Research - (2020) Volume 8, Issue 1
Effect of Dentine Bonding and Laser on Prevention of Teeth Discoloration Produced by Gray Mineral Trioxide Aggregate and Triple Antibiotic Paste: A Comparative In vitro Study
Farah Abdul Razzaq1* and Anas F Mahdee2
*Correspondence: Farah Abdul Razzaq, Department of Conservative Dentistry, University of Basrah, Iraq, Email:
Abstract
Aim: To evaluate the preventive efficacy of two dentine tubule occlusion methods (dentine bonding and laser) against discoloration caused by Gray Mineral Trioxide Aggregate (GMTA) and triple-antibiotic paste (TAP).
Materials and methods: Sixty extracted human maxillary first premolar were used in this study. Access cavities were prepared and endodontic instrumentation was performed for the buccal canals. The specimens then were randomly divided into two groups (n=30) as GMTA or TAP groups. Each of these were further divided into 3 subgroups according to surface treatment for the pulp chamber walls into: dentine bonding (Bon), Er,Cr:YSGG laser(Las), or left without treatment as control (Con). The buccal canal totally filled with GMTA or TAP. The color measurements were performed from a specific region on the buccal surface by using VITA Easyshade Advance at baseline, after 3 weeks, and 4 months incubation periods. The color lightness (L*) and the total color differences (ΔE*) during different stages of the study were calculated and analyzed.
Results: Although both bonding and laser treatment increase color lightness at the baseline measurements, this lightness decreased with time. At 3 weeks, the bonding subgroup of GMTA illustrated the highest measured color lightness in comparison to others. At the second incubation period (4 months), all subgroups showed decrease in color lightness with the highest detected values within TAP subgroups.
Conclusion: the use of dentine bonding and laser can prevent color changes produced by GMTA and TAP, however their efficacy appear to be time dependent.
Keywords
Triple-antibiotic paste, Gray MTA, Discoloration, Dentine tubule, Er, Cr:YSGG, Laser, Dentine bond
Introduction
Bacterial infection that resulting during hard tissue replacement with prosthesis is a big complication and inhibit tissue regeneration [1,2]. For the prevention of this infection a number of metals with antibacterial properties have been incorporated for the formula of replacement prosthesis such as the incorporation of zinc in calcium site or OH channel of hydroxyapatites to provide infection controlling in addition to the biocompatibility of the implant [3]. As well as Zn+2 and Co+2 chemical adsorption in to Sic added porcelain surface of the replacement prosthesis which lead to favorable antibacterial action against E. coli and S. aureus [4].
Revascularization is an endodontic procedure for pulp-dentine complex regeneration. This treatment is a substitute for the apexification of immature permanent dentition with necrotic pulp [5], to allow the continuance of physiological growth of the root [6]. One of the most essential phases of the revascularization is the disinfection of the canals [7]. Some research workers have advocated the usage of a TAP to kill the microbial flora present in the canals [8,9]. The existence of any antibiotic of tetracycline family, in TAP composition adversely affected tooth esthetic causing severe discoloration due to the it’s binding to ca+2 by chelation [10].
GMTA is a biocompatible repairing substance with positive sealing and biologic properties. It has been applied in surgical and non-surgical procedures including apical root-end fillings, repair of perforations, direct pulp capping procedures, apexification and revascularization of the pulp [11]. Several studies also reported discoloration after its usage in teeth within aesthetic zone [12,13]. The reason of discoloration created by GMTA related to its contents of metallic ions including Bi2O3 [14], Al2O3, MgO and Fe2O3 [15,16].
An effort of sealing of the dentinal tubules on the walls of the pulp chamber by the using dentine bonding had been reported as a viable approach to prevent discoloration [7]. In the last decade, dental laser such as Nd: YAG and Er.Cr:YSGG have become a popular choice for the management of DH. Various theories are asserted to describe the impact of laser irradiation on dentine, which including the sealing of dentinal tubule by fluid vaporization, melting and re-solidification [17,18]. There is only one report that recognized that dentine bonding, dentine desensitizing agent and laser can be used as preventive methods for tooth discoloration caused by TAP with short incubation time [19]. However, none of the previous research identifies the effect of these preventive procedures on different tooth discoloring materials and on extended period of incubation. Therefore, the aim of this study was to evaluate the preventive efficacy of two dentin tubule occlusion (dentine bonding and Er,Cr:YSGG laser methods against discoloration caused by GMTA and TAP on different incubation periods.
