Research - (2020) Volume 8, Issue 3
Comparative Study Between Three Different Types of Denture Base Material for the Ability to form the Biofilm of Bacteria
Mohmmed R1, Karama T Al-Taee2* and Anas Hammad Abed3
*Correspondence: Karama T Al-Taee, Department of Basic sciences, College of Dentistry, Al-Anbar University, Iraq, Email:
Abstract
Aim: The aim of this study was to compare between three different types of denture base material heat cure acrylic resin (conventional acrylic), flexible acrylic and compact acrylic (acrylic block) according to the ability of their surfaces for bacterial adhesion and biofilm formation to know their susceptibility for contamination during dentures placement.
Materials and Methods: In our study, we were using two types of bacteria Pseudomonas aeruginosa and Staphylococcus epidermidis. That was previously tested for their ability to produce biofilm. To detect and measure the formation of biofilm, Quantitative biofilm assay and Scanning Electron Microscope were used.
Results: According to the results of (Quantitative) biofilm assay, the surfaces of the three dental materials showed significant differences in their ability to form biofilm. The results adhesion of P. aeruginosa and S. epidermidis on the three surfaces showed that, the conventional acrylic surface has the most ability to accumulate these bacteria and forming biofilm after contamination and incubation 24 hours, while the compact acrylic (block) showed less ability to forming biofilm, but the flexible acrylic was in between them.
Conclusion: Compact acrylic (acrylic block) in construction of dentures by the CAD-CAM technology has the advantage of decreasing the accumulation of bacteria (P. aeruginosa and S. epidermidis) on the surface of dentures in comparison with using the conventional acrylic and flexible acrylic by the conventional method (flasking procedure). In addition to that the P. aeruginosa was more prevalent than the S. epidermidis on the surfaces of the samples.
Keywords
Denture base material, Biofilm, Bacteria
Introduction
The introduction of the digital technology (CAD-CAM) in the construction of CDs will overcome the complications that are associated with the conventional method (flasking procedure), enhance their properties and offer better results [1]. This technology uses the acrylic block for construction of dentures (Figure1A). Flexible dentures are widely used today versus conventional rigid acrylic dentures because of resistance to fracture, aesthetic, excellent biocompatibility [2] and show little effects on the mucosa of dentures bearing area and little changes on the mucosa [3]. Biofilms are extremely significant to the infections of the human, and these communities that composed from aggregation of different microorganisms can promote on living or non-living surfaces [4] medical instruments made from Metallic and non-metallic, like catheters, implants, materials of the dentures are good environments for colonization of different kinds of microorganisms [5]. The rough surfaces enhance the accumulation of the biofilm than smooth surfaces [6]. The biofilms are composed of microorganisms that live in matrix produced by these microorganisms of hydrated extracellular polysaccharide [7]. The major dental diseases are dental caries and periodontal diseases may cause by biofilm [8] also systemic diseases, lung disease, diseases of cardiovascular system, diabetes mellitus that related to periodontal diseases [9]. The nature of the biofilm structure increases the microorganism resistance to the immune system defences of the host and antimicrobial factors [10].

Figure 1a. Compact acrylic (block).
One of the important virulence factors of Staphylococcus epidermidis and P. aeruginosa is the formation of biofilm. [11-13]. P. aeruginosa exhibited high ability to forming biofilm on denture base material [14]. The oral cavity may the source of the respiratory disease if P. aeruginosa present in saliva and sputum [9,13]. P. aeruginosa gramnegative bacilli well adapted to found in watery environments and produce a slimy alginate extracellular material that represent the texture of biofilm. S. epidermidis gram positive cocci colonizes the skin as microflora and forming biofilm by producing polysaccharide outside the cells [15]. The biofilm accumulates in greater amount on the surface of the denture because of the topography and the structure of these biomaterials ’ surfaces [16]. The using of disinfection material for cleaning the denture increases the roughness of the denture surface [17] this roughness leading to enhancement biofilm production [18]. New researches have demonstrated a link between systemic diseases and biofilm accumulation on the denture in older people [19]. SEM is a best tool for studying the biofilm details and the morphological microbial accumulation on the surface of the denture material. The SEM images showed that the biofilm is a bacterial cell enclosed by amorphous matrix that considered extracellular polymeric material [20]. The target of our study is that to compare between three different types of denture base material (heat cure acrylic resin, flexible acrylic and compact acrylic) for the ability of their surfaces to forming biofilm.
