Research - (2021) Volume 9, Issue 6
Incidence, Diagnosis and Treatment of Brain Tumours
Buthayna G Elshaikh1*, Hiba Omer1, MEM Garelnabi2, Abdelmoneim Sulieman3, Nawader Abdella1, Sahar Algadi1 and Hind Toufig4
*Correspondence: Buthayna G Elshaikh, Department of Basic Sciences, Deanship of Preparatory Year and Supporting Studies, Imam Abdulrahman bin Faisal University, Saudi Arabia, Email:
Abstract
This review article studies extensively brain tumours. Brain tumors are among the leading causes of death. Here we introduce the different types and grades of brain tumor, their causes, symptoms and worldwide distribution. The article also discusses the different diagnostic facilities starting from those that do not require radiation diagnostic imaging facilities such as testing physical and cognitive performance, electrical activities, chemical testing, and ultrasound. We then present diagnostic imaging for brain tumor including x-ray, CT scans, MRI and nuclear medicine imaging. Biopsy, which usually follows these exams, is also explained. It then furthers into explaining therapy of brain tumor for example, surgery, radiotherapy, radiosurgery, alternating electric field therapy and laser interstitial thermal therapy. Medicinal treatment, targeted therapy and immunotherapy are also briefly explained. Finally, examples for multi-modality or hybrid therapies are given.
Keywords
Brain tumors, Benign, Malignant, Diagnosis, Therapy
Introduction
The central nervous system (CNS) which consists of brain, spinal cord and associated nerve cells control the entire human body. The human brain is surrounded by cranial bones, meninges and cerebrospinal fluid (CSF), which provide protection and support. The main components of the brain are gray matter forms the outer cerebral cortex, and the white matter contains tracts called myelinated axons, based on their appearance [1]. The brain is anatomically divided into 3 regions and is composed of two categories neurons and glial cells. The former aids in communication and processing and the latter protect the neurons [2].
Brain tumours
The abnormal growth of tissue in the brain can seriously harm the function of the brain, and eventually the entire body. It is associated with progressive worsening of emotional and mental health status resulting in a poor quality of life. Brain tumor originating from the brain itself is defined as primary, otherwise it is called metastatic. Non-cancerous tumors are benign. They usually are localized, slow growing and have homogenous structure. Cancerous or malignant tumors on the other hand are non-homogenous in their structure, invasive with high proliferating rate [3].
Examples of brain tumours include
Benign: Chordomas, Glomus jugulare, Craniopharyngiomas, Meningiomas and pituitary adenomas.
Malignant brain tumors: The most common malignant tumors are Gliomas which originate from glial cells. There are many times of glial cells cancers including: Astrocytomas, Medulloblastomas and Glioblastoma multiforme
Pediatric brain tumors are commonly dependent on age. They are defined as supratentorial tumors that are common in infants and children up to 3 years of age and again after age 10; and infratentorial tumors which are common between ages 4 to 10 years.
Classification of brain tumours
According to the WHO classifications brain tumors are classified into four grades on histopathologic features.
Grades I (pilocytic astrocytoma) and II (astrocytomas and oligodendrogliomas) are usually benign. That is, they look quite normal under microscopic examinations and are slow growing. Life expectancy is more for patients with grades I and II. Grade II, which is more aggressive can convert into malignant brain tumors and may recur after treatment.
Grade III (anaplastic astrocytomas and oligodendrogliomas) and Grade IV (glioblastoma multiforme) are malignant, with IV being more aggressive and metastatic. They look abnormal under a microscope and have high recurrence rates. Prognosis of patients with these tumors are very poor [4-7] Figure 1 shows a scan of a normal brain. Figure 2 shows scans for the four grads of brain tumors.
Figure 1. Normal brain CT scan.