Materials and Methods
Sixty recently extracted human maxillary 1st premolar were used. All teeth were disinfected with Thymol 1g/100ml in D.W for 48 hrs. All teeth were examined and selected on the basis of the absence of any defect or abnormality. Each specimen was sectioned using a diamond disk under water cooling to obtain a standardized root length of 10 mm apical to CEJ. The access cavities were opened, and the canals were prepared by SX Protaper gold rotary file (Densply, Maillefer, Switzerland) using endomotor (D-smart I, china) before canal enlarging by peaso reamer #2 (China ) for final shaping. The canals were then irrigated using 3 ml of 5.25% NaOCl, 3 ml of 17% EDTA for 2 min, followed by 5ml of normal saline to simulate clinical irrigation before sealing the apical openings with GIC (Shanghai Rongxiang Dental Material Company, China). A standardized circular composite frame with a 7 mm diameter was created to 1 mm above CEJ on the buccal surface of the crown to standardize the measurement region.
Specimens were randomly allocated to the following three groups (n=20) bonding application (Bon), Er,Cr:YSGG laser application (Las), and no surface treatment (Con). Group Bon: The inner surfaces of pulp chamber were treated with Universal adhesive ( 3M, Germany) according to the following protocol a thin layer of bond was applied by micro brush then air dried for 5 sec then cured with LED (LINGCHEN, china) for 10 sec. This process was repeated for two times for each specimen in this group. Group Las: The inner surfaces of pulp chamber were treated with Er,Cr:YSGG laser (WaterLase, Biolase, USA ) by the setting (0.5 W/cm2 power, 20 Hz. frequency, 1% water and 1% air) using side firing tip (Water Lase, Biolase, USA ). This type of tip direct the beam laterally toward the pulp chamber walls at a standardized distance of 1mm by using an endodontic stopper, and for 2 min of emission. Group con: No surface treatment was applied to this group (control). After that the application of freshly mixture of either GMTA or TAP was placed immediately within the whole canal space.
The TAP prepared by mixing of (one scope which equal to 0.12 g of each of the following: tetracycline, metronidazole and ciprofloxacin) with D.W in (3:1) P/L ratio, and the mixture was immediately placed in the canals by a disposable syringe with intracanal application tip. The GMTA was freshly mixed according to the manufacturer instruction with D.W, and the wet sand -like mixture were placed in the root canal using MTA applicator (CERKAMED MEDICAL COMPANY, Poland) and adapted by using suitable endodontic plugger. After which the access cavity was sealed with temporary restoration (Cavimed, MODERN MEDICAL EQUIPMENT LLC.).
Color first measurement (baseline) by using VITA Easy Shade Advance spectrophotometer (Vident, Germany). This measurement was repeated for each sample after (3 weaksk) and (4 months) incubation period, Since the revisualization procedure require bacterial free environment, at the period of tissue regeneration, this need to placement of the disinfecting material for several weeks [20], and (4 months) incubation period to investigate more discoloration effect of the used materials by increased period of incubation. After each measurement samples were incubated humidifier at 37°C.
The spectrophotometer recorded the measurements using the CIE Lab system (Commission International de L’Eclairage, Vienna, Austria) recording 3 color parameters (L*, a* and b*). ΔE* was also calculated which represented the distance difference between the first and second colors measurements. Therefore, ΔE1 and ΔE2 were calculated for each group according to the following formula [21].
