Research - (2019) Volume 7, Issue 3
Effect of Hydroxyapatite Coating on Plasma Nitrided Surface of Commercially Pure Titanium
Sabreen W Ibrahim1*, Nagham B Kamil1, Husam M Khalaf1 and Tebarak AA Al-Slmani2
*Correspondence: Sabreen W Ibrahim, College of Dentistry, Al-Mustansiriya University, Iraq, Email:
Abstract
Introduction: Dental implants were widely used being considered as successful treatment option for replacing missing teeth. Different methods were used to alter surface morphology and topography to accelerate healing and improve oseointegration. The aim of this study was to assess corrosion resistance and in vitro bioactivity in simulated body fluid (SBF) of plasma nitrided surfaces with and without hydroxyapatite coating.
Material and Methods: Commercial pure titanium grade II surfaces were treated using glow plasma nitride apparatus for 10 h, then coated with hydroxyapatite by dip coating, The electrochemical corrosion test was performed once the samples were inserted in Hank’s solution, the polarization potential and corrosion rate were measured. Treated and coated samples immersed in SBF for 7 days to evaluate the in vitro bioactivity. Phase analysis by X-ray diffraction (XRD) and scanning electron microscope (SEM) have been carried out on the surfaces before and after immersion in SBF.
Results: Coating with hydroxyapatite on pre-nitrided titanium surfaces increases the bioactivity of titanium and increases the corrosion resistance compared to nitrided and untreated surfaces.
Conclusion: Coating on pre-nitrided surfaces can be considered an effective way to enhance the formation of bone-like material on titanium surfaces and increase the corrosion resistance of titanium.
Keywords
Titanium, Plasma nitride, Hydroxyapatite, Dip coating, Bioactivity, Corrosion
Introduction
Now-a-days dental implants are considered the most successful treatment option for all concerned patients. Prosthetic rehabilitation of patients with lost teeth to restore functioning and esthetics was the main cause of using implants [1].
Being rather biocompatible and bioinert, titanium, in comparison with other implant materials in use, has adequate yield strength, hardness, little foreign body reaction, and direct connection with bone [2].
In spite of the acceptable success rates of using dental implants, poor bone quality and metabolic disease may result in failure of dental implants. Hence, nowadays researchers are attracted towards surface modification of biomaterial in order to improve the bone-implant contact to enhance immediate or early loading of final prosthesis after placement, and decrease or eliminate the waiting period, which is usually uncomfortable for the patients [3].
After a healing period, an implant is considered osseointegrated, when there is no movement between the implant and bone under normal conditions of loading as a result of direct bone formation on an implant surface without deposition of fibrous encapsulation, which is poorly vascularized collagenous capsule [4].
Titanium has superior characteristics compared to other implant materials, but the osteoconductivity of titanium is less than ceramics, so coating titanium with ceramics is a way to overcome this drawback [5].
Modification of the surface properties of titanium by plasma nitride is considered a simple, dry technique, which is echo-friendly with low cost and does not affect the intrinsic properties of the material except its surface (increasing both of the surface roughness and wettability) [6].
Hydroxyapatite coating can accelerate bone regeneration on implant surface. Because hydroxyapatite is a main inorganic component of bone, it can activate the osteoblasts and provide free calcium ions during bone formation [7].
Dip coating is a technique utilized to precipitate ceramic coatings on the implant surface, which can deposit thin layers of coating (less than 10 μm thickness). This method improves the biological activity of the titanium, thence accelerate the osseointegration [8,9].
Simulated Body Fluid (SBF) test is an efficient, suitable, quick and economical test for evaluating the bioactivity of biomaterial. This test was introduced by Kokubo et al. [10]. This procedure involves formation of bone-like apatite layer on the surface of implant which is considered as a positive biological response, the SBF solution represent the inorganic part of human blood plasma [10].
The primary aim of this study was to evaluate the in vitro bioactivity in simulated body fluid (SBF) and corrosion resistance of plasma nitrided surfaces with and without hydroxyapatite coating.
