Proximal | Distal | Chi-square | P-value |
|---|---|---|---|---|---|
| A | 14 | 6 | 8 | 4.44 | 0.21 |
| B | 14 | 2 | 12 | ||
| AB | 10 | 1 | 9 | ||
| O | 27 | 7 |
Table 2: Blood group.
Socioeconomic status
In our study 49 patients (75.38%) were from the lower socioeconomic strata. The P value was 0.02, which was significant. This is due to the difficult access to healthcare, more H. Pylori infections, lack of knowledge about symptoms, and because our hospital generally attracts and provides healthcare facilities to the poor (Table 3).
| Socioeconomic status | Overall | Proximal | Distal | Chi-square | P-value |
|---|---|---|---|---|---|
| Low | 49 | 9 | 40 | 2.52 | 0.02 |
| Middle | 14 | 4 | 10 | ||
| High | 2 | 2 | 0 |
Table 3: Socioeconomic status.
Diet
In our study, a higher number of patients (42; 64.61%) consumed a non-vegetarian diet. Vegetarian diet was taken by a smaller percentage (23, 35.38%) of patients, but the difference was not significant, as reflected in the P value of 0.77.
Smoking and alcohol consumption
In our study, 32(49.23%) patients were chronic smokers, and 16 (24.61%) patients were regular alcohol consumers. The P values were 0.71 and 0.89 respectively, which was insignificant.
Previous ulcer treatment
In our study, there were 22 (33.84%) patients who had taken treatment for ulcer before being diagnosed with gastric adenocarcinomas. Out of these, 6 (27.27%) patients had proximal lesions and 16(72.72%) patients had distal lesions. The P value was 0.79 which was not significant (Table 4).
| Overall | Proximal | Distal | Chi-square | P-value | |
|---|---|---|---|---|---|
| Ulcer treatment | 22 | 6 | 16 | 0.07 | 0.79 |
Table 4: Previous ulcer treatment.
Previous gastric surgery
In our study, 6 (9.2%) patients out of 65 had undergone a gastric surgery previously. The surgery was truncal vagotomy with gastrojejunostomy in all the 6 cases. All 6 patients had distal adenocarcinomas. None of the patients in our study had a family history of gastric cancer.
Clinical presentation symptoms
In our study, 60 (92.30%) patients had dyspepsia; out of this 14 (23.33%) patients had proximal lesions and 46(76.66%) had distal lesions. The P value was 0.70 which was not significant. Out of the 63 (96.92%) patients who had anorexia, 14 (22.22%) patients had proximal lesions and 49(77.77%) patients had distal lesions. The P value was 0.52, which was not significant. In our study weight loss documented in 47 (72.30%) patients overall, 13 (27.65%) of which had proximal lesions, and 34 (72.34%) had distal lesions. The P value was 0.28, which was insignificant. Dysphagia was seen in 12 (18.46%) patients overall; all of who had proximal lesions. This was a very significant finding with P value of 0.001. This finding suggests that proximal lesions present with Dysphagia much more often than the distal tumors do. Persistent vomiting was found in 9 (13.8%) patients overall and all these patients had distal lesions, despite which the P value was 0.18. This was not significant. 31 (47.69%) of the patients presented with melena as a symptom. Out of these 4 (12.9%) had proximal lesions, and 27 (87.09%) had distal lesions which was not significant statistically (P value-0.12). Jaundice was seen in two patients one each from either group (Table 5).
| Overall | Proximal | Distal | Chi-square | P-value | |
|---|---|---|---|---|---|
| Dyspepsia | 60 | 14 | 46 | 0.15 | 0.7 |
| Anorexia | 63 | 14 | 49 | 0.4 | 0.52 |
| Weight loss | 47 | 13 | 34 | 1.18 | 0.28 |
| Dysphagia | 12 | 12 | 0 | 43.8 | 0.001 |
| Vomiting | 9 | 0 | 9 | 1.81 | 0.18 |
| Malena | 31 | 4 | 27 | 2.45 | 0.12 |
| Jaundice | 2 | 1 | 1 | 0 | 1 |
Table 5: Previous gastric surgery.