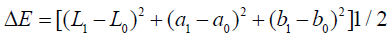
The recorded data were statistically analyzed by parametric statistical tests. ANOVA for repetitive measures was used to compare statistical among baseline, 3 weeks and 4 months records for lightness (L*) at confident level (95%) and p≤0.05. If this test showed statistical significance, further and bonferroni corrections comparisons between groups were performed using Bonferroni test. While, paired t-test were used to statistical comparisons between ΔE1 and ΔE2 for the respective measurements. ANOVA and Bonferroni test were also used to compare between Con, Bon, and Las subgroups at different time interval measurements of color lightness (L0=Baseline, L1=3 weeks, and L2=4 months), while unpaired t-test was used to compare between the respective ΔE* values. For statistical comparing between TAP and GMTA groups, the unpaired t-test was used. All computational work was performed using SPSS statistical software (SPSS v20.0; SPSS Inc.).
Results
The lightness measurements (L*) for different periods of the study are illustrated in Figure 1. All groups show decrease in color lightness with time. The highest change in color lightness is clearly detected within the second stage measurements (L2) for both GMTA and TAP. For GMTA groups, bonding treatment decrease the effect of GMTA on color lightness. This is clearly apparent in the first stage measurements (L1), which shows the highest lightness value (88.6 ± 2.6) and become less after 4 months (L2=84.9 ± 2.7). While, laser treatment shows little effect on maintaining the color lightness for GMTA groups at the different stages of the study. On the other hand, both bonding and laser treatment appear to have undetectable effect on color lightness within TAP groups in comparison to their control group measurements (Figure 1).
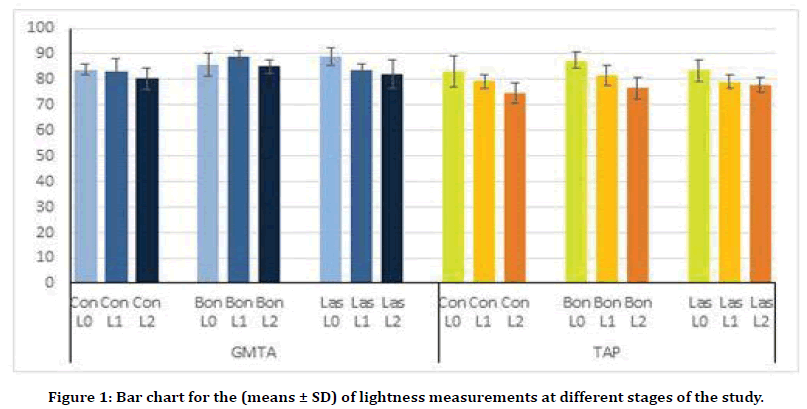
Figure 1. Bar chart for the (means ± SD) of lightness measurements at different stages of the study.
The total color change (ΔE*) are represented in Figure 2. In GMTA groups, the highest value for ΔE1 is for bonding group followed by laser and control groups (10.3 ± 3.01, 8.5 ± 3.4 and 7.02 ± 2.9 respectively). While the ΔE2 shows the highest value in the laser group (12.9 ± 5.6) in comparison to bond (8.5 ± 2.05) and control (6.7 ± 3.9) groups. However, total color changes in the TAP groups show increase in ΔE* with time i.e. higher ΔE2 values in comparison to ΔE1. The highest ΔE2 value in TAP was recorded in the bond group (12.9 ± 3.2) (Figure 2).
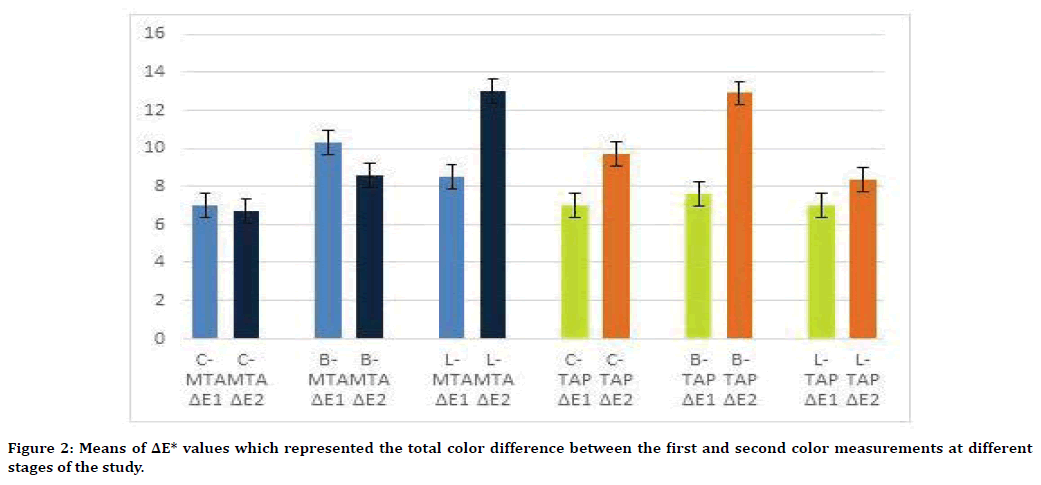
Figure 2. Means of ΔE* values which represented the total color difference between the first and second color measurements at different stages of the study.