Materials and Methods
Preparation of the denture base material
Three different dentures base materials table 1 (heat cure acrylic resin or conventional acrylic, flexible acrylic resin and compact acrylic or acrylic block Figure 1A were used to prepare two types of samples, type A to SEM analysis (10 × 10 × 3 mm) and type B for spectrophotometer analysis (10 × 10 × 4 mm) Figure 1B.
| Denture base materials | Type | Brand name | Properties | Mode of using |
|---|---|---|---|---|
| Heat cure acrylic resin | PMMA | Veracril | Conventional Rigid acrylic | Pack- pressing polymerization |
| Flexible acrylic resin | Thermoplastic Nylon | IRIS | Flexible acrylic | Injection moulding polymerization |
| Compact acrylic resin (block) | PMMA made under high pressure and temperature | IRIS | Acrylic made under high pressure and temperature | Digital CAD-CAM technology (Milling) |
Table 1: Types of dentures base materials.

Figure 1b. Image of samples.
Isolation of bacteria
Two types of bacteria P. aeruginosa and Staph epidermidis were used in our study. P. aeruginosa was obtained from the water samples of dental chair unite in the clinics of dentistry college, the samples of water were collected and cultured on blood agar and MacConkey agar, the plates of media were incubated at 37ºC for 24 hrs. Staph epidermidis was obtained from cotton swaps of denture patient, cultured on blood agar and Manitole salt agar incubated in 37ºC for 24 hrs. According to the morphological characters and biochemical tests the bacteria were diagnosed [21]. The bacteria tested for producing biofilm, according to Stepanović, et al. [22].
Quantitative biofilm assay
For the purpose of examining the biofilm formation on the surfaces of the three different denture base materials (samples), the P. aeruginosa and Staph epidermidis were cultured on Brain Heart infusion broth and incubated at 37ºC for 24 hrs. BHI broth was prepared with 5% sucrose [23] and the samples were sterilized by using Ethylene oxide and then placed in tubes in triplicates and then contaminated with P. aeruginosa and Staph. epidermidis after diluting to 1:100 and then incubated in 37ºC for 24 hrs [14]. Uninoculated tubes containing sterile BHI broth supplemented with 0.5% sucrose with the samples considered to be the negative controls are used as blanks. The biofilm was visualized following staining with 0.5% crystal violet for 10 min [24]. The samples were stained with crystal violet were washed twice with PBS to discharge the stain and then dried. The dye of biofilms that covered the samples were resolubilized by 50 μl 95% ethanol for P. aeruginosa and 33% glacial acetic acid for Staph epidermidis [25]. Then the coloured solution transfer to wells of Microtiter plate to measure by spectrophotometer at 620 nm by a microplate reader [14,21]. After measuring OD, cut of value was calculated as below.
Odc (cut of value): Average OD of -ve control+(3 SD) of - ve control
The cut of value important in interpreting the results when used in this formula
OD isolates: Average OD of isolate – ODc If -the value obtained lack of biofilm production, while +ve value indicates biofilm production [26].
Biofilm formation for SEM examination
Brain Heart infusion broth was prepared and added 5% sucrose. (Ethylene oxide) was used to sterilize the samples of denture material. The samples were placed in triplicates tube, and contaminated with diluted 1:100 P. aeruginosa overnight BHI broth and then incubated in 37ºC for 24 hours and 4 days. The culture medium was changed to fresh BHI medium without P. aeruginosa. The medium was changed every 2 days. The control was BHI broth without bacteria to each sample [20]. The control group was composed of un inoculated sample of each denture base material in BHI broth. In order to observe the biofilm characteristics on the surface of samples, after incubation, each sample was gently removed and washed with 0.1 M PBS one time. Samples were fixed with 2.5% (glutaraldehyde) for 18hrs at laboratory temperature and then washed three times in 0.1 M phosphate buffer saline (pH 7.3) for 10 min. After fixation, all samples were dehydrated further in a graded ethanol series for 20 min [14]. The samples were dried and examined by SEM after 24 hours and after 4 days to describe the morphological characteristics of the surfaces after the biofilm formation. The examination was focused on surface morphology for biofilm only.
