Research - (2021) Volume 9, Issue 4
Effects of Bronchodilator in Obstructive Airway Diseases
Aparajeet Kar, Suryanarayana V and Krishnarajasekhar OR*
*Correspondence: Krishnarajasekhar OR, Department of Respiratory Medicine, Sree Balaji Medical College & Hospital, Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, India, Email:
Abstract
The present study was a prospective study to assess the severity of Obstructive Airway Disease in patients attending our outpatient department and to assess the Bronchodilator response in terms of change in FEV1 in such patients to a single dose of salbutamol nebulization. FEV 1% Post-bronchodilator changes is insignificant in patients suffering from Bronchial Asthma with older age group. FEV 1% Post-Bronchodilator changes is significant in patients suffering from COPD GOLD Stage I and II. No significant post-Bronchodilator FEV1% changes were seen in patients suffering from COPD GOLD stage III and IV. There were no significant Post-Bronchodilator FEV1% changes in patients suffering from Bronchiectasis, Post-TB OAD. The effect of Bronchodilator is not found to be useful in Bronchiectasis Salbutamol is a potent drug for treating patients with Bronchial Asthma and COPD stage I and II.
Keywords
Bronchodilator, OAD, Salbutamol, Emphysema, Spirometry, Chronic, Airway
Introduction
Obstructive Airway Disease (OAD) is a disease characterized by airflow limitation that is either fully or partially reversible or not reversible. The diagnosis of OAD is based on spirometry evidence of airflow obstruction. Evidence now suggests that a considerable proportion of patients thought to be having irreversible airway obstruction show significant bronchodilator reversibility after using medication for some time. The complexity and inherent variability of a patient’s acute response to a bronchodilator and the lack of a standardized procedure for assessing bronchodilator reversibility have led to significant confusion surrounding this concept. Although bronchodilator reversibility commonly is defined based on improvement in FEV 1, lung volume-based measures of pulmonary function may be of particular importance in patients with severe OAD. The usefulness of acute reversibility to bronchodilators in predicting a patient’s long-term response to bronchodilator maintenance therapy is also unclear, although most studies suggest that a lack of acute response to bronchodilators does not preclude a beneficial long-term response to maintenance bronchodilator treatment. At this juncture, we can recollect the GOLD guidelines revised definition of COAD emphasizing its “partially reversible” nature with a corresponding sense of hope replacing the old sense of despair with regards to the management of COAD [1-7].
We grouped the patients with OAD as those diagnosed as:
✓ Emphysema
✓ Chronic Bronchitis
✓ Bronchial Asthma
✓ Bronchiectasis
✓ Post Tuberculosis- Bronchiectasis
Chronic Bronchitis and emphysema are the two diseases which go hand in hand and mostly co-exist in almost all the patients with these diseases with one of them as the predominant one and grouped by some as Chronic obstructive pulmonary disease (COPD), and is characterized by airflow limitation that is usually partially reversible in some patients only and progressive and associated with an enhanced chronic inflammatory response to noxious substances. Exacerbations and comorbidities contribute to the overall severity and prognosis in individual patients."
Chronic bronchitis—Chronic bronchitis is a clinical diagnosis and can be defined as a chronic productive cough on most days for three months in each of two successive years in a patient in whom other causes of chronic cough such as tuberculosis and bronchiectasis have been excluded. “COPD is a disease characterized by the partially reversible obstruction of the airways. The obstruction is usually progressive, with an abnormal inflammatory response to the noxious particles and gases, associated with systemic manifestations” [1-20].
Emphysema—Emphysema is a pathological diagnosis and can be defined as abnormal and permanent enlargement of the airspaces distal to the terminal bronchioles that is accompanied by destruction of the airway walls, without obvious fibrosis (ie, there is no fibrosis visible to the naked eye). Asthma - The Global Initiative for Asthma gives the following definition of asthma: "Asthma is a heterogenous disease, usually characterized by chronic airway inflammation. It is defined by history of respiratory symptoms such as wheeze, chest tightness, shortness of breath and cough that vary over time and intensity, together with variable expiratory airflow limitation” [21]. Bronchiectasis–Bronchiectasis is characterized by abnormal, permanent dilation and distortion of the bronchial tree, resulting in chronic obstructive lung disease. This condition is the result of a variety of pathophysiologic processes, usually including some combination of infection and impaired mucus drainage or airway obstruction. The condition may be confined to a single pulmonary lobe or segment or may involve multiple pulmonary segments or lobes.