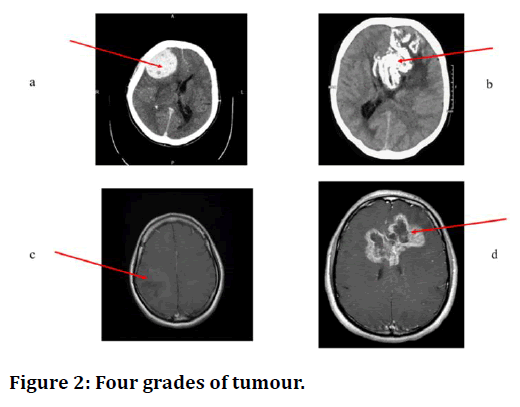
Figure 2. Four grades of tumour.
Contrast enhanced axial CT Image showing meningioma, a WHO grade I tumor [8].
CT axial Non-contrast Image showing oligodendroglioma, a WHO grade II tumor [9].
Axial MRI T1wI with contrast of anaplastic astrocytoma, a WHO grade III tumor [10].
MRI, axial T1WI with contrast showing a glioblastoma NOS, which is a WHO grade IV tumor [11].
Causes of brain tumours
The causes of brain tumours cannot be pointed precisely, although several risk factors are extensively studied. These include:
• Ionizing and non-ionizing radiation.
• Family history, genetic and racial factors.
• Life style.
• Nutrition.
• Alcohol.
• Smoking.
• Aspartame [12].
Symptoms of brain cancer
The symptoms of brain cancer depend on many factors including: The type and nature (benign or malignant) of tumour, its location and size. Generally, the symptoms include:
Severe headache and seizures, change in visual, audition, balance and other cognitive abilities, personality and mood changes including aggression and hallucination, in addition to excessive fatigue and reduced muscle control [13].
Demographic distribution of brain tumour
Brain tumors are among the highest (10th) causes of mortality. The incidences of primary malignant brain tumors are slightly more in men than women, while women have higher non-malignant tumors for example meningiomas. The rates are also higher in developed countries but this could be attributed to the advanced diagnostic facilities.
Metastatic brain tumors: Primary lung cancer, breast cancer, melanoma, renal cancer, and colorectal cancer are the most common primary cancers responsible for brain metastasis, where lung cancer seems to be the leading cause for metastases [14] presented a retrospective study on brain metastasis that compared the incidence based on: race, sex, age and grade of tumor at the diagnosis In their study there was a significant difference by race in metastasis from lung, melanoma and breast cancer; where the probability was significantly higher in African Americans compared to Caucasian American. This was opposite for metastases from renal cancer and no difference was realized for brain cancer metastases for colorectal cancer. The study also showed that men had higher probabilities of brain metastases for almost all type of systemic cancer except breast cancer and lung cancer compared to women. Regarding age, the probability of developing brain metastases was the highest for the patients with lung cancer diagnosed at age 40–49 years. For ages 50-59 the probability was higher for those suffering from with primary melanoma, renal or colorectal cancer. For relatively young ages, example 20-39 years primary breast cancer was the most effective cause of metastases. Except for melanoma, patients above 70 years had similar chances for all primary cancers, this category had lower probabilities for metastases. The probability for brain metastases was higher for patients with distant stage of primary melanoma (the highest probability for brain metastases), lung, breast, colorectal and kidney cancers [38]. Table 1 shows new cases and deaths from in 2020 by Sung et al. [15].