Results
Quantitative biofilm assay
The results of OD in Table 2 show differences in the ability of denture base materials for accumulated biofilm. The highest mean value within all the denture base materials was acquired with P. aeruginosa followed by S. epidermidis Figure 2.
| Material | Bacterial species | Test (average) OD | SD | Control | odc | Interpretation |
|---|---|---|---|---|---|---|
| F | S. epidermidis | 0.43 | 0.068 | 0.2 | 0.2238 | non supportive |
| P. aeruginosa | 0.7 | 0.027 | Supportive | |||
| C | S. epidermidis | 0.3 | 0.034 | 0.133 | 0.2165 | Supportive |
| P. aeruginosa | 0.61 | 0.1 | Supportive | |||
| H | S. epidermidis | 0.62 | 0.032 | 0.314 | 0.405 | Supportive |
| P. aeruginosa | 0.75 | 0.039 | Supportive |
Table 2: Types of samples, names of microorganisms and biofilm production, according to Quantitative biofilm assay. obsorbtion on 620 nm.OD Optical density, SD=standard deviation, odc=Cut of value, F=Flexible acrylic resin, C=Compact acrylic resin, H=Heat cure acrylic resin.
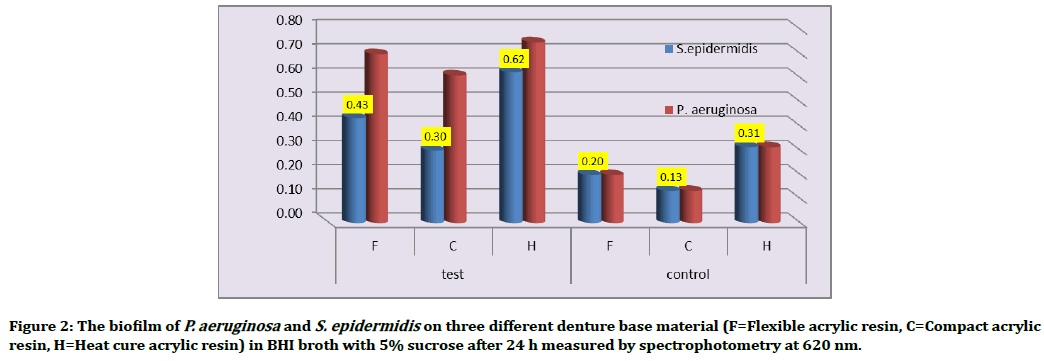
Figure 2. The biofilm of P. aeruginosa and S. epidermidis on three different denture base material (F=Flexible acrylic resin, C=Compact acrylic resin, H=Heat cure acrylic resin) in BHI broth with 5% sucrose after 24 h measured by spectrophotometry at 620 nm.
SEM analysis
In our study SEM images of dental base material surfaces showed different surface topography and Lesser smoothness (Figure 3).

Figure 3. SEM images of biofilm formations of P. aeruginosa on H1 and H2-Heat F1and F2-Flexible C1 and C2-Compect acrylic. H1, F1C1 after 24 hours. incubation in BHI broth H2, F2, C2 after 4 days incubation. different morphology shape for accumulation of P. aeruginosa on the three different surfaces of denture base materials.
Discussion
The current study showed that Heat cure resin shows a high ability to allow bacteria to form biofilm (0.747) for P. aeruginosa and (0.623) for S. epidermidis, while compact acrylic shows less ability to accumulate bacteria (0.609) for P. aeruginosa and (0.297) for S. epidermidis, according to our study results. The highest mean value within all the denture base materials was obtained with P. aeruginosa followed by S. epidermidis Figure 2. These results are consistent with. [14,15] In both bacteria adhesion the flexible was in between with other types of base denture material of accumulating bacteria on their surfaces A cut of value can be used to classify the surfaces in their ability to adhesion of bacteria and form the biofilm, which is divided into a strong, moderate and weak. by using the formula below:
(OD isolate: Average OD of isolate–ODc)
The production or non-production of biofilm can be calculated, if the value obtained from this formula is -ve this indicates the lack of biofilm production, while +ve value indicates biofilm production (Table 2). In our study all the surface of three base denture material enhances the formation of biofilm except the compact acrylic surface exhibit less ability to accumulate bacteria.