Post-TB Bronchiectasis- In our study some patients who have taken treatment for Pulmonary Tuberculosis previously or have taken inadequate treatment showed variable amount of obstruction. Post-TB Bronchiectasis is manifested by irreversible dilatation of bronchi, destruction of elastic and muscular elements of bronchial walls. In this study we observe and relate the effects of bronchodilator i.e., Nebulized Salbutamol and FEV1 changes in the patients with OAD.
Materials and Methods
The present study “Effects of Bronchodilator in Obstructive Airway Diseases” was conducted in the Department of Pulmonary Medicine, Sree Balaji Medical College and Hospital, Chromepet, Chennai after obtaining the approval from Institutional Human Ethical committee.
Study period
The study is carried from April 2014 to November 2015.
Sample size
A total of 40 patients were enrolled for the study. Patients aged between 18 years to 80 years visiting the outpatient department of Pulmonary Medicine were taken up for study. Detailed history of these patients was taken and patients with symptoms of breathelessness, cough, chest pain were asked to undergo Chest X-ray and Pulmonary function test. CT Chest was done in a few as and when required. Detailed history of smoking and other occupational exposures were also recorded. Thorough clinical examination was done. Patients were selected on basis of the following inclusion and exclusion criteria.
sInclusion criteria
Patients more than 18 years of age.
Patients willing to be included in the study and willing for the investigations.
Patients with features of COPD-bronchitis and Emphysema, Bronchiectasis, Post –TB OAD status.
Patients with history of breathelessness with features suggestive of Obstruction in spirometry.
Exclusion criteria
Patients who came with Acute Exacerbation.
Recent myocardial infarction, arrhythmias, active hemoptysis.
Patients aged below 18 years of age and more than 80 years.
Patients who underwent recent surgeries, intracranial and ophthalmic surgery.
Patients unable to do spirometry.
Consent
An informed written consent was taken from all the patients enrolled in the study after a proper health education.
Methods
Patients who were fitting into the inclusion and exclusion criteria were taken into the study.
The following investigations were done in all the patients:
Routine investigations
Complete blood picture.
Erythrocyte sedimentation rate (ESR).
Random blood sugar.
Complete urine examination.
Chest X-ray P.A. view.
ECG.
HIV, HBsAg.
Sputum for acid fast bacilli.
CT Chest as and when required.
Pulmonary function test: Patients were asked to undergo spirometry and FEV1% were measured. Patients with FEV1% less than 80% were given Salbutamol nebulization and Spirometry was repeated. The changes in Pre- Bronchodilator and Post-Bronchodilator FEV1% were measured.
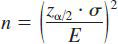
Where
n=Sample size
σ=Population standard deviation e=Margin of error
Z=The value for the given confidence interval the confidence level is estimated at 95% Standard deviation 23.6 with a z value of 1.96
The confidence interval or margin of error is estimated at +/- 8 Assuming that 80 percent as power of the study, minimum sample size required for the study was calculated to be 34. In our study 40 subjects were chosen.
In our study out of 40 patients 8 patients were in the age group of (18-40) years, 19 were in the age group of (41-60) years and 13 were in (61- 80) years of age. In our study while analysing the age distribution, majority of the study subjects belonged to the 41-60 years age class interval (n=19, 47.50%) followed by (61-80) years age class interval (n=12, 30.00%) (Table 1 and Figure 1). In our study amongst the 40 patients 19 were male and 21 were female (Table 2 and Figure 2).
