Research - (2020) Volume 8, Issue 1
Effect of Prescription of Magnesium Sulfate to have Controlled Hypotension during Septorhinoplasty
Shideh Marzban1, Soudabeh Haddadi1*, Arman Parvizi1, Ali Faghih Habibi2, Maedeh Zakeri Dana1, Zahra Atr Kar Roshan1 and Hajian AB1
*Correspondence: Soudabeh Haddadi, Department of Anesthesiology, Alzahra Hospital, Guilan University of Medical Sciences, Rasht, Iran, Email:
Abstract
Introduction: Controlled hypotension is a technique to decrease intra-operative bleeding. Magnesium sulfate would be a compatible choice for this target.
Aim: This study was conducted to evaluate effect of magnesium sulfate on controlling blood pressure during septorhinoplasty.
Materials and methods: In this double-blind randomized clinical trial study, 60 patients aged 18-50 years with ASA class I and II, underwent septorhinoplasty. They were randomly divided into two groups: magnesium sulfate (M) and control (C). In Group M, diluted magnesium sulfate (50%) (40 mg/kg in 100 ml of normal saline) was infused intravenously in 10 minutes, prior to the induction of general anesthesia then followed by 15 mg/kg/hr administration during surgery. In Group C, same volume of normal saline 0.9% was infused. Anesthesia was conducted through intravenous infusion of propofol (50 μg/kg/min) and remifentanil (0.1 μg/kg/min) for all. The mean arterial pressure was maintained 60-65 mm Hg mm Hg by injection of nitroglycerine (25-50 μg/min), if needed. Data was analyzed considering significant p<0.05.
Results: Most of participants were female (55 persons-91.7%). Mean length of surgery was 72.2 ± 14.8 min. Over 76% of controls (23 patients) needed to nitroglycerin infusion, while to none of magnesium receivers, nitroglycerin was delivered (p=0.001). Although, mean heart rate was different significantly between two groups prior to anesthesia induction (p=0.002), but its changes between two groups were not obvious at any other time of measurements. The incidence of pain feeling also was significantly lower in magnesium receivers in recovery room. There was no significant difference in amount of bleeding and surgeon satisfaction between groups.
Conclusion: Magnesium sulfate seems to be effective in inducing hypotension and accompanied with lower postoperative pain.
Keywords
Magnesium sulfate, Controlled hypotension, Septorhinoplasty
Introduction
All surgical procedures have a possibility for complication. Rhinoplasty is estimated to be associated with severe systemic or lifethreatening complications in 1.7–5% of all cases. Complications of rhinoplasty may be evaluated in four main categories according to the time as intraoperative (such as bleeding), immediate postoperative (in PACU), early postoperative, and late postoperative [1]. One of the challenges of anesthesiologists in reconstructive, cosmetic and microscopic surgery (microsurgery) is providing conditions that can minimize bleeding in the surgical field and helping the surgeon to have better vision [2-4]. To do so, certain methods such as the injection of vasoconstrictors, e.g. epinephrine, creating sufficient depth of anesthesia with controlled respiration and controlled hypotension, are used [1]. In fact, bleeding control via controlled hypotension approach is a goal to have better outcomes in almost surgical procedures [4-6]. The acceptable rate for controlled to hypotension is to maintain MAP (Mean Arterial Pressure) by 30% less than the normal defenition or the least MAP from 50 to 80 mmHg for patients classified as ASA-I (American Society of Anesthesiologists) and older, respectively [4,6]. Different drugs have been used to control hypotension, which include magnesium sulfate [2,6,7], sodium nitroprusside [3], nicaridine [6], hydralazine [8], esmolol [3], α2 agonist [4,6,9,10], labetalol [11] and high doses of potential inhalation anesthetics, such as isoflurane [12,13]. Magnesium (Mg) is the fourth common cation in the body and activates approximately 300 enzyme pathways, many of which are involved in metabolism of energy. Mg is a physiologic blocker of calcium channels and a non-competitive antagonist of NMDA receptors (N-methyl-D-Aspartate). Theoretically, it can play an important role in the molecular sensitization process and adjust postoperative pain and tremor through the blockage of NMDA receptors [9]. Magnesium sulfate induces hypotension through vasodilation and muscle relaxation by restricting calcium excretion from the sarcoplasmic network, increasing prostacyclin production, inhibiting angiotensinconverting enzyme (ACE) and catecholamine release prevention. It makes better coronary artery perfusion as well [2,14] Intraoperative hypotension refers to the drug’s powerful anesthetic and negative inotropic effect [15,16]. The onset time of Mg is immediately after the injection and lasts for about 30 min. It’s excreted through the kidneys [17,18]. Intraoperative infusion of magnesium sulfate would reduce MAP and contributed to significant decrease in hemorrhage and following blood transfusion [18]. Data have showed that remifentanil and magnesium sulfate had equal potency in controlling MAP and the latter additionally was preferable to decrease postoperative pain reduction [19,20-22]. As one of the common surgeries worldwide, is septorhinoplasty and because of the above-mentioned points, this study was conducted to investigate effects of magnesium sulfate on hemodynamic status and bleeding control among patients underwent septorhinoplasty.