Materials and Methods
Sample preparation
30 commercially pure titanium grade II disks (29 mm diameter and 2 mm thickness were used as substrates, and immersed in ultrasonic path filled with ethanol for 15 min after mirror polishing, to remove contamination and debris, then placed for 10 min in distilled water bath and left to dry. These disks were divided into 3 groups; 10 disks were left untreated (control group), 10 disks were treated by plasma nitride, and 10 disks were treated with plasma nitride and coated with HA.
Nitriding process
The D.C. power supply in a locally manufactured (Iraq) plasma nitriding system was connected to the cathode once the experimental substrates were placed on the center of cathode. For glow-discharge plasma nitriding, the following parameters were used (nitrogen gas, pressure of 2 × 10-1 mbar, feed voltage of 650 V, current of 0.02 mA, and 10 hours was the selected time for nitriding). Before the beginning of plasma nitriding process, the plasma chamber was evacuated to vacuum pressure of (5 × 10-4 mbar) to confirm the fully removal of the gas-like hydrocarbons, then all the experimental substrates were cleansed by argon (Ar) plasma sputtering for 15 min. The nitriding process started when the nitriding gas (N2) was allowed to enter into the evacuated chamber, the process of flow rate continues until the pressure stabilizes at (2 × 10-1 mbar), and the cathode voltage and discharging current keep on increasing up to plasma formation. At the end of this process, the samples were left to cool in the same gas (nitrogen), where cooling the samples in the same environment prevents oxidation of the nitrided substrates.
Coating solution preparation
8 nitrided disks were coated with HA by dip coating. Preparation of HA solution (HA Particles size 98.5%, <40 μm, sky spring nanomaterial, USA) was performed by dissolving 0.01 gm of P2O5 in 50 ml ethanol with continuous stirring on hot stirrer (Daihan Lab Tech/ Model: LSM-1003, Korea), for half an hour, then 7 gm of HA added to solution with continuous stirring on hot stirrer to obtain homogenous solution, after that the samples were sintered in tube furnace (Carbolite type MTF 12/38 A. Bamford England. Serial No.3/88/432, Maximum temperature 1200°C) at 400°C under inert gas (argon).
Tests performed
XRD analysis
Phase analysis was used for all samples (control, nitrided and HA coating on pre-nitrided surface before and after immersion in SBF) by Shimadzu Lab XRD-6000 Powder X-ray diffractometer (Japan), utilizing Cu Kα radiation. The 2Ө angles swept from 30°-80° at one degree increment. The peak indexing was analyzed based on the JCPDS (joint committee on powder diffraction standards).
Structural surface characterization
To study the surface morphological and topographical characteristics of experimental specimen, samples were imaged using scanning electron microscope (SEM) (AA 300 Angstrom Advanced Inc, USA). Energy dispersive Xray spectroscopy (EDX) analysis used to describe the atomic components of the experimental samples.
Bioactivity test in simulated body fluid (SBF)
Titanium disks are immersed in SBF fluid at (37°C, pH 7.4, 1 L) for 7days the prepared SBF followed the work of Kokubo et al. with ion concentration is nearly similar to that of body fluid, Hank's solution chemical composition include NaCl (8.00 g/L), KCl (0.40 g/L), CaCl2 (0.14 g/L), NaHCO3 (0.35 g/L), MgCl2.6H2O (0.60 g/L), MgSO4.7H2O (0.06 g/L), Na2HPO4 (0.06 g/L), KH2PO4 (0.06 g/L), Glucose.2H2O (1.0 g/L) [10].
Electrochemical corrosion test
The assessment of the corrosion behavior in relation to current and potential voltage was carried by anodic polarization plot. Better corrosion resistance is indicated by higher polarization voltage and lower current density. Corrosion rate unit, which is used in this study, is millimeter per year (mmpy).
Statistical analysis
Data of the experiment were statistically analyzed using SPSS program (version-23). Testing equality of means value by analysis of variance (one-way ANOVA) and LSD were used to compare the significance difference between means in this study, with statistical significance level was set at 0.05.
