Research - (2021) Volume 9, Issue 5
Correlation of Urine Albumin Creatinine Ratio with Hematological Indices in Patients with Diabetic Nephropathy
Rajini Samuel T and AJ Manjula Devi*
*Correspondence: AJ Manjula Devi, Department of Biochemistry, Sree Balaji Medical College and Hospital Affiliated to Bharath Institute of Higher Education and Research, India, Email:
Abstract
To Determine The correlation between urine albumin creatinine ratio with haematological indices in diabetic nephropathy patients with diabetic controls and determine the correlation of urine albumin creatinine ratio in diabetic nephropathy patients with c1 diabetic controls. Further determine the correlation of haematological indices in diabetic nephropathy patients with diabetic controls. We conclude our study by saying that anemia is more common in diabetes and is a frequent complication of diabetic nephropathy. Sustained microalbuminuria is the earliest warming sign of asymptomatic diabetic nephropathy patients and 1s a potent risk factor for the development of progressive renal disease. Urine albumin creatinine ratio (ACR) is an accepted and useful screening test for microalbuminuria. RDW was found to be significantly associated with diabetic nephropathy patients and hence it is a useful renal biomarker for diabetic complications.
Keywords
Urea, Diabetes, Nephropathy, Microalbuminuria, Hypertension
Introduction
Diabetic nephropathy (nephropatia diabetica), also known as Kimmelstiel-Wilson syndrome, or nodular diabetic glomerulosclerosis and interpapillary glomerulonephritis, is a progressive kidney disease caused by angiopathy of capillaries in the kidney glomeruli. It is characterized by nephrotic syndrome and diffuse glomerulosclerosis [1-3]. It is due to long starling diabetes mellitus and is a prime indication for dialysis in many developed countries. It is classified as a microvascular complication of diabetes Diabetic nephropathy (DN) is the most common cause of nephrotic syndrome in adults and of end-stage renal disease in the US, accounting for up to 80% of cases of the latter. The prevalence of renal failure is probably about 40% among patients with type 1 diabetes mellitus [4].
The prevalence of renal failure among patients with type 2 diabetes mellitus is usually stated as 20 to 30%, but this figure is probably low. Renal failure usually takes 2: 10 yr after the onset of nephropathy to develop; however, because type 2 diabetes is often present for several years before being recognized, nephropathy often develops < 1O yr after diabetes is diagnosed. Diabetic nephropathy manifests as slowly progressive albuminuria with worsening hypertension and renal insufficiency. Diagnosis is based on history, physical examination, urinalysis, and urine albumin/creatinine ratio [5-7].
Anem1i.. is a common accompaniment to diabetes, particularly in those with albuminuria or reduced renal function? Diabetes is the single most common cause of endstage renal disease and therefore the most common cause of renal anemia. In addition, anemia may be more common 1n diabetes and develop earlier than m patients with renal impairment from other causes. Red blood cell distribution width (RDW) is a measure of variability in size of circulating erythrocytes obtained in standard automated complete blood counts. RDW may represent an important clinical marker for diabetes complications independent of traditional risk factors and disease duration [8,9].
The study was taken up in our population of middle aged and older individuals to investigate the correlation between urine albumin creatinine ratio with haematological indices like RBC, HB, MCV, MCH, MCHC and RDW m patients with diabetic nephropathy with diabetic patients as control [10,11].
Materials and Methods
The study was conducted m a sample of 49 diabetic nephropathy patients and 49 diabetic control patients attending diabetic and nephrology department for check-up at Sree Balaji Medical College and Hospitals, Chromepet, Chennai.
Study individuals were divided into group 1 and group 2. Group 1: Comprises of 49 known diabetic patients belonging to the age group between 40 and 80.
Group2: Comprises of 49 known diabetic nephropathy patients belonging to the age group between 40 and 80.
This study was conducted between May 2013 and May 2014. The research and ethics committee of the university approved the study protocol. All participants were provided with written informed consent before enrolment in the study. Demographic data, age, gender, height, weight, DM duration, general history and medications were recorded.