The reason for the formation of the biofilm is the possession of bacteria the ability to produce extracellular polysaccharide as a molecular glue to adhere the bacteria on the surfaces, [27] this ability, increasing when the roughness of the surface is high and this is available in the surfaces of denture material [18]. Many studies show the roughness of denture material is increased by using the disinfectant therefore biofilm form increased during the time [28]. Our study suggests using quantitative biofilm assays to determine the formation ability of biofilm, this result like result from [29]. The process of biofilm formation begins with the initial attachment of bacteria to a surface, which is affected by several factors, including the characteristics of the surface to which the bacteria adhere [21]. The roughness and the topographic surface determine the ability of bacteria to form the thickness of the biofilm [18].
Biofilm formation can also act as an important virulence factor in many persistent infections. Therefore, our results highlight the necessity of regular microbiological monitoring of dental surfaces and implementing strict hygiene practices and control measures in order to minimize cross-contamination risks [29].
Many early biofilm studies used scanning electron microscopy (SEM), which showed in detail the surface morphologies of microbial biofilms and their structure. Direct SEM observation clearly revealed that bacterial cells in a biofilm were extensively surrounded by fibrous or amorphous matrices, which represented the EPS [20]. In our study SEM images of dental base material surfaces showed different surface topography and Lesser smoothness, Therefore, P. aeruginosa accumulation of biofilm was appeared in different forms. In general, P. aeruginosa adherence was observed in cluster forms and whole attached cells were viewed in irregular morphology.
Scanning electron microscopy analysis revealed a different biofilm accumulation forms on flexible, heat cure and compact acrylic Figure 3. The thickness of biofilm was influenced in the period of incubation. After 4 days H2, F2 and C2, the biofilm accumulation more than 24 hours incubation H1, F1 and C1. Therefore, our study suggested that the increased of an incubation period increased the biofilm forming.
Conclusion
This study presents that the use of compact acrylic (acrylic block) in construction of dentures by the CADCAM technology has the advantage of reducing the aggregation of bacteria (P. aeruginosa and S. epidermidis) on the surface of dentures in comparing with using the conventional acrylic and flexible acrylic by the conventional method (flasking procedure). The P. aeruginosa was more able to adhesion than the S. epidermidis on the surfaces of the samples.
Conflict of Interest
There are no conflicts of interest.
References
- Maeda Y, Minoura M, Tsutsumi S, et al. A CAD/CAM system for the removable denture. Part I: Fabrication of complete dentures. Int J Prosthodont 1994;7:17–21.
- Shamnur SN, Jagadeesh KN, Kalavathi SD, et al. Flexible dentures: An alternate for rigid dentures. J Dent Sci Res 2010; 1:74-79.
- Mustafa GM, AlBaki MAA, Naji SA. Comparing the effects of denture base materials on hygiene of mucosal denture bearing area. Tikrit J Dent Sci 2014; 3:71-76..
- Peters BM, Jabra-Rizk MA, O'May GA, et al. Polymicrobial interactions: Impact on pathogenesis and human disease. Clin Microbiol Reviews 2012; 25:193–213.
- Beech IB, Sunner JA, Arciola CR, et al. Microbially-influenced corrosion: Damage to prostheses, delight for bacteria. Int J Artificial Organs 2006; 29:443-452.
- Busscher HJ, Rinastiti M, Siswomihardjo W, et al. Biofilm formation on dental restorative and implant materials. J Dent Res 2010; 89:657-665.
- Flemming HC, Wingender J. The biofilm matrix. Nature Reviews Microbiol 2010; 8:623.
- Seneviratne CJ, Zhang CF, Samaranayake LP. Dental plaque biofilm in oral health and disease. Chinese J Dent Res 2011; 14:87.
- Gurenlian JR. The role of dental plaque biofilm in oral health. Am Dent Hygienists Assoc 2007; 81:116-116.
- Domenech M, Ramos-Sevillano E, García E, et al. Biofilm formation avoids complement immunity and phagocytosis of Streptococcus pneumoniae . Infection Immunity 2013; 81:2606-2615.