Results
XRD analysis
XRD patterns of untreated, plasma nitride cp Ti and HA coating on plasma nitrided cp Ti were fitted to those data reported for the hexagonal αTi (JCPDS-ICDD file #5-682), cubic TiN (JCPDS-ICDD file #38-1420), Ti2N (JCPDS-ICDD file #23-1455), Ti3N2-x (JCPDS-ICDD file #40-0958), Ti4N3-x (JCPDS-ICDD file #39-1015), (JCPDS-ICDD file #09-0169 for Ca3(PO4)2) and (JCPDS-ICDD file #9-432 for HA). The 2Ө angles were swept from 30°-80° in one degree increment.
The XRD analysis of the plasma-treated groups (Figure 1a) detects the presence of TiN phases on Ti surfaces which confirms nitrogen ion implantation in the titanium surfaces as compared with untreated titanium (Figure 1b), while Figure 1c shows HA peaks with Ti peaks.
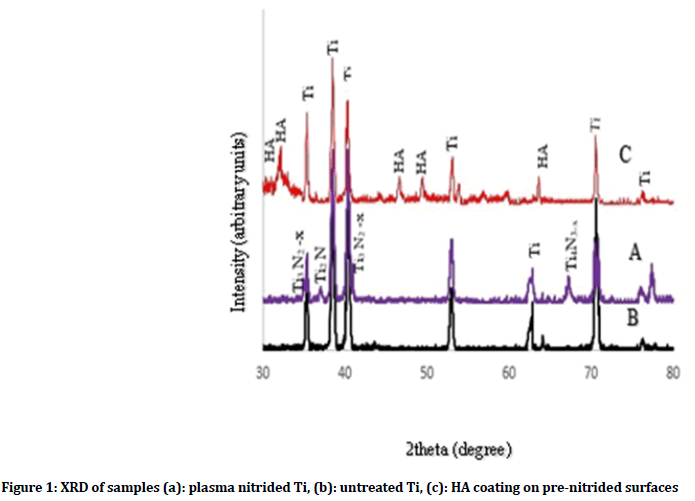
Figure 1: XRD of samples (a): plasma nitrided Ti, (b): untreated Ti, (c): HA coating on pre-nitrided surfaces
The presence of Ti peaks in the XRD pattern after coating process could be a result of the penetration of X-rays beyond the coated layer. Figures 2a and 3a show formation of HA layer after immersion in SBF fluid for 7 days as compared with untreated and plasma nitrided Ti as shown in (Figures 2a and 3a). On the other hand, Figure 4 shows XRD of HA coated samples on prenitrided surfaces after immersion in SBF for 7 days, which indicates low crystallinity of HA peaks similar to that of bone apatite as compared with XRD of HA coated samples on pre-nitrided surfaces before immersion in SBF, which indicates higher crystallinity and larger particles (Figure 4).
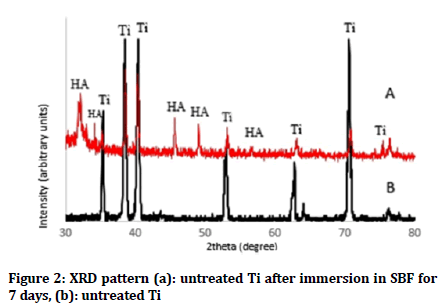
Figure 2: XRD pattern (a): untreated Ti after immersion in SBF for 7 days, (b): untreated Ti
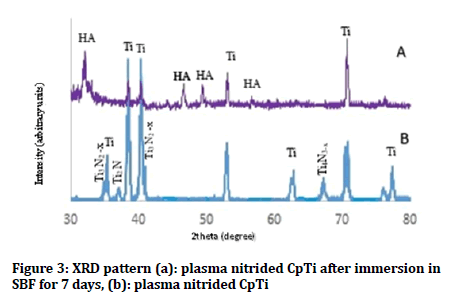
Figure 3: XRD pattern (a): plasma nitrided CpTi after immersion in SBF for 7 days, (b): plasma nitrided CpTi
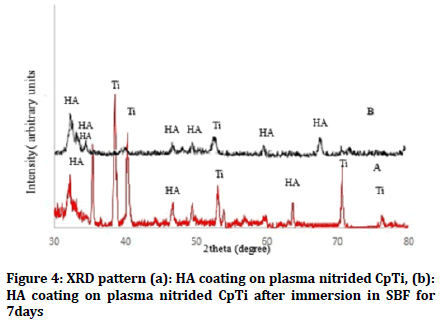
Figure 4: XRD pattern (a): HA coating on plasma nitrided CpTi, (b): HA coating on plasma nitrided CpTi after immersion in SBF for 7days
Scanning electron microscope (SEM)/ Energy dispersive X-ray spectroscopy (EDX)
Scanning electron microscope was executed to investigate the surface morphology of the samples. Plasma nitriding surface treatment results in homogenous microstructure of titanium nitride layer with distinct irregularities (Figure 5a) compared to polished surface, which shows polishing marks (Figure 5b). However, the coated samples show even distribution of HA layer over nitrided surface with thin cracks which are observed on the surface of the coated samples. Cracks are usually formed with sol gel coating because of the uneven shrinkage of the gel film during drying and heating treatment (Figure 5c).