Materials and Methods
Current study was conducted as a double-blind randomized clinical trial from 2016 to 2017 in Amiralmomenin Hospital, Rasht, Iran;after obtaining the research approval from Vice- Chancellor for Research and Technology, Guilan University, and also Ethics Committee (Code: IR.GUMS.REC.1395.375) and receiving the IRCT code from the clinical trials registration system (code: IRCT2017042221436N3). Finally 60 patients between 18 and 50 years of age, with ASA class I and II physical status, candidate for elective septorhinoplasty under general anesthesia after siging written consent to participate included in the study (Inclusion criteria). They all experienced general anesthesia and were operated by a single anesthetic and surgical team. Exclusion criteria were included of the history of heart, kidney, hematologic and neuromuscular disease, anemia, diabetic neuropathy, obesity (BMI>35), pregnancy, positive allergy history to magnesium or other drugs used in the study, calcium channelblocker, opioid, anticoagulants and magnesium supplement intake, and also unwillingness to participate. Patients were randomly divided into two groups with 30 persons in each, cases were magnesium sulfate-remifentanilpropofol (Group M) and for controls normal saline-remifentanil-propofol (Group C). The goal was to maintain systolic blood pressure (SBP) and MAP in range of 80-85 and 60-65 mm Hg, respectively. In operating room electrocardiography, SBP, MAP, and peripheralcapillary oxygen saturation (SPO2) percentage were monitored (Saadat-Iran). In both groups, general anesthesia (GA) was induced byMidazolam (0.02 mg/kg), fentanyl (2 μg/kg), lidocaine (1 mg/kg) and propofol (2-2.5 mg/kg). Endotracheal intubation was facilitated using atracurium besylate (0.5 mg/ kg) injection. O2 was prescribed as 4 l/min, expiratory volumeregulated at 6-8 ml/kg and respiratory rate maintained 10-12 breaths/ minute with alternating intermittentpositive pressure ventilation mode (IPPV). Cases were received diluted magnesium sulfate 50% (40 mg/kg) in 100 ml of 0.9% saline solution intravenously just 10 minutes before anesthesia induction following by 15 mg/kg/hr maintaining dose under continuous cardiac monitoring (HR: heart rate, SBP, MAP). Controls were received same volume of 0.9% normal saline solution. Maintenance of anesthesia in all patients was achieved by infusion of propofol (50 μg/kg/min) and remifentanil (0.1 μg/kg/min). In order to control overt hypotension (MAP≤60 mmHg) and bradycardia (HR<60), 5 mg of ephedrine and 0.5 mg of atropine were prescribed, subsequently if needed. Vital sign was recorded initially (before induction of anesthesia) as the baseline, immediately after inducing anesthesia (minute 0) and after wards every 5 min until the emergence. A single anesthesiologist was the only person who was aware of groups and type of the drug used. Amount of intra-operative bleeding in ml was measured by adding the number of filled bloody gauzes (10 mL) plus the blood existing in the suction device. Total bleeding volume was compared with Maximal Allowable Blood Loss- MABL (<10%, 10-20% and >20% of allowed bleeding). Length of surgery (minutes) and intraoperative nitroglycerin usage to maintain controlled hypotension were other recorded variables. Surgeon's satisfaction of the operation field was assessed on the scale of 0 to 10 based on the Beozaart scale [13] Serum magnesium level(mg/dL) was measured for group M about 30 min prior to estimated time for end of surgery. Either there was sign of drug toxicity or magnesium serum level ≥ 10 mg/dL, injection of magnesium was ceased and calcium gluconate 10% (10 ml) was intravenously injected slowly and repeated if needed. In post-anesthesia care unit, patients were observed for pain score on a 10 mm visual analog scale (VAS) every 10 min (0 =No pain,10=Unbearable pain) for three times. Pain (3<VAS) was treated using intravenous meperidine (0.5 mg/kg).