Blood pressure (systolic and diastolic) of the patient was measured in the sitting position after ten minutes of rest, and the average of two measurements (with a 5-minute interval) was used for analysis. Blood samples were collected for haematological indices and urine samples were collected for urine albumin creatinine ratio (ACR).
Inclusion criteria
Cases:
Diabetic nephropathy patients in the age group 40-80 years.
Control
Diabetic patients in the age group 40-80 years.
Exclusion criteria
Smokers.
Hypertension.
Alcoholics.
Chronic illness.
Sample collection
The study was explained to both cases and control and informed consent obtained from them before taking blood sample. The blood samples were collected by venepuncture under aseptic precautions.
Results
The study population comprised of a total of 98 individuals and of this 49 were known diabetic control patients (group 1) and 49 were known diabetic nephropathy patients (group 2). All the study individuals were in the age group between 40 and 80 · years. 49 diabetic controls (group 1) were age, sex and body mass index matched individuals.
All the biochemical study parameters were analysed using statistical product and service solutions (SPSS) 17 software. Statistical tests used were group statistics, independent samples test and Pearson' s correlation (Table 1). Both group 1 (control) and group 2 (cases) were compared with HB values on the bar diagram. Their significance and correlation is clearly visible in the graph (Figure 1). Both group 1 (control) and group 2 (cases) were compared with MCH values on the bar diagram. Their significance and correlation are clearly visible in the graph (Figure 2). Both group 1 (control) and group 2 (cases) were compared with RDW values on the bar diagram. Their significance and correlation are clearly visible in the graph (Figure 3).
| Group Statistics | |||||
|---|---|---|---|---|---|
| Group | N | Mean | Std. Deviation | Std. Error Mean | |
| Ratio mg of albumin / gram of creatinine | Control | 49 | 18.7416 | 6.95669 | 0.99381 |
| cases | 49 | 99.2276 | 72.06525 | 10.29504 | |
| RBC million /mm3 A,t | Control | 49 | 4.0516 | 0.67102 | 0.09586 |
| cases | 49 | 4.0069 | 0.69646 | 0.09949 | |
| HB g/dl | Control | 49 | 10.847 | 1.6989 | 0.2427 |
| cases | 49 | 9.035 | 1.2025 | 0.1718 | |
| PCV¾ | Control | 49 | 33.735 | 5.289 | 0.7556 |
| cases | 49 | 30.269 | 3.8854 | 0.5551 | |
| MCVfl | Control | 49 | 83.724 | 7.4925 | 1.0704 |
| cases | 49 | 76.655 | 9.2892 | 1.327 | |
| MCHpg | Control | 49 | 26.969 | 3.0303 | 0.4329 |
| cases | 49 | 23.037 | 3.8769 | 0.5538 | |
| MCHC¾ | Control | 49 | 32.159 | 1.1299 | 0.1614 |
| cases | 49 | 29.986 | 2.2789 | 0.3256 | |
| RDW-CV¾ | Control | 49 | 12.7288 | 1.17744 | 0.16821 |
| cases | 49 | 20.9918 | 2.5088 | 0.3584 | |
Table 1: T-Test.
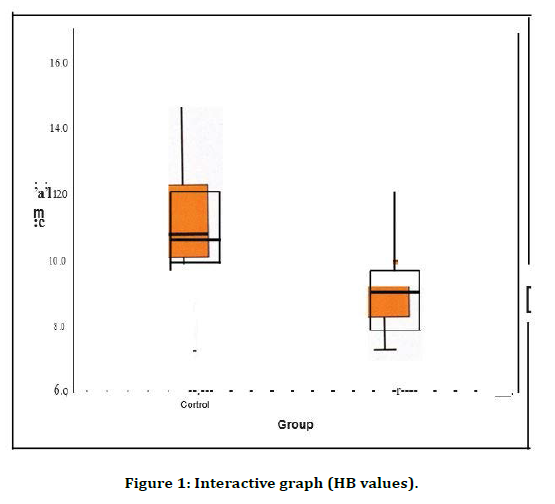
Figure 1. Interactive graph (HB values).