Table 1: Age distribution.
| Age | 18-40 Years | 41-60 Years | 61-80 Years |
|---|---|---|---|
| Total | 8 | 19 | 13 |
Table 2: Gender distribution–groups.
| Male | Female |
|---|---|
| 19 | 21 |
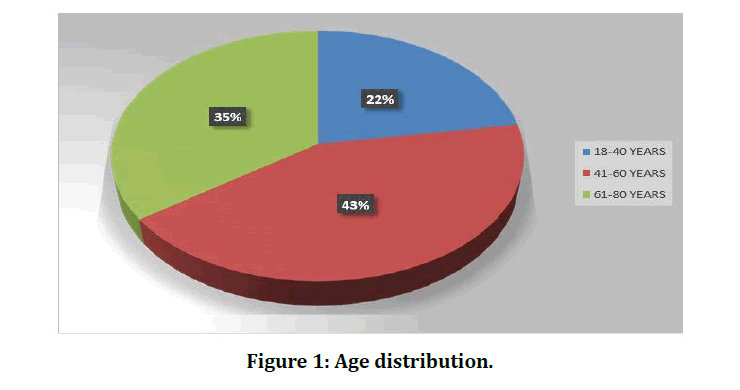
Figure 1: Age distribution.
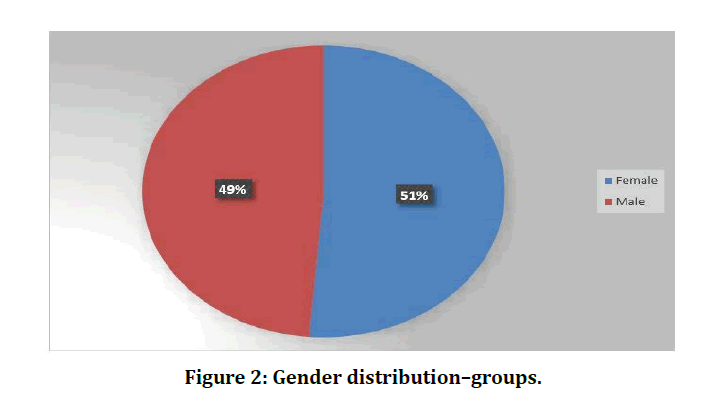
Figure 2: Gender distribution–groups.
In our study while analysing the symptom status, majority of the study subjects expressed breathlessness and cough as principal symptoms (Table 3 and Figure 3). In our study while analysing the duration of illness distribution, majority of the study subjects belonged to the ≤ 5 years duration of illness class interval (n=19, 47.50%) followed by 6-10 years duration of illness class interval (n=12, 30%) (Table 4 and Figure 4). In our study while analysing the smoking status, majority of the study subjects were nonsmokers (n=26, 65%) followed by smokers (n=14, 35%) (Table 5 and Figure 5).
Table 3: Symptoms.
| Symptoms | Cough | % | Fever | % | Breathlessness | % |
|---|---|---|---|---|---|---|
| Yes | 40 | 100 | 20 | 50 | 40 | 100 |
| No | 0 | 0 | 20 | 50 | 0 | 0 |
| Total | 40 | 100 | 40 | 100 | 40 | 100 |
Table 4: Duration of illness.
| Duration of Illness | Duration of Illness | % |
|---|---|---|
| ≤ 5 years | 19 | 47.5 |
| 6-10 years | 12 | 30 |
| 11-15 years | 5 | 12.5 |
| 16-20 years | 3 | 7.5 |
| >20 years | 1 | 2.5 |
| Total | 40 | 100 |
Table 5: Smoking status.
| Smoking Status | Number | % |
|---|---|---|
| Smoker | 14 | 35 |
| Non-Smoker | 26 | 65 |
| Total | 40 | 100 |
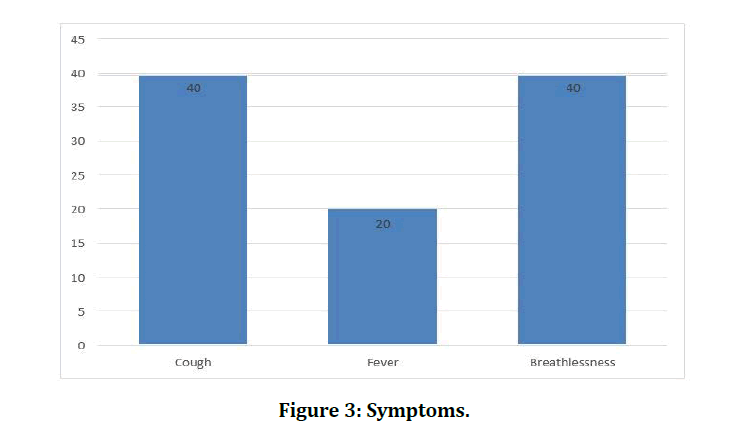
Figure 3: Symptoms.