The statistical comparisons L0, L1 and L2 measurements within the same group are shown in (Table 1). ANOVA for repetitive measured test was employed and showed statistical significant difference (p<0.05) between L0, L1, and L2 of all subgroups for both GMTA and TAP. The following Bonferroni shows statistical significant difference (p<0.05) between laser GMTA subgroups only with no statistical difference between control and bond subgroups. On the other hand, TAP subgroups showed statistical significant difference between bond subgroups (p<0.001), and between L0 and L1 for control and laser subgroups (p<0.05).
| Groups | ANOVA p-value | Stages | Bonferroni test p-value | |
|---|---|---|---|---|
| Stages | ||||
| L1 | L2 | |||
| Con-GMTA | 0.012 | L0 | 1.000 (NS) | 0.130 (NS) |
| L1 | 0.017 (S) | |||
| Bon-GMTA | 0.013 | L0 | 0.382 (NS) | 0.733 (NS) |
| L1 | 0.029 (S) | |||
| Las-GMTA | 0.005 | L0 | 0.003 (S) | 0.005 (S) |
| L1 | 0.478 (NS) | |||
| Con-TAP | 0.001 | L0 | 0.194 (NS) | 0.002 (S) |
| L1 | 0.005 (S) | |||
| Bon-TAP | 0 | L0 | 0 (S) | 0 (S) |
| L1 | 0.041 (NS) | |||
| Las-TAP | 0.037 | L0 | 0.077 (NS) | 0.024 (S) |
| L1 | 0.597 (S) | |||
There is statistical significant when p ≤ 0.05
Table 1: ANOVA for repetitive measures and Bonferroni test to compare between (L0, L1 and L2) within the same group for both GMTA and TAP.
Another analysis was made between the ΔE* values within the same subgroup (ΔE1 and ΔE2) by using paired t-test (Table 2). This test shows statistical significant difference (p<0.05) between GMTA Laser subgroup with no statistical significant difference (p>0.05) between GMTA control and bond subgroups. For TAP subgroups, there is statistical significant difference (p ≤ 0.05) between ΔE1 and ΔE2 of bond subgroups and no statistical significant difference (p>0.05) for Laser subgroup.
| Groups | Subgroups of GMTA | p-value |
|---|---|---|
| GMTA | Las-ΔE1 Vs. Las- ΔE2 | 0.013(S) |
| TAP | Con-ΔE1 Vs. Con-Δ E2 | 0.057(S) |
| Bon-ΔE1 Vs. Bon-Δ E2 | 0.006(S) |
There is statistical significant when p ≤ 0.05
Table 2: Paired T-test comparison between ( ΔE1 and ΔE2) for all subgroups of GMTA and TAP (the non-significant p values are not displayed).
For comparing between control, bond and Laser measurements for lightness (L) and ΔE values within the same group of either GMTA of TAP both ANOVA and Bonferroni tests (Table 3) were used. ANOVA test shows there is statistical significant difference only for the L0, L1 and ΔE2 comparisons of GMTA. Further statistical analysis by Bonferroni test shows statistical significant difference at L0 measurements between control and laser (p=0.01) and at L1 measurements between control and bond subgroups for GMTA (p=.006). Other color lightness measurement including all TAP subgroups show no statistical significant difference (p>0.05).