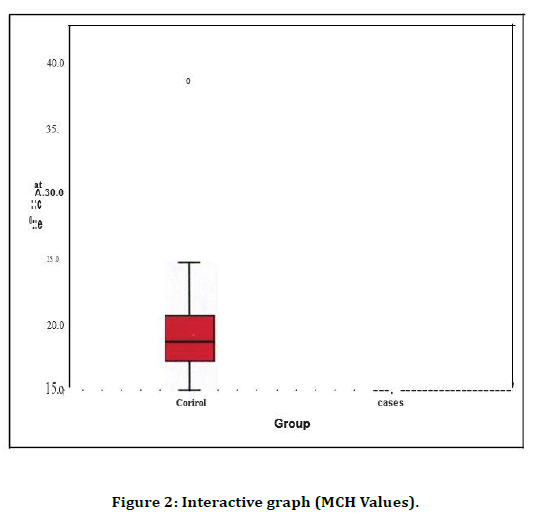
Figure 2. Interactive graph (MCH Values).
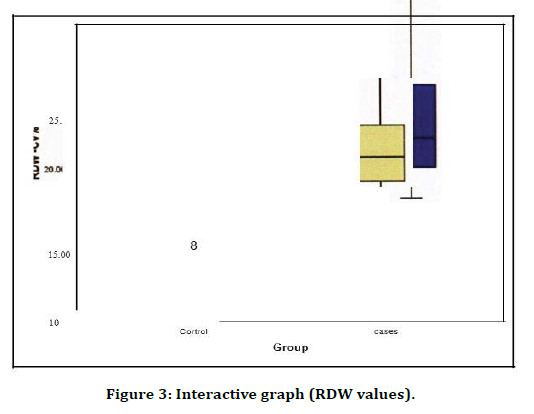
Figure 3. Interactive graph (RDW values).
The values are expressed in Mean ± S.D. The ratio of the mg of albumin per gram of creatinine among patients with diabetic nephropathy (99.2276 ± 72.06525) were significantly higher than controls(18.7416 ± 6.95669) The R-values among patients with diabetic nephropathy (4.0069 ± 0.69646) has no significance with controls( 4.0516 ± 0.67102) The HB values among patients with diabetic nephropathy (9.035 ± 1.2025) were significantly lower than controls(! 0.847 ± 1.6989)
The PCV values among patients with diabetic nephropathy (30.269 ± 3. 8854)) were significantly lower than controls (33.735 ± 5.2890) The pearsons correlation between RDW and RBC ( r=0.436 and p value =0.002) has significant positive correlation and between RDW and MCV ( r=-0.385 and p value =0 .006 , u nd between RDW and MCH ( r=-0.509) and p value- 0.000 ) and between RDW and MCHC ( r=- 0.549) and p value=0.000) were found to have significant negative correlation.
Discussion
DN begins as glomerular hyperfiltration (increased GFR). GFR normalizes with early renal injury and mild hypertension, which worsens over time. Microalbuminuria, urinary excretion of albumin in a range of 30 to 300 mg albumin/day, then occurs. Urinary albumin these consent ratios called microalbuminuria because detection of proteinuria by dipstick on routine urinalysis usually requires> 300 mg albumin/day. Microalbuminuria progresses to macroalbuminuria (proteinuria >300 mg/day at a variable course, usually over years. Nephrotic syndrome (proteinuria 3 g/day) precedes endstage renal disease, on average, by about 3 to 5 yr., but this timing is also highly variable [12-14].
In DN, the kidneys are usually of normal size or larger. Other risk factors include the following, duration and degree of hyperglycemia, hypertension, dyslipidemia, cigarette smoking, certain polymorphisms affecting the reninangiotensin- aldosterone axis, family history of diabetic nephropathy, genetic variables (decreased number of glomeruli) [15].