| Cancer site | No. of new cases (% of all sites) | No. of new deaths (% of all sites) | ||
|---|---|---|---|---|
| Female breast | 2,261,419 | -11.7 | 684,996 | -7 |
| Lung | 2,206,771 | -11.4 | 1,796,144 | -18 |
| Prostate | 1,414,259 | -7.3 | 375,304 | -4 |
| Nonmelanoma of skina | 1,198,073 | -6.2 | 63,731 | -1 |
| Colon | 1,148,515 | -6 | 576,858 | -6 |
| Stomach | 1,089,103 | -5.6 | 768,793 | -8 |
| Liver | 905,677 | -4.7 | 830,180 | -8 |
| Rectum | 732,210 | -3.8 | 339,022 | -3 |
| Cervix uteri | 604,127 | -3.1 | 341,831 | -3 |
| Esophagus | 604,100 | -3.1 | 544,076 | -6 |
| Thyroid | 586,202 | -3 | 43,646 | -0 |
| Bladder | 573,278 | -3 | 212,536 | -2 |
| Non-Hodgkin Lymphoma | 544,352 | -2.8 | 259,793 | -3 |
| Pancreas | 495,773 | -2.6 | 466,003 | -5 |
| Leukemia | 474,519 | -2.5 | 311,594 | -3 |
| Kidney | 431,288 | -2.2 | 179,368 | -2 |
| Corpus uteri | 417,367 | -2.2 | 97,370 | -1 |
| Lip, oral cavity | 377,713 | -2 | 177,757 | -2 |
| Melanoma of skin | 324,635 | -1.7 | 57,043 | -1 |
| Ovary | 313,959 | -1.6 | 207,252 | -2 |
| Brain, nervous system | 308,102 | -1.6 | 251,329 | -3 |
| Larynx | 184,615 | -1 | 99,840 | -1 |
| Multiple myeloma | 176,404 | -0.9 | 117,077 | -1 |
| Nasopharynx | 133,354 | -0.7 | 80,008 | -1 |
| Gallbladder | 115,949 | -0.6 | 84,695 | -1 |
| Oropharynx | 98,412 | -0.5 | 48,143 | -1 |
| Hypopharynx | 84,254 | -0.4 | 38,599 | -0 |
| Hodgkin lymphoma | 83,087 | -0.4 | 23,376 | -0 |
| Testis | 74,458 | -0.4 | 9334 | -0 |
| Salivary glands | 53,583 | -0.3 | 22,778 | -0 |
| Anus | 50,865 | -0.3 | 19,293 | -0 |
| Vulva | 45,240 | -0.2 | 17,427 | -0 |
| Penis | 36,068 | -0.2 | 13,211 | -0 |
| Kaposi sarcoma | 34,270 | -0.2 | 15,086 | -0 |
| Mesothelioma | 30,870 | -0.2 | 26,278 | -0 |
| Vagina | 17,908 | -0.1 | 7995 | -0 |
| All sites excluding nonmelanoma skin | 18,094,716 | 9,894,402 | ||
| All sites | 19,292,789 | 9,958,133 | ||
Table 1: New cases and deaths from cancer.
Materials and Methods
This article, written in English language is a retrospective study of the material regarding brain tumors available on PubMed, and other reputable journals as well as trusted websites.
It aims to educate researchers and public about the disease by presenting an explanation and a historical review on brain cancer including its nature, symptoms, global distribution, staging. It also includes the usual methods to diagnose the treat via surgical, thermal or radiation methods and means to treat it, whether systematic or palliative techniques. The prognosis is also presented in the article.
Diagnosis of brain tumours
Diagnosis of adult brain and spinal cord tumors are performed using one or more of the following procedures.
Physical exam and family history
During physical exam the general signs of disease such as existence of lumps are investigated. The general habits such as smoking, exercise as well as family history of the patients are also registered.
Neurological exam
The brain, spinal cord, and nerve function are usually investigated through asking the patient some questions to check: a patient’s neurocognitive capabilities, memory, expressions, coordination, simple calculations, vision (central and peripheral), audition, gait as well as muscular voluntary and involuntary refluxes work.
Electrical activity tests
The electrical activities are tested using Electroencephalography (EEG) and evoked potentials.
Tumor marker test
A sample of blood, urine, or tissue is checked to measure the levels of substances that are linked to certain types of cancer.
Gene test
Cells or tissue taken from the tumor are analyzed in the laboratory for changes in genes or chromosomes, which may indicate unwanted growth. These changes may be a sign that a person has or is at risk of having a specific disease or condition.
Ultrasound
Used to test for brain tumors in prenatal and postnatal conclusions, as well as cerebral arteries.
X-ray
Use electromagnetic radiation to image the skull and the bones that comprise it to detect tumors within the brain or the nasal sinuses. Xray imaging include.
Conventional non-invasive techniques as well as cerebral arteriograms or angiograms using a contrast medium injected into the veins of the patient.
Conventional Invasive X-Ray Methods include:
Pneumoencephalography here, lumbar or suboccipital approach is utilized to ingrain air into the cerebral ventricles and the subarachnoid spaces and then trace it using sophisticated x-ray procedure
Ventriculography through a trepanation opening, air is brought into every sidelong brain ventricle following collection of CSFs.