| Stages | ANOVA p -value | Subgroups | Bonferroni test | |
|---|---|---|---|---|
| Subgroups | ||||
| Bon | Las | |||
| GMTA-L0 | 0.012 | Con | 0.529 (NS) | 0.010(S) |
| Bon | 0.228(NS) | |||
| GMTA-L1 | 0.003 | Con | 0.006(S) | 1.000(NS) |
| Bon | 0.011(S) | |||
| GMTA- ΔE2 | 0.007 | Con | 0.579(NS) | 0.006(S) |
| Bon | 0.061(NS) | |||
There is statistical significant when p ≤ 0.05
Table 3: ANOVA and Bonferroni test to compare between (Con, Bon and Las) of both GMTA and TAP (the non-significant p values are not displayed).
Another statistical comparisons were made to detect the differences between GMTA and TAP groups by using unpaired t-test for color lightness (L*) and total color change (ΔE*) values (Table 4). For lightness values, the baseline measurements (L0) show no statistical significant difference between GMTA and TAP for control and bonding subgroups, while the laser shows statistical significant difference (p=0.005). Larger statistically significant difference between GMTA and TAP are clearly detected in the L1 and L2 measurements with highest significant differences (p<0.001) between bond subgroups. For total color change comparisons (ΔE*) the only detected statistical significant difference was between the bonding subgroups (p=0.03).
| Stages | Subgroups | P-value |
|---|---|---|
| L0 | Las-GMTA Vs. Las-TAP | 0.005 |
| L1 | Con-GMTA Vs. Con-TAP | 0.031 |
| Bon-GMTA Vs. Bon-TAP | 0 | |
| Las-GMTA Vs. Las-TAP | 0.001 | |
| L2 | Con-GMTA Vs. Con-TAP | 0.006 |
| Bon-GMTA Vs. Bon-TAP | 0 | |
| Las-GMTA Vs. Las-TAP | 0.039 | |
| ΔE1 | Bon-GMTA Vs. Bon-TAP | 0.038 |
| ΔE2 | Bon-GMTA Vs. Bon-TAP | 0.002 |
| Las-GMTA Vs. Las-TAP | 0.047 |
There is statistical significant when p ≤ 0.05
Table 4: Unpaired T-test to compare the color lightness values of GMTA Vs. TAP at different stages of study (the non-significant p values are not displayed).
Discussion
The results of this study confirmed those of preceding studies displaying that TAP and GMTA stained the teeth that underwent managements by them [10,14]. If crown discoloration occurs, treatment by intercoronal bleaching can be performed [22]. Although internal bleaching is a reliable method, it can sometimes lead to undesirable consequences such as root resorption. Severe discoloration requires additional bleaching, thus increasing the cost, time, and complications [23].
The modified procedure presented in this study is designed to avoid unwanted crown discoloration. This method is a noninvasive, easy, basic and cost-effective. Our study showed that dentin tubule occlusion methods had a preventive effect against the discoloration caused by TAP and GMTA. All dentin tubule occlusion methods are intended to mechanically seal the dentin tubules [24], thus preventing tetracycline and GMTA from entering the tubules. In addition, none of the previous studies used different discoloring materials with different prevention technique and on an extended period of time. Therefore, this study was conducted to evaluate the effect of Dentin bonding and Er:Cr:YSSG laser (Waterlase) as preventive treatment for discoloration caused by GMTA and TAP. In some studies, all procedures were performed from the apical aspect to avoid disruption of the intact crown and to prevent coronal microleakage [25,26], which did not imitate the clinical procedure. In the present study, all procedures were approached from the coronal aspect to simulate the clinical condition.