Figure 5: SEM of (a): plasma nitrided Ti, (b): untreated Ti, (c): HA coating on plasma nitrided Ti
After soaking in SBF, SEM of untreated surfaces shows formation of discrete small apatite crystals (Figure 6a). Nitrided samples after immersion in SBF present agglomeration of apatite particles mostly covering the surface (Figure 6b). Coated samples on pre-nitrided surface are completely covered by regular and dense apatite crystals (Figure 6c).

Figure 6: SEM of samples after immersion in SBF for 7days (a): untreated Ti, (b): plasma nitrided Ti, (c): HA coating on plasma nitrided Ti
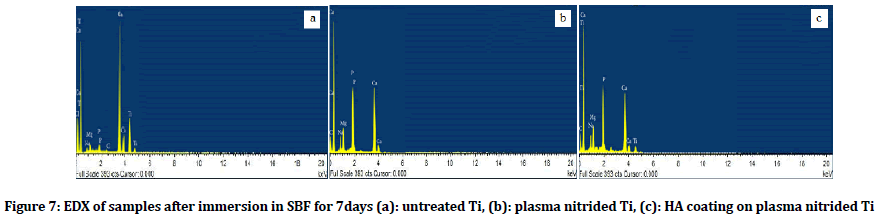
The total amount of Ca and P crystals and other elements were measured by EDX, the Ca/P ratio of titanium surface, plasma nitrided surfaces and HA coating on pretreated surfaces after immersion in SBF were 0.82, 1.59 and 2.15 respectively) as shown in (Figure 7).
Figure 7: EDX of samples after immersion in SBF for 7days (a): untreated Ti, (b): plasma nitrided Ti, (c): HA coating on plasma nitrided Ti
Electrochemical corrosion test
Potentiodynamic polarization curves: Figure 8 illustrates the anodic polarization plots for untreated, plasma nitrided CpTi and HA coating on pre-nitrided surfaces. The measured polarization voltage is found to be (-0.312 V, -0.38 V and -0.51 V) for HA coating on pre-nitrided, plasma nitrided and untreated surfaces respectively. While the current density for HA coating on pre-nitrided, plasma nitrided and untreated surfaces are (3.8 × 10-8 A/cm2, 7.5 × 10-8 A/cm2 and 1.4 × 10-7 A/cm2 respectively). The higher polarization and lower current density of HA coating on pre-nitrided surfaces clearly reveal the enhancement of the corrosion resistance of Cp Ti with coating over nitriding surfaces in comparison to nitriding of Cp Ti.