Signs
In our study, 44(67.69%) patients had anemia, out of which 10 (22.72%) had proximal lesions and 34 (72.27%) had distal lesions. P value was insignificant at 0.92. Mass abdomen was found in 26 (40%) of the patients overall, of which 7 (26.92%) had proximal tumors and 19 (73.07%0 had distal tumors. P value was 0.54, which was not significant. In our study, Hepatomegaly was found in 10 (15.38%) patients, 5 (50%) of these patients had proximal adenocarcinomas and 5 (50%) had distal tumors. P value was found to be 0.03, which was significant. This implies that proximal tumors have Hepatomegaly more often than the distal tumors. In our study, ascites was found in 7 patients overall and supraclavicular nodes were found in 6 patients overall. The difference in the incidence of these signs between proximal and distal tumors was not significant statistically (Table 6).
| Overall | Proximal | Distal | Chi-square | P-value | |
|---|---|---|---|---|---|
| Anemia | 44 | 10 | 34 | 0.05 | 0.92 |
| Mass abdomen | 26 | 7 | 19 | 0.36 | 0.54 |
| Hepatomegaly | 10 | 5 | 5 | 4.83 | 0.03 |
| Ascites | 7 | 2 | 5 | 0.01 | 0.91 |
| Supraclavicularnode | 6 | 0 | 6 | 0.18 | 0.36 |
Table 6: Signs.
Upper gastrointestinal endoscopy
In our study, we found that the average size of the tumor was 3.67 cm overall, with a range of 2.5 cm to 6.0 cm. In proximal tumors, the average size was 3.70 cm, while in distal tumors it was 3.67 cm (Table 7 and Figure 3).
| Total | Proximal | Distal | |
|---|---|---|---|
| Size (in cm) | 3.67 | 3.7 | 3.67 |
| Location | 65 | 15 | 50 |
Table 7: Upper gastrointestinal endoscopy.
Figure 3. Antral carcinoma endoscopic view.
Out of the 65 patients studied, 15 ( 23.07%) patients had proximal tumors and 50 (76.92%) patients had distal tumors. This finding implies that the incidence of proximal gastric adenocarcinomas is not increasing in our population, whereas in the Western hemisphere there is an alarming rise in proximal gastric adenocarcinomas to an extent that these tumors from about 45% of the total gastric adenocarcinomas (Figure 4).
Figure 4. Adenocarcinoma of lesser cuvature endoscopic view.
The morphology of the tumor on upper GI scopy was described as ulcerating, polypoid or superficial. Overall 21 (32.30%) were ulcerating growths; of which 2 (9.5%) were located proximally and 19 (90.47%) were located distally. 33 (50.76%) were polypoid growths, of which 12 (36.36 %) were located proximally, and 21 (63.63%) were located distally. Out of the 11 (16.92%) superficial growths 1 (9.09%) was located proximallly and 10 (90.90%) were distal growths. The P value was 0.03 which was significant (Table 8).
| Overall | Proximal | Distal | Chi-square | P-value | |
|---|---|---|---|---|---|
| Ulcerating | 21 | 2 | 19 | 6.67 | 0.03 |
| Polypoid | 33 | 12 | 21 | ||
| Superficial | 11 | 1 | 10 | ||
| Decreased distensibility of stomach | 18 | 2 | 16 | 1.18 | 0.27 |
| Abnormal pyloric function | 11 | 0 | 11 | 3.97 | 0.05 |
Table 8: Morphology of the tumor on upper GI scopy.
Distensibility of the stomach was decreased in 18 (27.69%) patients. Out of these 2 (11.11%) were in patients with proximal lesions, and 16 (88.88%) were in patients withdistal tumors. The difference was not statistically significant with a P value of 0.27. Abnormal pyloric function was found in 11 (16.92%) of the patients overall and all these patients had distal lesions. P value was significant at 0.05.
On histopathological examination the tumors were classified into well, moderately, and poorly differentiated adenocarcinoma. Out of the 17 (26.15%) well-differentiated tumors, 4 (23.52%) were located proximally and 13 (76.47%) located distally. Moderate differentiation was seen in 26 (40%) of the total lesions; of which 7 (26.92%) were proximal and 19 (73.07%) were distal. Poorly differentiated tumors were seen in 22 (33.84%) of the total; out of these 4 (18.18%) were proximal and 18 (81.81%) were distal tumors. The P value was not significant (Table 9).
| Differentiation | Overall | Proximal | Distal | Chi-square | P value |
|---|---|---|---|---|---|
| Well | 17 | 4 | 13 | 0.04 | 0.95 |
| Moderate | 26 | 7 | 19 | 0.36 | 0.55 |
| Poor | 22 | 4 | 18 | 0.13 | 0.72 |
Table 9: Histopathological examination.