Both pneumoencephalography and ventriculography are no longer used and are replaced by newer neuroimaging examinations which are less invasive and more advanced.
Myelography: using a dye injected in the cerebrospinal fluid and traced with x-ray (myelogram) to check if there is spread in other parts of the brain or the spinal cord.
Computed tomography (CT)
Brain tumor detection and characterization rely on standard imaging with CT and magnetic resonance imaging (MRI).
A CT scan is a computerized three-dimensional x-ray that can reveal the internal tissue and organs with high precision; for example, soft tissue, bone, and blood vessels. Any abnormalities e.g. changes of the skull bones. Leakage of fluids like blood or enlargement of brain ventricles or tumors are easily detected.
Magnetic resonance imaging (MRI)
Usually incorporated T1-weighted imaging: Spin echo, turbo spin echo gradient echo, dynamic reviews and 3D echo, or T2-weighted imaging: Dark-fluid, proton thickness and fluid-attenuated reversal recovery and other techniques
MRI can be used in other forms.
MRI spectroscopy to measure the level of metabolites in the body.
Perfusion MRI to detect blood flow.
Functional MRI (fMRI) to study the use of oxygen and blood flow during physical activity. fMRI can help plan for surgery by identifying the motor, sensory, visual and language centers of the brain [16].
CT scans are preferred for patients with pacemakers or metal implants who cannot undergo an MRI examination. It less time consuming than MRI and more effective in defining calcifications or abnormalities in the skull. MRI on the other hand, are more delicate and hence effective for soft tissue investigations. Figure 3 showsNormalized (MRI) images for different types of tumors in different planes [17].
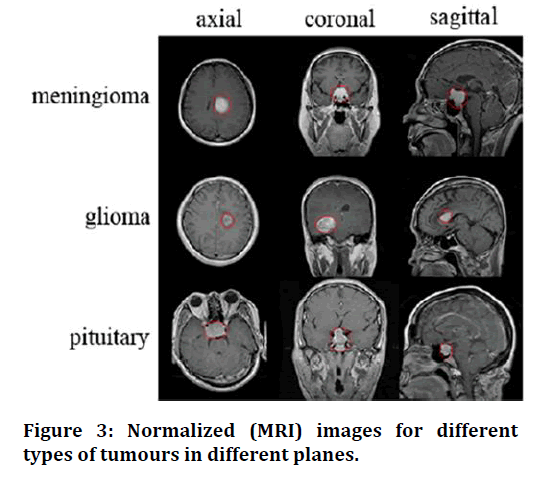
Figure 3. Normalized (MRI) images for different types of tumours in different planes.
Nuclear imaging
A non-invasive imaging technique that produces images of the distribution of radionuclide in patients, usually using a radiopharmaceutical that is specific for the diagnostic purpose, and a gamma camera. The most famous nuclear medicine techniques used foe detection of brain tumor are: Positron emission tomography (PET) and single photon emission computed tomography scan (SPECT). These techniques provide functional diagnosis of the diseases. To produce more accurate detections PET and SPECT are usually combined with CT scanning.
Positron emission tomography (PET)
Due to the high metabolic activity and thus sugar intake, a famous nuclear medicine test uses radiotracer with a glucose compound (18F-fluorodeoxyglucose, FDG) injected into the patient’s veins. This radiotracer eventually emits a positron which annihilate when it comes in contact with an electron producing produce two photons in opposite directions at a time. A special coincidence detection circuitry (dedicated gamma camera) is used to detect two photons simultaneously. Active cells absorb more of this tracer and thus more photons are produce at the tumour sites, showing more brightness. This allows detecting the position, size and activity of the tumour. PET scans also distinguish primary from metastatic tumours. Treglia et al. [16] conducted an evidence-based data met analysis for the different radiotracers used in the detection of brain tumour using PET scans including 18F-FDG, 11C-methionine, 18F-FET, 18F-FDOPA, 18F-FLT, and 11C-choline. Studies showed that the different tracers provided good diagnostic performance in detecting brain tumours, with the highest performance showed by radio-labelled amino acid tracers example methyl-[11C]-L-methionine ([11C]MET) [16]. Multiple PET scanners were successfully used to identify and classify gliomas with high precisions [18].