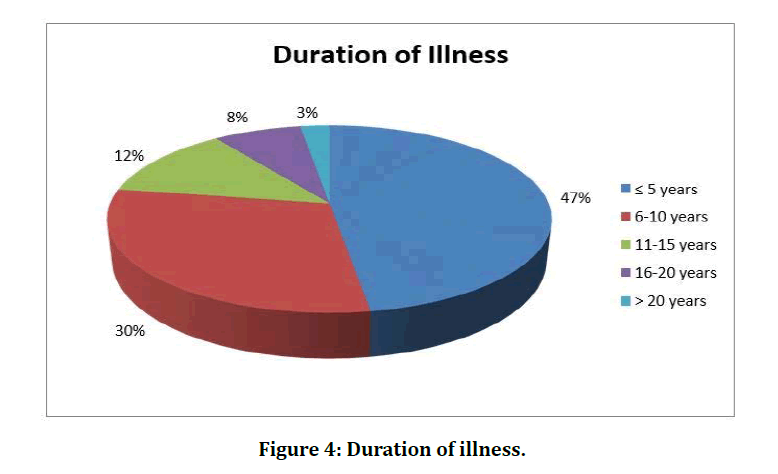
Figure 4: Duration of illness.
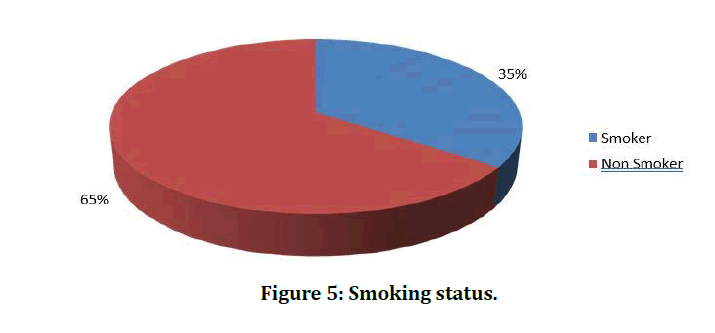
Figure 5: Smoking status.
Discussion
In our study we have observed that patients with lesser age group tend to show significant Post- Bronchodilator changes when compared to older age groups. In our study we have observed that there was more of female preponderance in the group of Bronchial Asthma and Bronchiectasis which has been also seen in many other studies. We have also observed that the patients with shorter duration of illness tends to show increased amount of Post bronchodilator changes when compared to those with chronic duration. In our study we have observed that non-smokers tend to show significant amount of Positive bronchodilator changes when compared to smokers. In our study Asthma group showed a significant Post-Bronchodilator FEV1% change by 12% and more which most of studies done previously co-relates. COPD GOLD Stage I showed significant Post- Bronchodilator FEV1% change by 12 % and more COPD GOLD stage II showed significant Post- Bonchodilator FEV1% changes in most of the patients by 12% We have observed in our study that no significant changes were seen in COPD GOLD Stage III and IV. We also observed that no significant changes were seen in Bronchiectasis and Post-TB obstructive airway disease [22-24].
Conclusion
The following conclusions were drawn after the statistical analysis of the data: FEV1% Postbronchodilator changes is insignificant in patients suffering from Bronchial Asthma with older age group FEV1% Post-Bronchodilator changes is significant in patients suffering from COPD GOLD Stage I and II. No significant post-Bronchodilator FEV1% changes were seen in patients suffering from COPD GOLD stage III and IV. There were no significant Post-Bronchodilator FEV1% changes in patients suffering from Bronchiectasis, Post - TB OAD. Effect of Bronchodilator is not found to be useful in Bronchiectasis. Salbutamol is a potent drug for treating patients with Bronchial Asthma and COPD stage I and II.