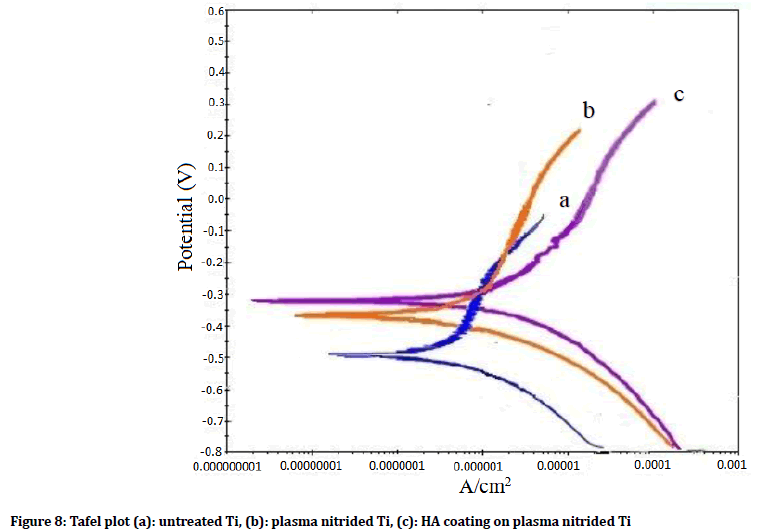
Figure 8: Tafel plot (a): untreated Ti, (b): plasma nitrided Ti, (c): HA coating on plasma nitrided Ti
Corrosion rate
Descriptive statistics of the corrosion rate of untreated, plasma nitrided surfaces and HA coating on plasma nitrided surfaces of CpTi are shown in Table 1. Untreated group shows the highest mean (1.34 × 10-2), while HA coated on pre-nitrided Ti shows the lowest mean in (0.36 × 10-2). The equality of means is tested by ANOVA test as shown in Table 2. This table presents highly significant differences in corrosion rate mean between 3 groups. The least significant difference (LSD) test used to differentiate between groups as shown in Table 3, manifests a highly significant difference between nitrided and HA coated on pre-nitrided surfaces when compared with untreated group, and significant difference of nitrided group when compared with HA coating on nitrided surface.
| Groups | No. | Mean × 10-2 | SD × 10-2 |
|---|---|---|---|
| Untreated | 6 | 1.24 | ± 0.23 |
| Nitrided | 6 | 0.72 | ± 0.20 |
| HA coating on nitrided surface | 6 | 0.45 | ± 0.24 |
Table 1: Summary statistics (Means and standard deviation) of corrosion rate for all experimental groups (mmpy)
| Groups | Sum of squares | df | Mean squares | F-test | Sig. |
|---|---|---|---|---|---|
| Between groups | 1.62 | 2 | 0.811 | 15.34 | 0 |
| Within groups | 0.79 | 15 | 0.053 | - | |
| Total | 2.414 | 17 | - | ||
Table 2: Equality of means of corrosion rate by ANOVA for all three groups
| Groups | Mean difference | Sig. | |
|---|---|---|---|
| Untreated (control) | Nitrided surfaces | 0.44 | 0.004 |
| HA coated on nitrided surface | 0.72 | 0 | |
| Nitrided surfaces | HA coated on nitrided surface | 0.27 | 0.05 |
The mean difference is significant at the 0.05 level.
The mean difference is highly significant at the 0.01 level.
Table 3: Multiple comparison (LSD) among all groups
Discussion
Titanium and its alloys are bio inert materials, which result in poor osseointegration, incapable to support bone bonding in long-term implants, and can produce host inflammatory response at the site of implantation by releasing titanium particles in the surrounding tissue. In order to solve these problems and stimulate bone formation for proper osseointegration, titanium implant surfaces are modified or coated with specifically engineered bioactive materials, and various elements may be implanted [11].
Plasma nitride was applied to different alloys such as, Ti6Al4V and CrCoMo, which resulted in enhancement of wear and friction resistance and improvement of surface hardness of Ti6Al4V alloy [12].
Kokubo et al. stated that the bioactivity of titanium surfaces is not high enough because of the slow bone formation on titanium surface and delayed osseointegration of titanium implant to bone that may take several months. Therefore numbers of studies were conducted to improve the interaction of titanium with biological environment and minimize the time required for proper osseointegration through surface modification of titanium [13].