Single photon emission computed tomography (SPECT)
Use radiotracers that generate a single photon or gamma decay which are captured by a dedicated camera to produce a 3-D image of the organ (e.g. brain) identifying normal and abnormal tissue (e.g. tumour) The highly energetic meta-stable isomer of technetium is commonly used. is converted to the stable by emitting a gamma photon. Similar to other nuclear medicine tests including PET scans, a gamma camera then rotates about the patient and acquires projections at different angles. They are used to differentiate post-therapeutic changes from tumour recurrence or metastases or potential nonneoplastic mimics. SPECT involves attaching the technetium isomer to a protein found in body: Annexin-V to the phospholipid bilayer. This isomer cannot penetrate the outer layer of healthy cells. This way technetium can be detected in malignant cells that are undergoing apoptosis [19].
Biopsy involves removal of some tissue from the tumour site using a fine needle for morphological and pathological test. It usually follows imaging, upon detection of a tumour. Imaging guidance usually assists in determining the details of the tumour. It allows staging and aggressiveness of the tumor by visualizing its characteristics. Nevertheless, biopsy may pose high risks such as infections due to its invasive nature [4].
Biopsy can be categorized as
Open biopsy: Involves surgical removal of a sample of the brain tissue for investigation. This can be followed by removal of the tumour if it was found to be malignant. Performed when imaging tests show that there may be a tumour that can be removed by surgery.
Lumbar puncture
Involves collecting a sample of the CSF to test for signs of malignancy.
Stereotactic biopsy
Performed when images show a deep-seated tumour that is hard to reach with the normal processes. It is usually an image-guided procedure.
The tests involved include
Microscopy, cytogenic analysis and Immunohistochemistry.
Treatment methods of brain tumours
The recommendations for treatment of brain tumours rely on several factors such as:
• The type (malignant or benign; primary or secondary, local or metastatic etc.).
• Size, proximity and effect e.g. putting pressure on vital parts of the brain.
• The grade or stage of the tumour.
• The possible side effects of the treatment.
• The general health and preferences of the patient, some patients refuse some therapies for example chemotherapy.
Treatment options include
Active surveillance
This refers to closely watching the condition of patients with asymptomatic, slow growing tumours without medical interference. The patient is monitored on a regular basis to avoid worsening of conditions that may lead to aggressive treatments which in turn may result in serious side-effects [20].
Surgery
The whole tumour or part of it in addition to some surrounding tissues is removed for diagnostic and therapeutic purposes as mentioned above. This may reduce the pressure of the tumour on nearby parts of the brain and hence improve neurological symptoms. Surgery may be the only treatment needed if the tumour is low-grade and localized. The main drawback of surgery is that it may miss tumours that are too small to be seen, which may lead to recurrence of the disease.
Radiation therapy
Uses megavoltage x-rays or charged particles for example electrons to kill or sterilize the tumour cells [21,22].
The most common type of radiotherapy modalities used for brain tumours are:
External-beam radiation therapy which consists
Conventional radiation therapy: The patient undergoes imaging using a simulator for example to locate the tumor: position, size and organs at risk, then the whole brain is irradiated following a treatment plan designed by an oncology team (medical physicist, dosimeters and oncologist).
3-D conformal radiation therapy: A more precise 3-D plan is referred to as 3-dimensional conformal radiation therapy. It applies more than one thin beam from of different intensities irradiated from different angles and orientations to focus the radiation beam on the tumour and preserve the healthy near-by organs [6].
Intensity modulated radiation therapy (IMRT)
IMRT techniques uses inverse planning methods together with biological complication models for organs at risk. Beam intensities are modelled using a computeroptimized planning to obtain highly conformal dose delivery. By replacing conformal beams with smaller beams of irregular fluence using maps obtained through optimization processes complex dose distributions with high precisions are achieved [23,24].