Color measurement was performed using VITA Easyshade Advance, which measures according to the CIELab system. This system enables the determination of color according to the internationally adopted ISO standards. The values of L* (lightness) were detected which indicates the darkening and lightening patterns were the ones of the biggest concern [27]. According to the condition of this study, and in comparison to the control groups, the bond and laser samples of GMTA showed higher lightness values during the baseline measurements. This was clearly illustrated within laser subgroup which reflects the whiting action of laser that may occur immediately after use. This could be due to the water vaporization action of laser that may lead to this opaque appearance [28]. After the first incubation period the bond subgroup of GMTA gave the highest lightness value among all tested groups. This can be explained that the application of 2 layers of dentin bonding was not only occluding the dentinal tubules and preventing the discoloration effect of GMTA, but it also may cause some sort of tooth whiting effect. The explanation for this is still unknown, but it could be due to the effect of these two layers of bonding material on the light reflection and refraction within the measured specimens, which may give this lighter pattern. This agreed with Akbari, et al. [16] who concluded that application of two layers of dentine bonding before using gray and white MTA may prevent tooth discoloration. Khim, et al [29] also recommended the application of dentin bonding as an additional step before endodontic obturation to seal the dentinal tubules and prevent sealer interactions which may reduce tooth discoloration. Whilst, coronal discoloration caused by GMTA and TAP progressed in a timedependent manner consistent with the previous research [30,31]. The reason for this might be due to the inherent composition of GMTA and TAP, which undergo certain chemical changes leading to discoloration. As the time progresses, further chemical reactions and seepages of discoloration products which may lead to more tooth discoloration. This agreed with the findings of Khim, et al. [29] study who showed that dentine bonding could not prevent coronal discoloration completely, but it could effectively reduce the risk of coronal discoloration caused by root canal sealer remnants. Other studies evaluated the efficacy of dentin bonding in preventing coronal discoloration caused by triple antibiotic paste [32]. On the other hand, the laser subgroups discoloration could be related to the alteration of the optical properties for the lased surface. This possibly due to the laser thermal effect that can induce alterations in the physical and chemical composition [33]. The present study resulted in darker laser specimens when comparing with bond. This may relate to the micro morphological changes reported in the lased dentine surface [34,35]. This possibly produced rough and pitted surface of the lased dentin which may increase in the amount of the GMTA and TAP ruminants in contact with the dentin surface.
The present results also found that the prevention methods had less effect on TAP groups in comparison to GMTA. This could be due to the difference in the composition between the two materials. TAP composed of tetracycline which is able to chelate with the dentin surface calcium [36]. This may produce darker and much difficult tooth discoloration products than the main discoloring component of GMTA (Bi2O3). Also the difference in the mixing consistency and wettability of the fresh mixture between GMTA and TAP could be another cause. Since GMTA workable consistency was a form of wet sand like material, while TAP fresh mixture is creamier and paste like consistency. This may affect the adaptation and manipulation between the two materials and this may affect their discoloration actions.
ΔE* between the initial and final color measures demonstrates the degree of total color change represented by a numerical value. This means that each parameter presents within ΔE* equation (L*, a* and b*) is basically part of the color change that totally measured in one number. Therefore, ΔE* could not exactly demonstrate the direction of the color axis on the CIE Lab space [37]. According to the results of this study, the statistical comparisons between ΔE* recorded significant p values only within TAP groups which showed higher changes in all color parameters including L*. While the smaller color differences which recorded statistical significance within L* values in the GMTA groups were not detected by ΔE. Additionally, most of the previous studies detecting color changes for external discoloring materials were using the ΔE values in their statistical comparisons [38,39]. While, other studies that detected color changes caused by internal discoloring materials such as pulp treatment materials were much focused on the L* values in addition to ΔE values [40]. This possibly due to that the external discoloring material could cause faster and severer changes in the CIE Lab parameters detected by the spectrophotometer. On the other hand, the internal discoloring materials could take longer time to be detected externally by the same device. Therefore, and according to the limitations of this study, the L* values rather than ΔE has been determined within this study to detect the amount of sample discoloration. In a clinical situation, the effect of the degenerative products from blood cannot be easily excluded from the internal discoloring materials. Moreover, the use of TAP is normally accompanied with GMTA as part revascularization procedure. These can be considered as limitations for the present study, which only used totally blood free samples with only one pulp treatment material.
Conclusion
Within the limits of this study, it can be concluded that dentin tubule occlusion methods use within this study have preventive effect against discoloration caused by TAP and GMTA. However this prevention is limited by the type of the discoloring material and the period of incubation. Further studies are required to identify tooth discoloration within clinical situations using several preventive measures.