Plasma nitride is widely utilized to modify surface topography, and increases the surface roughness and wettability [14].
Surface treatment of Cp Ti via plasma nitride may decrease the non-functional time during healing period after implantation, the patient ’ s discomfort, and the failure rates [15].
According to results of EDX, the ratio of Ca/P of the untreated titanium surface was (0.82) which is less than Ca/P ratio of hydroxyapatite (1.67), while Ca/P ratio of plasma nitrided titanium after soaking in SBF was (1.59), which indicates formation of bone-like crystals, and the Ca/P ratio of HA coating on pre-nitrided surfaces was (2.15), this refers to precipitation of calcium rich amorphous calcium phosphate [16].
The nitride layer that is formed on implant surface resulted in increased roughness and other topographical features which could be very important for bone regeneration, adhesion, scattering and cellular proliferation accelerated on the rough surface. Rough surfaces would favor the proliferation of osteoblasts, while smooth surfaces would favor the proliferation of fibroblasts [17].
Surface treatment of titanium implant by plasma nitride resulted in formation of nitride layer that improves the deposition of calcium phosphate phase, which is considered as a very effective way to enhance the precipitation of bone-like apatite material on the implant surface this result agrees with Cordioli et al. [18].
Oxynitride phases present on the surface of plasma nitrided titanium allows negatively charged surface centers to act as nucleating sites for the adsorption of Ca2+ ions from the body fluid, then incorporation of negatively charged phosphate groups with formation of calcium phosphate [19].
Strnad et al. [20] stated that the osteoconductive properties of HA resulted in formation of calcium phosphate layer over HA coated samples.
The SBF is similar to blood plasma in that it is highly saturated with calcium and phosphorous which are required to apatite deposition and they have similar ion composition and concentrations in a stable phase, therefore, Hank’s solution as an SBF is a simple test that can be used to understand the initial phase of bone-like mineral formation on titanium surface [21].
Corrosion resistance of plasma nitrided alloy, are higher than those of the non-treated one. The nitride layer provides a more protective, and resistant layer that reduces the ion release rate and alloy toxicity, which agree with Gordin et al. [22].
The samples coated by HA on plasma nitrided surfaces has an increased bioactivity as indicated by the increase in Ca/P ratio, compared to the samples treated with plasma nitrogen ion implantation only. This clearly indicates that surface roughness, which increases surface area along with surface chemistry and similarity of the chemical composition of HA with bone, may have an excellent ability to induce bioactivity of titanium, where this result agrees with Thirugnanam et al. [23].
Conclusion
HA layer is coated on plasma nitrided CpTi by dip coating. According to the in vitro SBF test, HA coating on plasma nitrided titanium accelerate formation of calcium phosphate on their surfaces compared with nitrided and untreated surfaces. The formation of Ti2N peaks as shown in XRD spectra indicating diffusion of nitrogen ions into the titanium surface. The corrosion resistance increases on HA coating on plasma nitrided titanium when compared to the nitrided and untreated surface
Conflict Of Interest
The authors declare that there is no conflict of interest regarding the publication of this manuscript.
References
- Stanford CM, Schneider GB. Functional behaviour of bone around dental implants. Gerodontology 2004; 21:71-7.
- Brunette DM, Tengvall P, Textor M, et al. Titanium in medicine: Material science, surface science, engineering, biological responses and medical applications. SSBM 2012.
- Lee SW, Hahn BD, Kang TY, et al. Hydroxyapatite and collagen combination-coated dental implants display better bone formation in the peri-implant area than the same combination plus bone morphogenetic protein-2-coated implants, hydroxyapatite only coated implants, and uncoated implants. J Oral Maxillofac Surg 2014; 72:53-60.
- Mavrogenis AF, Dimitriou R, Parvizi J, et al. Biology of implant osseointegration. J Musculoskelet Neuronal Interact 2009; 9:61-71.
- Heimann RB. Materials science of crystalline bioceramics: A review of basic properties and applications. CMU J 2002; 1:23-46.