Histopathology
The tumors were classified as either intestinal type or diffuse type according to Lauren classification. The intestinal type were 42 (64.61%) overall; of which 13 (30.95%) were proximal tumors and 29 (69.04%) were distal. 13 out of the 15 proximal lesions (86.66%), were of the intestinal type. The P value (0.04) was significant. The diffuse type was 23 (35.38%) overall; of which 2 (8.6%) were proximal tumors and 21 (91.30%) were distal. The P value (0.04) was significant (Table 10).
| Lauren classification | Overall | Proximal | Distal | Chi-square | P-value |
|---|---|---|---|---|---|
| Intestinal | 42 | 13 | 29 | 4.15 | 0.04 |
| Diffuse | 23 | 2 | 21 | 4.15 | 0.04 |
Table 10: Histopathology.
Staging
There was no significant difference between the proximal and distal gastric adenocarcinomas when all the stages were considered together, the chi-square being 3.99 and p value being 0.26. But when stage III and IV was considered alone, it was found that more number of proximal tumors had presented at a later stage than the distal tumors (Table 11).
| Overall | Proximal | Distal | |
|---|---|---|---|
| I A | 0 | 0 | 0 |
| I B | 6 | 0 | 6 |
| II | 13 | 3 | 10 |
| III | 17 | 1 | 16 |
| III B | 6 | 3 | 3 |
| IV | 23 | 8 | 15 |
Table 11: Staging.
Hepatic metastasis (H)
Hepatic deposits were found in 17 (26.15%) patients overall. Of these 7 (41.17%) were due to proximal tumors and 10 (58.82%)n were due to distal tumors. The P value was 0.04 which was significant (Table 12 and Figure 5).
| Overall | Proximal | Distal | Chi-square | P-value | |
|---|---|---|---|---|---|
| H | 17 | 7 | 10 | 4.25 | 0.04 |
Table 12: Hepatic metastasis (H).
Figure 5. CT image showing liver metastasis.
Peritoneal metastasis (P)
In our study, peritoneal metastases were found in 9 (13.84%) patients overall. Out of these 4 (44.4%) were due to proximal tumors, and 5 (55.55%) were due to distal tumors. The P value was 0.22, which was insignificant (Table 13).
| Overall | Proximal | Distal | Chi-square | P value | |
|---|---|---|---|---|---|
| P | 9 | 4 | 5 | 1.47 | 0.22 |
Table 13: Peritoneal metastasis (P).
Conclusion
In our study, there was no difference in the degree of differentiation between the proximal and the distal tumors. The proximal tumors were more often of the intestinal type when compared to the distal tumors, which was statistically significant. This was also in accordance with the findings in other studies. Proximal tumors tend to present at a higher stage than the distal tumors. But this was not statistically significant. Proximal tumors have hepatic metastasis at presentation more frequently than the distal tumors, which was a statistically significant finding in our study.
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgments
The encouragement and support from Bharath University, Chennai is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- American cancer society: cancer facts and figures 2005. Atlanta, American cancer society, 2005.
- Parikh AA, Mansfield P: gastric adenocarcinoma. In Cameron JL (ed): current surgical therapy, 8th ed. St. louis, mosby-year- book, 2004:95-100.
- Blot WJ, devesa SS, kneller RW, Fraumeni JF Jr: rising incidence adenocarcinoma of the esophagus and gastric cardia. JAMA 265:1287 -1289,1991.
- Meters WC, Damiano RJ Jr, Rotolo FS, Postlethwait RW: Adenocarcinoma of the stomach. Changing patterns over the last 4 decades. Ann surg 2051 - 8,1987.
- Salvon- Harman JC, Cady B, Nikulasson S, et al: shifting proportions of gastric adenocarcinomas. Arch surg 129:381-388, discussion 388-389,1994.
- Surveillance, epidemiology and end results program, 1975-2001. Do. Cca. P Sciences (ed): national cancer institute, 2004.
- Lauren P: the two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. An attempt at a histo-clinical classification. Actapathol microbial scand 64:31 -49.
- Kaneko E, nakamura T, Umeda N, et al: outcome of gastric carcinoma detected by gastric mass surgery in Japan. Gut 18:626-630,1977.
- Lowy AM, Mansfield PF, Leach SD, A jani J: Laparoscopic staging for gastric cancer surgery 119: 611-614, 1996.
- Burke EC, Karpeh MS Jr, conlon KC Brennan MF :Peritoneal lavage cytology in gastric cancer: an independent predictor outcome. Ann surgoncol 5:411 - 415,1998.
- Papachristou DN, Agnanti N D Agostino H, Fortner JG: Histologically positive esophageal margin in the surgical treatment of gastric cancer. Am j Surg 139: 711-713, 1980.
- Bozzetti J. Bonfanti G, Bufalino R, et al: Adequacy of margins of resection in gastrectomy for cancer. Ann surg 196: 685-690,1982.
- Siewert JR, Feith M. Stein HJ: Biologic and clinical variations of adenocarcinoma at the esophago-gastric junction: relevance of a topographic-anatomic subclassification. J SurgOncol 90:139 -146, discussion 146,2005.