Statistical analysis
Data was analyzed by SPSS v.21. Demographic data and differences between two groups were assessed using unpaired student’s t-test and Chi-squared test. Vital signs were compared by repeated measures ANOVA. Comparison of changes in VAS was performed using the Wilcoxon test within each group. In the case of normality, t-test was used for data based on Kolmogorov–Smirnov test and, otherwise (nonnormality), Mann-Whitney U test statistical test was applied between the two studied groups. For comparing side effects, Chi-squared test was used. All data were presented as mean± SD and/ or percent. P-value <0.05 was considered statistically significant.
Results
Table 1 shows demographic characteristics of participants.
| Group M | Group C | Total | P | |||
|---|---|---|---|---|---|---|
| Gender | Male | No (%) | 3 (10) | 2 (6.7) | 5 (8.3) | 0.64 |
| Female | 27 (90) | 28 (93.3) | 55 (91.7) | |||
| ASA class | I | 26 (89.7) | 29 (96.7) | 55 (93.2) | 0.284 | |
| II | 3 (10.3) | 1 (3.3) | 4 (6.8) | |||
| Nitroglycerin usage | No | 30 (100) | 7 (23.3) | 37 (61.7) | 0.0001 | |
| Yes | 0 (0) | 23 (76.7) | 23(38.3) | |||
| Dosage of nitroglycerin (µg) | Mean ± sd | 0±0.00 | 280 ± 327.37 | 140 ± 269.46 | - | |
| Age (year) | 26.53±4.73 | 28.70 ± 6.24 | 27.62 ± 5.60 | 0.135 | ||
| Weight (kg) | 57.60 ± 11.4 | 61.77±12.47 | 59.68±11.86 | 0.176 | ||
| Duration of surgery (min) | 72.25 ± 14.77 | 73.17±13.93 | 71.35±15.75 | 0.64 | ||
Table 1: Demographic characteristics of the studiedpatients.
Data showed that the HRdifference between the two groups was statistically significant just before the general anesthesia induction (p=0.002). Although, repeated measures ANOVA showed no obvious difference for HR in time points after GA induction between both groups, but according to Mauchly's test and Greenhouse- Geisser trend to decrease changes between groups was significantly different (p<0.001) (Table 2).
| Group M | Group M | P* | ||
|---|---|---|---|---|
| Heart Rate (beats/min) | Mean ± SD | |||
| Before GAinduction | 91.43 ± 14.99 | 105 ± 17.15 | 0.002 | |
| After GAinduction | 89.23 ± 15.84 | 87.40 ± 12.36 | 0.619 | |
| 5 min | after GA | 87.43 ± 21.38 | 81.50 ± 13.10 | 0.2 |
| 10 min | 79.77 ± 12.56 | 79.87 ± 12.85 | 0.976 | |
| 15 min | 76.80 ± 12.48 | 80.90 ± 10.43 | 0.173 | |
| 20 min | 75.90 ± 10.54 | 78.70 ± 11.34 | 0.326 | |
| 25 min | 75.67 ± 11 | 75.87 ± 10.23 | 0.942 | |
| 30 min | 74.30 ± 13.28 | 77.33 ± 9.88 | 0.32 | |
| 45 min | 72.30 ± 10.31 | 75.27 ± 11.49 | 0.297 | |
| 60 min | 71 ± 9.50 | 75.20 ± 9.07 | 0.085 | |
| P** (Interaction of time and group) | Observed power=0.987, Partial Eta Squared (Effect size)=0.081 | 0.001 | ||
| P** (Time effect) | 0.001 | |||
| P** (Group effect) | 0.366 | |||
| P*=Independent t-test; P**=Repeated measures ANOVA; Greenhouse-Geisser; SD=Standard Deviation | ||||
Table 2: Comparing HR during the study time points.