DN is asymptomatic m early stages. Sustained microalbuminuria is the earliest warning sign. Hypertension and some measure of dependent edema eventually develop in most untreated patients. In later stages, patients may develop syn oms and signs of uremia (eg, nausea, vomiting, anorexia) earlier (i.e., with higher GFR) than do patients without DN, possibly because the combination of end-organ damage due to diabetes (eg, neuropathy) and renal failure worsens symptoms. 1 3 23 28 Diagnosis is done by screening of all patients with diabetes with random urine albumin/creatinine ratio.20 48 In our study, the statistical results and analysis showed that the ratio of the mg of albumin per gram of creatinine among patients with diabetic nephropathy were significantly higher than diabetic control patients [16].
Diabetes is a Major health problem and for these patients, anemia is a common problem and can occur in up to 25% of people with the disease. This is because having diabetes for a long time can affect the kidneys and the nerves. These effects are known as diabetic nephropathy and diabetic neuropathy and may contribute to the development of anemia. Anemia is an unrecognized and irregularly treated condition among diabetics that can seriously affect their health and well -be in g. In our study the statistical results and analysis showed that the HB values and PCV values among patients with diabetic nephropathy were significantly lower than the diabetic control patients. Pearson’s correlation between the ratio of mg of albumin per gram of creatinine and HB and PCV in both cases (diabetic nephropathy patients) and controls (diabetic patients) were found insignificant [17,18].
When diabetes affects the kidneys (diabetic nephropathy), they may not be able to produce enough erythropoietin, a hormone that controls the production of red blood cells. Since the red blood cells carry oxygen, having fewer. d blood cells mean the body's organs may not get enough oxygen. This lack of oxygen produces the symptoms they may feel when suffering from anemia. Diabetic nephropathy may affect the way the body responds to anemia. Normally, when anemia develops, the body will try to correct it by increasing its production of erythropoietin in order to make more red blood cells. When diabetes affects the kidneys, the body may not be able to increase the production of erythropoietin in response to anemia. People with diabetes often have nutritional deficiencies which may or may not be directly caused by diabetes, but often can result in anemia [19].
The main causes of Anemia m Diabetic patients are Diabetic nephropathy which reduces production of erythropoietin, Diabetic neuropathy which affects the nervous system's anemia response, Nutritional deficiencies and some medications given for other conditions [20.21] related Red blood cell distribution width (RDW) is a measure of variability in size of circulating erythrocytes obtained in standard automated complete blood counts. RDW may represent an important clinical marker for diabetes complications independent of traditional risk factors and deceased [22].
RDW was shown to be significantly associated with diabetic nephropathy in a type2 diabetic population. In our study the statistical results and analysis showed that the RDW values among patients with diabetic nephropathy were significantly higher than controls. Pearson’s correlation between ratio of mg of albumin per gram of creatinine and RDW values were found significant positive.
Conclusion
Diabetes is the single most common cause of endstage renal disease and therefore the most common cause of renal anemia. In addition, anemia may be more common in diabetes and develop earlier than in patients with renal impairment from other causes. Anaemia 1s a frequent complication of diabetic nephropathy. It has only recently been recognized that in diabetic patient’s anaemia is seen not only in preterminal renal failure, but also frequently in patients with only minor derangement of renal function. Microalbuminuria provides early warning of renal damage. Maintaining good blood glucose control also appears to be effective in preventing the progression.
We conclude our study by saying that anemia is more common in diabetes and is a frequent complication of diabetic nephropathy. Sustained microalbuminuria is the earliest warning sign of asymptomatic diabetic nephropathy: patients and 1s a potent risk factor for the development of progressive renal disease. Urine albumin creatinine ratio (ACR) is an accepted and useful screening test for microalbuminuria. RDW was found to be significantly associated with diabetic nephropathy patients and hence it is a useful renal biomarker for diabetic complications.
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgments
The encouragement and support from Bharath University, Chennai, is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- Rho YH, Choi SJ, Lee YH, et al. The prevalence of metabolic syndrome in patients with gout: A multicenter study. J Korean Med Sci 2005; 20:1029-1033.
- Mellen PB, Bleyer AJ, Erling er TP, et al. Serum uric acid predicts incident hypertension in a biethnic cohort: The atherosclerosis risk in community’s study. Hypertension 2006; 48:1037-1042.