Funding
No funding sources.
Ethical Approval
The study was approved by the institutional ethics committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgement
The encouragement and support from Bharath University, Chennai is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- https://www.who.int/whr/2000/en/
- https://www.who.int/en/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
- Viegi G, Maio S, Pistelli F, et al. Epidemiology of chronic obstructive pulmonary disease: health effects of air pollution. 2006; 11:523-532.
- White paper of the specialty of physical medicine and rehabilitation in Europe-European union of specialist physicians (UEMS)-Section of physical medicine and rehabilitation, "Carol Davila" University Publishing House, Bucharest, 2006.
- https://www.uptodate.com/contents/chronic-obstructive-pulmonary-disease-definition-clinical-manifestations-diagnosis-and-staging
- Redman BK. The practice of patient education: a case study approach. 10th Edn St. Louis, Mosby, 200., Mosby;
- https://ginasthma.org/
- National asthma education and prevention program: Third expert panel on the diagnosis and management of asthma. Bethesda (MD): National Heart, Lung, and Blood Institute (US) 200.
- Marandi Y, Farahi N, Hashjin GS. Asthma: Beyond corticosteroid treatment. Archives Med Sci 2013; 9:521.
- Partridge MR. Examining the unmet need in adults with severe asthma. European Respiratory Review 2007; 16:67-72.
- https://www.who.int/publications/journals/bulletin/
- http://globalasthmareport.org/
- Yıldız F, ASIT study group. Factors influencing asthma control: results of a real-life prospective observational asthma inhaler treatment (ASIT) study. J Asthma Allergy 2013; 6:93.
- James T, Fine M. Monitoring asthma control using claims data and patient-reported outcomes measures. Pharmacy Therapeut 2008; 33:454.
- Agarwal R, Dhooria S, Aggarwal AN, et al. Guidelines for diagnosis and management of bronchial asthma: Joint ICS/NCCP (I) recommendations. Lung India: Official Organ of Indian Chest Society. 2015; 32:S3.
- Neves PC, Guerra M, Ponce P, et al. Non-cystic fibrosis bronchiectasis. Interactive Cardiovas Thoracic Surg 2011; 13:619-25.
- Rosen MJ. Chronic cough due to bronchiectasis: ACCP evidence-based clinical practice guidelines. Chest 2006; 129:122S-31S.
- Salkin D. Tuberculosis as a cause of upper lobe bronchiectasis. California Med 1950; 73:577.
- Rao PV, Rao GN, Bhanu P, et al. A clinical study of post-tubercular bronchiectasis. Indian J Mednodent Allied Sci 2014; 2:1-5.
- https://www.hopkinsmedicine.org/health
- Ingram RH. Chronic bronchitis, emphysema, and airways obstruction. In: Wilson JD, Braunwald E, Isselbacher KJ, et al. Eds. Harrison’s Principles of internal medicine. 13th Edn. New York: McGraw-Hill, 1994; 1197–205.
- https://goldcopd.org/gold-reports/
- https://ginasthma.org/pocket-guide-for-asthma-management-and-prevention-in-children-5-years-and-younger/
- Montuschi P, Malerba M, Santini G, et al. Pharmacological treatment of chronic obstructive pulmonary disease: From evidence-based medicine to phenotyping. Drug Discovery Today 2014; 19:1928-35.
Author Info
Aparajeet Kar, Suryanarayana V and Krishnarajasekhar OR*
Department of Respiratory Medicine, Sree Balaji Medical College & Hospital, Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Aparajeet Kar, Suryanarayana V, Krishnarajasekhar OR, Effects of Bronchodilator in Obstructive Airway Diseases, J Res Med Dent Sci, 2021, 9 (4):257-262.
Received: 20-Mar-2021 Accepted: 12-Apr-2021