SBP records demonstrated no statistically significant difference at any of the measurement times between two groups. Results for MAP for both groups revealed a decreasing trend prior to the end of the study (p<0.001). Generally, either SBP findings or the interaction between time and group effects were not different during the study time points (p=0.8). The DBP findings were also similar to those of SBP (p>0.05). However, the trend of DBP change in both groups was to decrease from the time before the induction of anesthesia until minute 60 of surgery (p<0.001). No significant difference for MAP was reported between two groups.
Although lower VAS was detected among magnesium sulfate receiver than do in controls but there was no difference in recovery room (p>0.05). Severe pain requiring intervention (3<VAS) at the first 10 min of recovery time among magnesium receivers (3.3%) was significantly lesser than do in controls (30%) (p=0.001). While there was no obvious difference was seen between participants in case of pain felling (p=0.4) generally, the interaction between time and group was showed no difference either (p=0.6) (Figure 1).
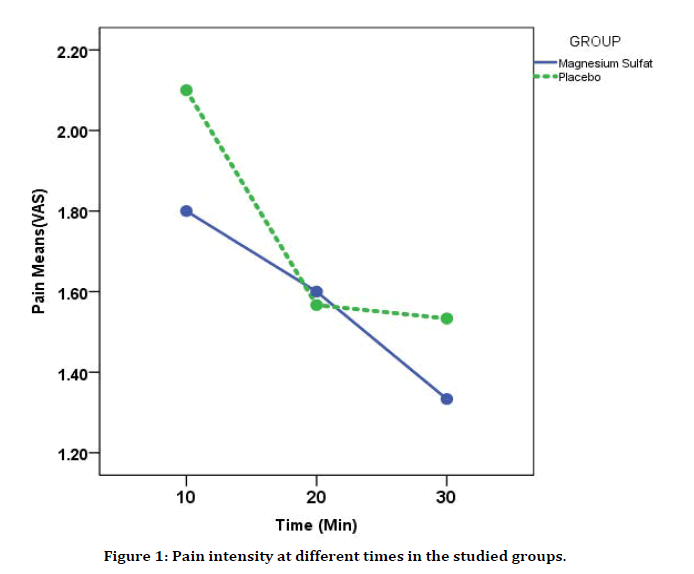
Figure 1. Pain intensity at different times in the studied groups.
Drug induced complications including bradycardia, nausea and shivering had same prevalence in both groups(p>0.05). The amount of bleeding and satisfaction of surgeon was similar with no statistically significant difference. Need for nitroglycerin injection was significant (p<0.001), despite 76.7% of controls, no member of magnesium receiver underwent injection (Table 3).
| Group C | Group M | P | |||
|---|---|---|---|---|---|
| No (%) | No (%) | ||||
| Bleeding based on MABL percentage | <10 | 6 (20) | 6 (20) | 0.8 | |
| 10-20 | 10 (33.3) | 8 (26.7) | |||
| >20 | 14 (46.7) | 16 (53.3) | |||
| Surgeon Satisfaction | 0-1 | 0 (0) | 2 (2.7) | 0.6 | |
| 2-3 | 11 (36.7) | 11 (36.7) | |||
| 4-5 | 15 (50) | 12 (40) | |||
| 6-7 | 3 (10) | 4 (13.3) | |||
| 8-9 | 1 (3.3) | 1 (3.3) | |||
| Treatment in Recovery unit (every 10 minutes) |
1st | Ondansetron(4 mg) | 0 (0) | 0 (0) | - |
| Warmer | 1 (8.3) | 1 (25) | 0.9 | ||
| Atropine (0.5 µg) | 0 (0) | 1 (25) | 0.5 | ||
| Pethidine(30 mg) | 11 (91.7) | 2 (50) | 0.005 | ||
| 2nd | Ondansetron | 0 (0) | 0 (0) | - | |
| Warmer | 0 (0) | 0 (0) | - | ||
| Atropine | 0 (0) | 0 (0) | - | ||
| Pethidine | 2 (100) | 3 (100) | 0.5 | ||
| 3rd | Ondansetron | 0 (0) | 0 (0) | - | |
| Warmer | 0 (0) | 0 (0) | - | ||
| Atropine | 0 (0) | 0 (0) | - | ||
| Pethidine | 1 (100) | 1 (100) | 0.9 | ||
Table 3: Drug consumption, hemorrhage rate and surgeon satisfaction.