- Harrison LE, Karpeh MS, Brennan MF: Total gastrectomy is not necessary for proximal gastric cancer. Surgery 123: 127 -130, 1998.
- Bozzetti F, Marabini E, Bonfanti G, et al: Subtotal versus total gastrectomy for gastric cancer: five-year survival rates in a multicenter randomized Italian trial. Italian gastrointestinal tumor study group. Ann surg 230: 170-178, 1999.
- Takekoshi T, Baba Y, Ota H, et al: Endoscopic resection of early gastric carcinoma: results of a retrospective analysis of 308 cases. Endoscopy 26:352-358, 1994.
- ParsonnetJ.Friedman GD. Vandersteen DP, et al: helicobacter pylori infection and the risk of gastric carcinoma. N Engl J Med 325:1127 -1131, 1991.
- Huntsman DG, Carneiro F. Lewis RF, et al: Early gastric cancer young, asymptomatic carriers of germ- line E-cadherin mutations. N Engl J Med 344:1904 - 1909,2001.
- Marrelli D. Pinto E, De Stefano A, et al: clinical utility of CEA, CA 19 -9,and CA 72-4 in the follow - up of patients with respectable gastric cancer. Am J Surg 181:16-19,2001.
- Willis S. Truongs, Guibnitz S, et al: Endoscopic ultrasonography in the preoperative staging of gastric cancer: accuracy and impact on surgical therapy. SurgEndosc 14:951-954, 2000.
- Kuntz C, Herfarth C: imaging diagnosis for staging of gastric cancer. Seminsurgoncol 17:96 -102,1999.
- Chen J. Cheong JH, Yun MJ, et al: improvement in preoperative staging of gastric adenocarcinoma with positron emission tomography. Cancer 103:2383 - 2390,2005.
- Burke EC, Karpeh MS, Conlon KC, Brennan MF: Laparoscopy in the management of gastric adenocarcinoma. Ann Surg 225:262 -267, 1997
- Romijn MG, van Overhagen H, SpillenaarBilgen EJ, et al: Laparoscopy and laparoscopic ultrasonography in staging of esophageal and cardiac carcinoma. Br J S urg 85: 1010-1012,1998.
- Hayashi H. Ochiai T. Suzuki T. et al: superiority of a new UICC TNM staging system for gastric carcinoma.Surgery 127:129 1352000.
- Lim L. Michael M. Mann GB. Leong T: Adjuvant therapy in gastric cancer. J ClinOncol 23:62200 -6232, 2005.
- Msika S, Benhamiche AM, Jouve JL, et al. Prognostic factors after curative resection for gastric cancer. A population-based study. Eur j cancer 36: 390 -396, 2000.
- Ekbom GA, Gleystecen JJ: Gastric malignancy: Resection for palliation. Surgery 88: 476 -481, 1980.
- Thompson AM, Rapson T, Gilben FJ, Park KG: Endoscopie palliative treatment for esophageal and gastric cancer. Techniques, complications, and survival in a population-based cohort of 948 patients. Surgendosc 18:1257-1262, 2004.
- P Piso et al. Proximal versus distal gastric carcinoma - What are the differences? Annals of Surgical oncology, 7 (7): 520 -525, 2000.
- JR Siewert MD, HJ Stein MD, and U Fink MD. Surgical resection for cancer of the cardia. Seminars in Surgical Oncology 1999, 17: 125 -131.
- JR Siewert, K Bottcher, HJ Stein, JD Roder, R Busch. Problem of proximal third gastric carcinoma. World J Surg. 19, 523-551, 1995.
- JR Siewert, K Bottcher, HJ Stein, JD Roder and the German Gastric Carcinoma Study Group. Relevant prognostic factors in gastric cancer. Annals of Surgery 228, n 44494611998
- TbSakaguchi et al. Characteristic and clinical outcome proximal-third gastric cancer. J Am Coll of Surg 1998; 187: 352-357.
- LE Harrison, MS Karpeh and MF Brennan. Proximal gastric cancers resected via a transabdominal-only approach. Annals of Surgery, 225, n°6, 678 -685, 1997.
Author Info
Ashwini Krishnamoorthy and K Kuberan*
Department of General Surgery, Sree Balaji Medical College & Hospital, Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Ashwini Krishnamoorthy, K Kuberan, Clinico Pathological Profiles of Patients with Proximal Versus Distal Gastric Adenocarcinoma, J Res Med Dent Sci, 2021, 9 (4):459-466.
Received: 22-Mar-2021 Accepted: 27-Apr-2021