- Arciola CR, Campoccia D, Montanaro L. Detection of biofilm-forming strains of Staphylococcus epidermidis and S. aureus. Expert Review Mol Diagnostics 2002; 2:478-484.
- Olivares E, Badel-Berchoux S, Provot C, et al. The BioFilm Ring Test: A rapid method for routine analysis of Pseudomonas aeruginosa biofilm formation kinetics. J Clin Microbiol 2016; 54:657-661.
- Caldas RR, Le Gall F, Revert K, et al. Pseudomonas aeruginosa and periodontal pathogens in the oral cavity and lungs of cystic fibrosis patients: A case-control study. J Clin Microbiol 2015; 53:1898-1907.
- Sahin C, Ergin A, Ayyildiz S, et al. Effect of biofilm formation, and biocorrosion on denture base fractures. J Adv Prosthodont 2013; 5:140-146.
- Kalishwaralal K, Barath Mani Kanth S, Pandian SRK, et al. Silver nanoparticles impede the biofilm formation by Pseudomonas aeruginosa and Staphylococcus epidermidis. Colloids Surfaces B: Biointerfaces 2010; 79: 340-344.
- Valentini F, Luz M S, Boscato N, et al. Biofilm formation on denture liners in a randomised controlled in situ trial. J Dent 2013; 41:420-427.
- Yuzugullu B, Acar O, Cetinsahin C, et al. Effect of different denture cleansers on surface roughness and microhardness of artificial denture teeth. J Adv Prosthodont 2016; 8:333-338.
- Gharechahi M, Moosavi H, Forghani M. Effect of surface roughness and materials composition on biofilm formation. J Biomater Nanobiotech 2012; 3:541-546.
- O'donnell LE, Smith K, Williams C, et al. Dentures are a reservoir for respiratory pathogens. J Prosthodont 2016; 25:99-104.
- Asahi Y, Miura J, Tsuda T, et al. Simple observation of Streptococcus mutans biofilm by scanning electron microscopy using ionic liquids. AMB Express 2015; 5:6.
- Winn WC. Koneman's color atlas and textbook of diagnostic microbiology. Lippincott williams and wilkins. 2006
- Stepanović S, Vuković D, Hola V, et al. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. J Pathol Microbiol Immunol 2007; 115:891-899.
- Souza J, Mota R, Sordi MB, et al. Biofilm formation on different materials used in oral rehabilitation. Brazilian Dent J 2016; 27:141-147.
- Wakimoto N, Nishi J, Sheikh J, et al. Quantitative biofilm assay using a microtiter plate to screen for enteroaggregative Escherichia coli. Am J Tropical Med Hygiene 2004; 71:687-690.
- O'Toole GA, Pratt LA, Watnick PI, et al. Genetic approaches to study of biofilms. Methods Enzymol 1999; 310:91-109.
- https://www.intechopen.com/books/antimicrobials-antibiotic-resistance-antibiofilm-strategies-and-activity-methods/the-methods-for-detection-of-biofilm-and-screening-antibiofilm-activity-of-agents
- Limoli DH, Jones CJ, Wozniak DJ. Bacterial extracellular polysaccharides in biofilm formation and function. Microbiol Spectrum 2015; 3:1-2.
- Odagiri K, Sawada T, Hori N, et al. Evaluation of denture base resin after disinfection method using reactive oxygen species (ROS). Dent Mat J 2012; 2011:155.
- Torlak E, Korkut E, Uncu AT, et al. Biofilm formation by Staphylococcus aureus isolates from a dental clinic in Konya, Turkey. J Infect Public Health 2017; 10:809-813.
Author Info
Mohmmed R1, Karama T Al-Taee2* and Anas Hammad Abed3
1Departement of Prosthetic, College of Dentistry, Al-Anbar University, Iraq2Department of Basic sciences, College of Dentistry, Al-Anbar University, Iraq
3Department of in Oral and Maxillofacial Surgery, Oral and Maxillofacial Surgery, College of Dentistry, Al-Anbar University, Iraq
Citation: Mohmmed R, Karama T Al-Taee, Anas Hammad Abed, Comparative Study Between Three Different Types of Denture Base Material for the Ability to form the Biofilm of Bacteria, J Res Med Dent Sci, 2020, 8(3): 73-78
Received: 17-Mar-2020 Accepted: 29-Apr-2020