The mean duration of surgery was 72.2 ± 14.8 min with no obvious difference between groups. Calculated serum level of Mg among cases was 3.5±0.49 mg/dL (minimum: 2.4 and maximum: 4.2). These were all in safe window and no drugtoxicity was expected.
Discussion
This study investigated outcomes of magnesium sulfate on having controlled hypotension intraoperatively, and also postoperative pain feeling in case of elective septorhinoplasty. Our findings were in lined with other study that showed the positive effect of magnesium sulfate on making controlled hypotension in mastoidectomy. Surprisingly, current study has revealed magnesium was preventive to use nitroglycerine injection in order to control the blood pressure. It may be of the vasodilatory effect and also inhibitory role of ACE enzyme by magnesium sulfate resulting in MAP decrease [2].
Although, this study showed that just prior to induction of general anesthesia, HR differentiated between groups significantly, but it had decrescendo trend in both groups, probably due to anesthetic agents. In contrast, an author reported that mean HR among controls was obviously less during rhinoplasty (P=0.001) [2]. The study mentioned that the decreasing HR in magnesium receiver was fairly constant, however, it was fluctuated in controls needed to usual nitroglycerine injection. The difference might be due to stabilizing effect of magnesium sulfate on cardiocytes through inhibitory role of calcium channels function [14,23].
Recorded changes in blood pressure, neither for SBP nor for DBP was not divergent between groups whether pre or intra-operatively, however, blood pressure tended to be decreased in all participants, probably due to anesthesia effects. Our findings showed similar results for MAP, as for blood pressure, in contrast with the other statement said that magnesium sulfate prescription was contributed to lowering MAP [24]. Regarding this contrast, the probable causes may include injected dose of Mg, patient’s response characteristics to the drug and also anesthesia effect. The pain feeling score for two groups during the first 30 minutes of recovery time was equivalent, although it was a little more among controls. Nevertheless, in first postoperative 10 minutes, magnesium receivers suffered lesser than do controls obviously. This was in lined with other findings [21].
Magnesium sulfate was not accused for complication in this study. Furthermore, some advocates showed postoperative nausea and vomiting, shivering and pain were even considerably lower in magnesium receivers [24].
Present study revealed that operative hemorrhage and surgeon satisfaction from field of surgery were similar among all participants. These were incompatible with those of others who reported that either the lesser blood loss or surgeon satisfaction were associated with Mg prescription [2,24,25]. Although, the role of magnesium sulfate in BP reduction is well understood but contrast findings imply on to perform further investigations to bright facts.
Conclusion
This study showed that prescription of Magnesium sulfate was associated with lower hemodynamic fluctuations and decreasing requirement for antihypertensive agents. Magnesium sulfate seems to be effective in inducing hypotension and accompanied with lower postoperative pain.
Conflict of Interest
There was no conflict of interest.
References
- M Kar, C Cingi, R Thomas. All around the nose. Springer 2020.
- Rahimi M, Yaraghi A, Hashemi SJ, et al. Effects of magnesium sulphate in deliberated hypotension for mastoidectomy surgery. J Isfahan Med School 2007; 25:23-31.
- Degoute CS, Ray MJ, Manchon M, et al. Remifentanil and controlled hypotension: Comparison with nitroprusside or esmolol during tympanoplasty. Can J Anaesth 2001; 48:20–27.
- Tobias JD. Controlled hypotension in children: A critical review of available agents. Paediatr Drugs 2002; 4:439–53.
- Rodrigo C. Induced hypotension during anesthesia with special reference to orthognathic surgery. Anesth 1995; 42:41-58
- Degoute CS. Controlled hypotension: A guide to drug choice. Drugs 2007; 67:1053–1076.
- Homaee MM, Farazmehr K, Soleimani M, et al. Blood loss and quality of controlled hypotension induced by magnesium sulfate vs. remifentanil in PSF surgery. Anesth Pain Official J ISRAPM 2012; 2:52-61.
- Khamestan M, Samadi K, Forouzan M. Evaluating effect of Remifentanil infusion vs. single dose intravenous hydralazine plus propranolol to provide controlled hypotension in patients undergoing rhinoplasty in Khalili hospital of Shiraz. J Anesth Pain 2016; 6:21-29.