- Zhu Y, Pandya BJ, Choi HK. Comorbidities of gout and hyperuricemia in the US general population: NHANES 2007-2008. Am J Med 2012; 125:679-687.
- Kim TH, Lee SS, Yoo JH, et al. The relationship between the regional abdominal adipose tissue distribution and the serum uric acid levels in people with type 2 diabetes mellitus. Diabetol Metabolic Syndrome 2012; 4:1-7.
- Robert I. Disorders of purine and primidine metabolism. In: Horrison,s Principles of internal medicine. Eds, Kasper and Braunwald. 15th. Mc Grawhill, New York, 2001; 2268-2271.
- Nebulas I, Yuko I, Toda EI, et al. Association between serum uric acid, metabolic syndrome, and carotid atherosclerosis in Japanese individuals. Arterioscler Thromb Vase Biol, 2005; 25:1038-1044.
- Ramachandran A. Epidemiology of type II diabetes mellitus in India, Diabetes research centre & mv hospital for diabetes, Chennai. J Indian Med Assoc 2002; 100:425-427.
- Alberti KG, Zimmet P, Shaw J. IDF epidemiology task force consensus group. The metabolic syndrome new worldwide definition. Lancet 2005; 366:1059-1062.
- Chen L, Magliano DJ, Zimmet PZ. The worldwide epidemiology of type 2 diabetes mellitus: present and future perspectives. Nature Reviews Endocrinol 2011.
- https://www.niddk.nih.gov/
- https://diabetesatlas.org/en/
- Menke A, Casagrande S, Geiss L, et al. Prevalence of and trends in diabetes among adults in the United States, 1988-2012. JAMA 2015; 314:1021-1029.
- Barlow SE. Expert committee recommendations regarding the prevention, assessment, and treatment of childhood and adolescent overweight and obesity: Summary report. Paediatrics 2007; 120:S164-S192.
- Lang IA, Galloway TS, Scarlett A, et al. Association of urinary bisphenol A concentration with medical disorders and laboratory abnormalities 1n adults. JAMA 2008; 300:1303-1310.
- Rother KI. Diabetes treatment-bridging the divide. N Engl J Med 2007; 356:1499-1501.
- Fujioka K. Pathophysiology of type 2 diabetes and the role of incretin hormones and beta-cell dysfunction. JAAPA 2007; 3-8.
- Garcia-Roves PM. Mitochondrial pathophysiology and type 2 diabetes mellitus. Arch Physiol Biochem 2011; 17:177-187.
- Kim SY, Guevara JP, Kim KM, et al. Hyperuricemia and risk of stroke: A systematic review and meta-analysis. Arthritis Rheum 2009; 61:885-892.
- Nakagawa T, Tuttle KR, Short RA, et al. Hypothesis: Fructose-induced 9yperuricemia as a causal mechanism for the epidemic of the metabolic syndrome. Nat Clin Pract Nephrol 2005, I:80-86.
- Takahashi MM, de Oliveira EP, de Carvalho AL, et al. Metabolic syndrome, and dietary components are associated with coronary artery disease risk score in free-living adults: A cross-sectional study. Diabetol Metab Syndr 2011; 3:7.
- Roch-Ramel F, Guisan B. Renal transport of urate in humans. News Physiol Sci 1999; 14:80-84.
- Maxwell SR, Thomason H, Sandler D, et al. Antioxidant status in patients with uncomplicated insulin-dependent and non-insulin-dependent diabetes mellitus. Eur J Clin Invest 1997; 27:484-490.
Author Info
Rajini Samuel T and AJ Manjula Devi*
Department of Biochemistry, Sree Balaji Medical College and Hospital Affiliated to Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Rajini Samuel T, AJ Manjula Devi, Correlation of Urine Albumin Creatinine Ratio with Hematological Indices in Patients with Diabetic Nephropathy, J Res Med Dent Sci, 2021, 9 (5):258-263.
Received: 06-May-2021 Accepted: 24-May-2021