References
- Roy M, Fielding GA, Beyenal H, et al. Mechanical, in vitro antimicrobial, and biological properties of plasma-sprayed silver-doped hydroxyapatite coating. ACS Appl Mater Interfaces 2012; 4:1341-1349.
- Darwiche H, Barsoum WK, Klika A, et al. Retrospective analysis of infection rate after early reoperation in total hip arthroplasty. Clin Orthop Related Res 2010; 468:2392-2396.
- Bhattacharjee A, Gupta A, Verma M, et al. Site-specific antibacterial efficacy and cyto/hemo-compatibility of zinc substituted hydroxyapatite. Ceramics Int 2019; 45:12225-12233.
- Bhattacharjee A, Gupta A, Prem Anand M, et al. Adsorption effect of Zn+2 and Co+2 on the antibacterial properties of SiC‐porcelain ceramics. Int J Appl Ceramic Tech 2020; 17:327-332.
- Petrino JA, Boda KK, Shambarger S, et al. Challenges in regenerative endodontics: A case series. J Endod 2010; 36:536-541.
- Iwaya SI, Ikawa M, Kubota M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent Traumatol 2001; 17:185-187.
- Reynolds K, Johnson J, Cohenca N. Pulp revascularization of necrotic bilateral bicuspids using a modified novel technique to eliminate potential coronal discolouration: A case report. Int Endod J 2009; 42:84-92.
- Tawfik H, Abu‐Seida A, Hashem A, et al. Regenerative potential following revascularization of immature permanent teeth with necrotic pulps. Int Endod J 2013; 46:910-922.
- Bezgin T, Yılmaz A, Celik B, et al. Concentrated platelet‐rich plasma used in root canal revascularization: 2 case reports. Int Endod J 2014; 47:41-49.
- Kim JH, Kim Y, Shin SJ, et al. Tooth discoloration of immature permanent incisor associated with triple antibiotic therapy: A case report. J Endod 2010; 36:1086-1091.
- Parirokh M, Torabinejad M. Mineral trioxide aggregate: A comprehensive literature review—part III: Clinical applications, drawbacks, and mechanism of action. J Endod 2010; 36:400-413.
- Karabucak B, Li D, Lim J, et al. Vital pulp therapy with mineral trioxide aggregate. Dent Traumatol 2005; 21:240-243.
- Bortoluzzi EA, Araújo GS, Tanomaru JMG, et al. Marginal gingiva discoloration by gray MTA: A case report. J Endod 2007; 33:325-327.
- Parirokh M, Torabinejad M. Mineral trioxide aggregate: A comprehensive literature review—part I: Chemical, physical, and antibacterial properties. J Endod 2010; 36:16-27.
- Ioannidis K, Mistakidis I, Beltes P, et al. Spectrophotometric analysis of crown discoloration induced by MTA- and ZnOE-based sealers. J Appl Oral Sci 2013; 21:138-144.
- Akbari M, Rouhani A, Samiee S, et al. Effect of dentin bonding agent on the prevention of tooth discoloration produced by mineral trioxide aggregate. Int J Dent 2012; 2012:1-3.
- Kimura Y, Wilder-Smith P, Yonaga K, et al. Treatment of dentine hypersensitivity by lasers: A review. J Clin Periodontol 2000; 27:715-721.
- Markowitz K, Pashley DH. Discovering new treatments for sensitive teeth: The long path from biology to therapy. J Oral Rehab 2008; 35:300-315.
- Küçükekenci F, Küçükekenci A, Cakici F. Evaluation of the preventive efficacy of three dentin tubule occlusion methods against discoloration caused by triple-antibiotic paste. Odontology 2018; 107:186-189.
- Diogenes A, Henry MA, Teixeira FB, et al. An update on clinical regenerative endodontics. Endodontic Topics 2013; 28:2-23.
- Yuan JC, Brewer JD, Monaco EA, et al. Defining a natural tooth color space based on a 3-dimensional shade system. J Prost Dent 2007; 98:110-119.
- Yasa B, Arslan H, Akcay M, et al. Comparison of bleaching efficacy of two bleaching agents on teeth discoloured by different antibiotic combinations used in revascularization. Clin Oral Invest 2015; 19:1437-1442.