- Davoudi M, Zolhavareih T. Evaluation effect of intravenous magnesium sulfate on postoperative pain after cesarean section under spinal anesthesia. Sci J Hamadan University Med Sci 2013; 19:20-26.
- Taghi MM, Ansari M, Gojazadeh M, et al. Results and consequences during and after rhinoplasty. Med J Tabriz University Med Sci 2012; 35:88-91.
- Helen H, Safarlo F. Investigating level of consciousness and remembering in rhinoplasty surgery with local anesthesia and venous sedation. Med Sci J Islamic Azad University 2010; 20:131-135.
- Prasant MC, Rastogi S, Hada P, et al. Comparative study of blood loss, quality of surgical field and duration of surgery in maxillofacial cases with and without hypotensive anesthesia. J Int Oral Health 2014; 6:18-21.
- Marzban S, Haddadi S, Mahmoudi Nia H, et al. Comparing surgical conditions during propofol or isoflurane anesthesia for endoscopic sinus surgery. Anesth Pain Med 2013; 3:234-238.
- Reinhart RA. Clinical correlates of the molecularand cellular actions of magnesium on the cardiovascular system. Am Heart J 1991; 121:1513-21.
- Yosri M. Controlled hypotension in adults undergoing choroidal melanoma resection: Comparison between efficacy of nitroprusside and magnesium sulfate. Eur J Anaesth 2008; 25:891–896.
- Sanders GM, Sim KM. Is it feasible to use magnesium sulfate as a hypotensive agent in oral and maxillofacial surgery? Annals Academic Med 1998; 27:780-785.
- Buvanendran A, Kroin JS. Intrathecal magnesium prolongs fentanyl analgesia: A prospective, randomized, controlled trial. Anesth Analg 2002; 95:661-6.
- https://www.medscape.com.
- Sadrosadat SH, Zehtab MJ, Imani. F, et al. A systematic review of methods designated for control of bleeding during major operations, especially those performed upon the spinal column. Iranian J Surg 2006; 14:1307-1315.
- Juibari HM, Arabion HR. Intravenous magnesium sulfate to deliberate hypotension and bleeding after bimaxillary orthognathic surgery: A randomized double-blind controlled trial. J Dent Shiraz Univ Med Sci 2016; 17:276-82.
- Levaux C, Dewandre PY, Brichant JF. Effect of intra-operative magnesium sulfate on pain relief and patient comfort after major lumbar orthopedic surgery. Anesth 2003; 58:131-135.
- Telci L, Esen F, Akcora D, et al. Evaluating effects of magnesium sulfate on reducing intraoperative anesthetic requirements. Br J Anaesth 2002; 89:594-8.
- Mireskandari SM, Jafarzadeh A, Makarem J, et al. The effectiveness of intravenous magnesium sulfate for deliberate hypotension in rhinoplasty. Archives Anesth Critical Care 2015; 1:112-115.
- Abbasi H, Behdad S, Ayatollahi V, et al. Comparison oftwodoses of tranexamicacid on bleeding and surgery site quality duringsinus endoscopy surgery. Advances Clin Exp Med 2011; 21:773-780.
- Gyamlani G, Parikh C, Kulkarni AG. Benefits of magnesium in acute myocardial infarction: Timing is crucial. Am Heart J 2000; 139:703-711.
Author Info
Shideh Marzban1, Soudabeh Haddadi1*, Arman Parvizi1, Ali Faghih Habibi2, Maedeh Zakeri Dana1, Zahra Atr Kar Roshan1 and Hajian AB1
1Department of Anesthesiology, Alzahra Hospital, Guilan University of Medical Sciences, Rasht, Iran2Department of ENT, Head and Neck Surgery, Amiralmomenin Hospital, Guilan University of Medical Sciences, Iran
Citation: Shideh Marzban, Soudabeh Haddadi, Arman Parvizi, Ali Faghih Habibi, Maedeh Zakeri Dana, Zahra Atr Kar Roshan, Hajian AB, Effect of Prescription of Magnesium Sulfate to have Controlled Hypotension during Septorhinoplasty, J Res Med Dent Sci, 2020, 8(1):181-186.
Received: 14-Oct-2020 Accepted: 07-Feb-2020