Proton therapy
Relatively heavy positively charged protons are used due to their high relative biological effectiveness which allows destroying the tumour cells more efficiently while resulting in minimal damage to neighbouring healthy organs. This modality is useful for tumours that are at proximity to bones, example brain tumours close to the base of the skull or near the optic nerves [25].
Stereotactic radiosurgery
This is a non-invasive form of surgery that applies a single large dose of radiation aimed directly at the tumour. This prevents unnecessary irradiation of healthy tissue [26]. It is quite effective in many metastatic tumours but more effective in localized benign tumours. It is performed using different types of machines including: a modified linear accelerator, a gamma knife, a cyber or robotic knife.
Fractionated stereotactic radiation therapy: which is divided into small daily doses or fractions, given over several days or weeks, is commonly used for tumours located close to sensitive structures, such as the optic nerves or brain stem [27].
Alternating electric field therapy
A non-invasive modality that is performed using electric fields produced by electrodes at the head. It is very effective in treatment of newly diagnosed or recurrent glioblastoma. The survival rate is longer and the side effects are fewer than chemotherapy for this disease.
LASER interstitial thermal therapy (LITT)
This is a minimally invasive cytoreductive treatment that uses real-time MR-thermography guidance and applies LASER to cause hyperthermia or thermo-necrosis on the tumour [28-30].
Medicinal treatment
As mentioned in part 1symptoms of brains tumour can be severe and seriously affect the quality of lives of the patients and their families. Certain medications are used to manage the symptoms. They can also be used to provide supportive care to improve the quality of lives; for example, controlling seizures and fluid buildup or any other swelling in the brain. These medicines include Dexamethasone, a corticosteroid that helps reducing the swelling and hence the pressure and pain. Other medicines include anti-seizure medications. Medicinal treatment may also be used to destroy tumour cells; known as systemic therapy [31,32].
An example of systemic therapies is Chemotherapy. Here, one or more drugs; preferably those that can pass the blood-brain barriers (the physical and chemical barrier that actively regulates tracking blood-borne molecules, pathogens, and immune cells into the brain) easily are delivered to the patients orally or intramuscularly or intravenously to kill tumor cells or sterilize or slow down the growth of the tumour [33,34].
Targeted therapy
This treatment modality targets the tumour’s specific genes, proteins, or the tissue environment that enhance the growth and spread of brain tumour cells without harming the healthy tissue [35].
The most famous targeted therapies for brain tumours include anti-angiogenesis therapy using for example, Bevacizumab which works on starvation of the tumour [36]. Other targeted therapies as Larotrectinib work on genetic change (NTRK fusion) found in brain and other tumors. This type is specifically useful for metastatic brain tumours and other tumours that are difficult to remove surgically. Monoclonal antibody therapy on the other hand are antibodies that can identify substances that enhance tumour growth (for example the protein called vascular endothelial growth factor (VEGF) that helps creation of new blood vessels needed by the tumours to grow) , attach to them and eventually kill them or stop their growth and spreading. Bevacizumab is a targeted therapy used in the treatment of recurrent glioblastoma. Other new forms of targeted therapies such as VEGF and tyrosine kinase inhibitors are being investigated.
Immunotherapy
Immunotherapy also known as biotherapy or biologic therapy uses the body’s immune system to fight cancer by boosting or restoring the body’s natural defenses against the disease. Examples include gene and dendritic cell vaccine therapies [37].
Hybrid therapies (A combination of two or more therapies)
Sometimes more than one modality or regimen is needed to cure brain tumours or eliminate or reduce the sideeffects. For example, radiation therapy and chemotherapy are used post-surgery if visible tumours remain or are suspected. Examples include Gliadel wafers used to deliver carmustine drug are placed in the area of the removed tumour. Radiation therapy and chemotherapy such as temozolomide are used to treat patients with glioblastoma and high-grade glioma [38].
A combination of 3 drugs: lomustine, procarbazine and vincristine are also used along with radiation therapy for treating patients with grade III oligodendroglia.
Surgery with electrical simulation is used when surgery is not quite convenient due to the proximity of the tumour to a delicate part of the brain or spinal cord. Sometimes, the patient is awakened once the surface of the brain is surgically exposed, then electrical stimulation techniques are used. This allows surgeons to remove the tumour without causing damage to the delicate part, for example the speech centre [39].