- Ari H, Üngör M. In vitro comparison of different types of sodium perborate used for intracoronal bleaching of discoloured teeth. Int Endod J 2002; 35:433-436.
- Orchardson R, Gillam DG. Managing dentin hypersensitivity. J Am Dent Assoc 2006; 137:990-998.
- Yasa B, Arslan H, Akcay M, et al. Comparison of bleaching efficacy of two bleaching agents on teeth discoloured by different antibiotic combinations used in revascularization. Clin Oral Investig 2015; 19:1437-1442.
- Akcay M, Arslan H, Yasa B, et al. Spectrophotometric analysis of crown discoloration induced by various antibiotic pastes used in revascularization. J Endod 2014; 40:845-848.
- Kirchhoff A, Raldi D, Salles A, et al. Tooth discolouration and internal bleaching after the use of triple antibiotic paste. Int Endod J 2015; 48:1181-1187.
- Brodbelt R, O'brien W, Fan P, et al. Translucency of human dental enamel. J Dent Res 1981; 60:1749-1753.
- Khim TP, Sanggar V, Shan TW, et al. Prevention of coronal discoloration induced by root canal sealer remnants using dentin bonding agent: An in vitro study. J Conservative Dent 2018; 21:562-568.
- Parsons JR, Walton RE, Ricks-Williamson L. In vitro longitudinal assessment of coronal discoloration from endodontic sealers. J Endod 2001; 27:699-702.
- Ioannidis K, Mistakidis I, Beltes P, et al. Spectrophotometric analysis of crown discoloration induced by MTA-and ZnOE-based sealers. J Appl Oral Sci 2013; 21:138-144.
- Shokouhinejad N, Khoshkhounejad M, Alikhasi M, et al. Prevention of coronal discoloration induced by regenerative endodontic treatment in an ex vivo model. Clin Oral Investig 2018; 22:1725-1731.
- Bachmann L, Diebolder R, Hibst R, et al. Changes in chemical composition and collagen structure of dentine tissue after erbium laser irradiation. Spectrochimica Acta Part A: Molecular Biomol Spectroscopy 2005; 61:2634-2639.
- Chou JC, Chen CC, Ding SJ. Effect of Er, Cr: YSGG laser parameters on shear bond strength and microstructure of dentine. Photomed Laser Surg 2009; 27:481-486.
- Ishizaki NT, Matsumoto K, Kimura Y, et al. Thermographical and morphological studies of Er, Cr: YSGG laser irradiation on root canal walls. Photomed Laser Therapy 2004; 22:291-297.
- Kim JH, Kim Y, Shin SJ, et al. Tooth discoloration of immature permanent incisor associated with triple antibiotic therapy: A case report. J Endod 2010; 36:1086-1091.
- https://sensing.konicaminolta.us/blog/identifying-color-differences-using-l-a-b-or-l-c-h-coordinates/
- Abdulla HA, Majeed MA. Assessment of bioactive resin-modified glass ionomer restorative as a new CAD/CAM Material Part II: Fracture strength study. J Res Med Dent Sci 2019; 7:74-79.
- Obaid AA, AlMawash A, Alyabis N, et al. An in vitro evaluation of the effect of polishing on the stainability of different CAD/CAM ceramic materials. Saudi Dent J 2019; 31:1-7.
- Kirchhoff AL, Raldi DP, Salles AC, et al. Tooth discolouration and internal bleaching after the use of triple antibiotic paste. Int Endod J 2015; 48:1181-1187.
Author Info
Farah Abdul Razzaq1* and Anas F Mahdee2
1Department of Conservative Dentistry, University of Basrah, Iraq2Department of Restorative and Aesthetic Dentistry, University of Baghdad, Iraq
Citation: Farah Abdul Razzaq, Anas F Mahdee, Effect of Dentine Bonding and Laser on Prevention of Teeth Discoloration Produced by Gray Mineral Trioxide Aggregate and Triple Antibiotic Paste (A Comparative In vitro Study), J Res Med Dent Sci, 2020, 8(1):187-194.
Received: 27-Jan-2020 Accepted: 11-Feb-2020