- Ou KL, Shih YH, Huang CF, et al. Preparation of bioactive amorphous-like titanium oxide layer on titanium by plasma oxidation treatment. Appl Surf Sci 2008; 255:2046-51.
- Morra M, Cassinelli C, Cascardo G, et al. Multifunctional implant surfaces: Surface characterization and bone response to acid‐etched Ti implants surface‐modified by fibrillar collagen I. J Biomed Mater Res A 2010; 94:271-9.
- Anil S, Anand PS, Alghamdi H, et al. Dental implant surface enhancement and osseointegration. In Implant dentistry-A rapidly evolving practice. IntechOpen 2011.
- Šimůnek A, Strnad J, Štěpánek A. Bioactive titanium implants for shorter healing period. Clin Oral Implants Res 2002; 13.
- Kokubo T, Takadama H. How useful is SBF in predicting in vivo bone bioactivity?. Biomaterials 2006; 27:2907-15.
- Nautiyal VP, Mittal A, Agarwal A, et al. Tissue response to titanium implant using scanning electron microscope. Natl J Maxillofac Surg 2013; 4:7.
- Ueda M, Wei R, Reuther H. Plasma-based ion implantation treatments under industrially relevant conditions. In solid state phenomena. Trans Tech Publications 2005; 107:31-6.
- Kokubo T, Kim HM, Kawashita M. Novel bioactive materials with different mechanical properties. Biomaterials 2003; 24:2161-75.
- Sykaras N, Iacopino AM, Marker VA, et al. Implant materials, designs, and surface topographies: Their effect on osseointegration. A literature review. Int J Oral Maxillofac Implants 2000; 15.
- Sá JC, de Brito RA, Moura CE, et al. Influence of argon-ion bombardment of titanium surfaces on the cell behavior. Surf Coat Technol 2009; 203:1765-70.
- Abdelrahim RA, Badr NA, Baroudi K. Effect of anodization and alkali-heat treatment on the bioactivity of titanium implant material (an in vitro study). J Int Soc Prev Community Dent 2016; 6:189.
- Khang D, Lu J, Yao C, et al. The role of nanometer and sub-micron surface features on vascular and bone cell adhesion on titanium. Biomaterials 2008; 29:970-83.
- Cordioli G, Majzoub Z, Piattelli A, et al. Removal torque and histomorphometric investigation of 4 different titanium surfaces: an experimental study in the rabbit tibia. Int J Oral Maxillofac Implants 2000; 15.
- Piscanec S, Ciacchi LC, Vesselli E, et al. Bioactivity of TiN-coated titanium implants. Acta Materialia 2004; 52:1237-45.
- Strnad Z, Strnad J, Povysil C, et al. Effect of plasma-sprayed hydroxyapatite coating on the osteoconductivity of commercially pure titanium implants. Int J Oral Maxillofac Implants 2000; 15:483-90.
- Weizbauer A, Modrejewski C, Behrens S, et al. Comparative in vitro study and biomechanical testing of two different magnesium alloys. J Biomater Appl 2014; 28:1264-73.
- Gordin DM, Busardo D, Cimpean A, et al. Design of a nitrogen-implanted titanium-based superelastic alloy with optimized properties for biomedical applications. Mater Sci Eng C 2013; 33:4173-82.
- Thirugnanam A, Kumar TS, Chakkingal U. Bioactivity enhancement of commercial pure titanium by chemical treatments. Trends Biomat Artif Organs 2009; 32:76-85.
Author Info
Sabreen W Ibrahim1*, Nagham B Kamil1, Husam M Khalaf1 and Tebarak AA Al-Slmani2
1College of Dentistry, Al-Mustansiriya University, Baghdad, Iraq2College of Education, Al-Iraqia University, Baghdad, Iraq
Citation: Sabreen W Ibrahim, Nagham B Kamil, Husam M Khalaf, Tebarak AA Al-Slmani, Effect of Hydroxyapatite Coating on Plasma Nitrided Surface of Commercially Pure Titanium, J Res Med Dent Sci, 2019, 7(3): 86-92.
Received: 15-Apr-2019 Accepted: 10-May-2019