Conclusion
This study equips readers with specific but in-depth information about brain tumor; nature, causes, symptoms and prevalence. It also provides knowledge about the different diagnostic and therapeutic facilities that are useful for brain tumours. It also guides researchers to different original and review articles that are found in the internet, which can save their precious time.
References
- Benson CC, Deepa V, Lajish VL, et al. Brain tumor segmentation from MR brain images using improved fuzzy c-means clustering and watershed algorithm. In 2016 International Conference on Advances in Computing, Communications and Informatics (ICACCI) 2016.
- Tortora GJ, Derrickson BH. Principles of anatomy and physiology. John Wiley & Sons; 2018.
- Gumaste PP, Bairagi VK. A hybrid method for brain tumor detection using advanced textural feature extraction. Biomedical and Pharmacology Journal. 2020 Mar 28;13(1):145-57.
- Tandel GS, Balestrieri A, Jujaray T, et al. Multiclass magnetic resonance imaging brain tumor classification using artificial intelligence paradigm. Computers Biol Med 2020; 122:103804.
- Chen R, Smith-Cohn M, Cohen AL, et al. Glioma subclassifications and their clinical significance. Neurotherapeutics 2017; 14:284-97.
- Gupta A, Dwivedi T. A simplified overview of World Health Organization classification update of central nervous system tumors 2016. J Neurosci Rural Practice 2017; 8:629.
- Louis DN, Perry A, Reifenberger G, et al. The 2016 World Health Organization classification of tumors of the central nervous system: A summary. Acta Neuropathol 2016; 131:803-20.
- https://radiopaedia.org/cases/meningioma-35?lang=gb
- https://radiopaedia.org/cases/glioblastoma-nos-butterfly-morphology?lang=gb
- https://radiopaedia.org/cases/anaplastic-astrocytoma-who-grade-iii?lang=gb
- https://radiopaedia.org/cases/oligodendroglioma-idh-mt-1p19q-codeleted?lang=gb
- Raghavapudi H, Singroul P, Kohila V. Brain tumour: Causes, symptoms, diagnosis and radiotherapy treatment. Current Med Imaging 2021.
- Ghandour F, Squassina A, Karaky R, et al. Presenting Psychiatric and neurological symptoms and signs of brain tumors before diagnosis: A systematic review. Brain Sci 2021; 11:301.
- Barnholtz-Sloan JS, Sloan AE, Davis FG, et al. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the metropolitan detroit cancer surveillance system. J Clin Oncol 2004; 22:2865-72.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin 2021; 71:209-49.
- Treglia G, Muoio B, Trevisi G, et al. Diagnostic performance and prognostic value of PET/CT with different tracers for brain tumors: a systematic review of published meta-analyses. Int J Mol Sci 2019; 20:4669.
- Badža MM, Barjaktarović MČ. Classification of brain tumors from MRI images using a convolutional neural network. Applied Sci 2020; 10:1999.
- Miyake K, Suzuki K, Ogawa T, et al. Multiple positron emission tomography tracers for use in the classification of gliomas according to the 2016 World Health Organization criteria. Neuro Oncol Adv 2021; 3:172.
- Sadaghiani MS, Sheikhbahaei S, Rowe SP, et al. Cellular and molecular imaging with SPECT and PET in brain tumors. Radiologic Clin 2021; 59:363-75.
- Lallana EC, Abrey LE. Update on the therapeutic approaches to brain tumors. Expert Review Anticancer Therapy 2003; 3:655-70.
- Brown PD, Jaeckle K, Ballman KV, et al. Effect of radiosurgery alone vs radiosurgery with whole brain radiation therapy on cognitive function in patients with 1 to 3 brain metastases: A randomized clinical trial. JAMA 2016; 316:401-9.
- Minniti G, Filippi AR, Osti MF, et al. Radiation therapy for older patients with brain tumors. Radiation Oncol 2017; 12:1-4.
- Omer HB. Intensity modulated radiotherapy using Monte Carlo for routine postmastectomy radiotherapy. Polish J Med Physics Eng 2012; 18:49-58.
- Gupta M, Kant R, Nautiyal V, et al. A dosimetric comparison between three-dimensional conformal radiation therapy and intensity-modulated radiation therapy in the treatment of posterior fossa boost in medulloblastoma. J Cancer Res Therapeutics 2017; 13:1027.
- Sato M, Gunther JR, Mahajan A, et al. Progression‐free survival of children with localized ependymoma treated with intensity‐modulated radiation therapy or proton‐beam radiation therapy. Cancer 2017; 123:2570-8.
- León-Triana O, Pérez-Beteta J, Albillo D, et al. Brain metastasis response to stereotactic radio surgery: A mathematical approach. Mathematics 2021; 9:716.
- Scaringi C, Agolli L, Minniti G. Technical advances in radiation therapy for brain tumors. Anticancer Res 2018; 38:6041-5.
- Mohammadi AM, Schroeder JL. Laser interstitial thermal therapy in treatment of brain tumors–the neuroblate system. Expert Rev Med Devices 2014; 11:109-19.
- Patel NV, Mian M, Stafford RJ, et al. Laser interstitial thermal therapy technology, physics of magnetic resonance imaging thermometry, and technical considerations for proper catheter placement during magnetic resonance imaging–guided laser interstitial thermal therapy. Neurosurgery 2016; 79:S8-16.
- Schulze PC, Vitzthum HE, Goldammer A, et al. Laser-induced thermotherapy of neoplastic lesions in the brain–underlying tissue alterations, MRI-monitoring and clinical applicability. Acta Neurochirurgica 2004; 146:803-12.
- Moravan MJ, Fecci PE, Anders CK, et al. Current multidisciplinary management of brain metastases. Cancer 2020; 126:1390-406.
- Moravan MJ, Fecci PE, Anders CK, et al. Current multidisciplinary management of brain metastases. Cancer 2020; 126:1390-406.
- Desland FA, Hormigo A. The CNS and the brain tumor microenvironment: Implications for glioblastoma immunotherapy. Int J Mol Sci 2020; 21:7358.
- https://www.surgonc.theclinics.com/article/S1055-3207(16)30026-6/abstract
- Sharma P, Debinski W. Receptor-targeted glial brain tumor therapies. International journal of molecular sciences. 2018; 19:3326.
- Kim MM, Umemura Y, Leung D. Bevacizumab and glioblastoma: past, present, and future directions. Cancer J 2018; 24:180-6.
- Pierpont TM, Limper CB, Richards KL. Past, present, and future of rituximab—the world’s first oncology monoclonal antibody therapy. Frontiers Oncol 2018; 8:163.
- Oya S, Ikawa F, Ichihara N, et al. Effect of adjuvant radiotherapy after subtotal resection for WHO grade I meningioma: A propensity score matching analysis of the brain tumor registry of Japan. J Neuro-Oncol 2021; 153:351-60.
- Sun YS. Direct-current electric field distribution in the brain for tumor treating field applications: A simulation study. Computational Mathematical Methods Med 2018; 2018.
Author Info
Buthayna G Elshaikh1*, Hiba Omer1, MEM Garelnabi2, Abdelmoneim Sulieman3, Nawader Abdella1, Sahar Algadi1 and Hind Toufig4
1Department of Basic Sciences, Deanship of Preparatory Year and Supporting Studies, Imam Abdulrahman bin Faisal University, Saudi Arabia2Sudan University of Science and Technology, College of Medical Radiological Science, Khartoum, Sudan
3Department of Radiology and Medical Imaging, College of Applied Medical Sciences, Prince Sattam Bin Abdulaziz University, Saudi Arabia
4College of Medicine, King Faisal University, Kingdom of Saudi Arabia
Citation: Buthayna G Elshaikh, Hiba Omer, MEM Garelnabi, Abdelmoneim Sulieman, Nawader Abdella, Sahar Algadi, Hind Toufig,Incidence, Diagnosis and Treatment of Brain Tumours , J Res Med Dent Sci, 2021, 9(6): 340-347
Received: 07-Jun-2021 Accepted: 23-Jun-2021
