Research - (2020) Volume 8, Issue 6
Pattern of Haematological Changes in Patients with Chronic Kidney Disease Attending Nephrology Clinic in a Tertiary Hospital in North Central Nigeria
Emmanuel Oladipo Sanni1*, Hannah Oluwayemisi Olawumi2, Idayat Adenike Durotoye2, Abiola Samuel Babatunde2, Olasunkanmi Akeem Shittu AO2, Sikiru Abayomi Biliaminu3, Khadijat Olaitan Omokanye KO4, Mutiat Kehinde Ogunfemi4, Olabisi Omolara Akinwumi OO5, Dayo Oyedepo6 and Senol Dane7
*Correspondence: Emmanuel Oladipo Sanni, Department of Haematology, Faculty of Basic Clinical Sciences, Nile University of Nigeria, Nigeria, Email:
Abstract
Introduction: The impact of the disease of the kidney is enormous in developing countries like Nigeria. With the increasing use of erythropoietin stimulating agents in anaemic patients with CKD, there is a need to evaluate the changes in haematological parameters in these patients. This study assessed the haematological changes in patients with known chronic kidney disease.
Materials and methods: Our research was a cross-sectional study that assessed and compared the haematologic parameters of 113 patients who had established chronic kidney disease and 113 -healthy controls who were age–and sex-matched. The full blood count (FBC) was determined by SysmexKX21(Sysmex, Japan). The test of significance was set at a P- value of <0.05.
Results and Discussion: The mean age of patients with CKD was 55.00 + 15.37 years, whilst that of controls was 52.73±13.59 years. (p-value 0.240) The values of the mean of the Hb and RBC among the patients were 9.69+ 2.25g/dl and 3.62 ± 0.92 x 1012/L respectively and were found to be significantly lower than that seen among controls (13.62 ± 1.45 and 4.94 ± 0.62 respectively) with a P-value<0.001. Significantly higher values of the mean WBC and platelet count were found in the patients when compared with the control participants (p-value<0.001). A significant proportion of our patients had microcytic hypochromic anaemia (32%).
Conclusion: Careful evaluation of the cause of anaemia and urgent interventions are needed in the managements of these patients.
Keywords
Anaemia, Erythropoietin stimulating agents, Chronic kidney disease, Haematological changes
Introduction
The steady rise in the incidence of chronic kidney disease (CKD) globally is a great concern to both the patients and the clinicians [1]. Diseases of the kidney account for a substantial quota of the burden resulting from diseases in developing countries [2,3]. The approximate prevalence of CKD globally is put at 8-16% but this varies across countries and regions [3]. Prevalence of CKD in Ilorin communities of Kwara State, Nigeria was found to be 15.8% [4]. The recurrent haematological abnormalities seen among patients diagnosed with CKD are anaemia and its severity increases with a decline in the function of the kidney [5].
Anaemia is usually seen at GFR less than 60mls/ min but more evident when its below 30mls/min (stage 4 and 5 of CKD) [6,7]. In Nigeria, variation in anaemia prevalence among people living with CKD is reported to be between 77.5 and 94% [8-10]. Anaemia affects almost every organ of the body due to reduced delivery of oxygen and use in the body tissues and this impact negatively on the quality of life and contributes significantly to several morbidities seen in these patients [11]. Anaemia as defined by the World Health Organization is Hb level in men less than 13.0g/dl as well as in women who are postmenopausal with a value of < 12g/dl in premenopausal women [12].
Several causes of anaemia in patients with CKD have been reported, but predominantly it is caused by erythropoietin (EPO) deficiency resulting from reduced renal synthesis of EPO [13]. Iron deficiency and hypo responsiveness to the effect of erythropoietin are the other major contributory factors to anaemia seen in patients with CKD.
Patients with CKD and anaemia often suffer from reduced quality of life, recurrent need for blood transfusion, increased hospital admissions, cognitive impairment, cardiovascular comorbidities, and higher mortality [14]. With the increasing use of erythropoietin stimulating agents and iron supplements among patients with CKD in our center, a reduction in anaemia prevalence should be expected. This study however, assessed the haematologic profile of patients with CKD and compared it with healthy controls.
Materials and Methods
Study Design
This study was a cross-sectional study done in the hospital. Ethical Approval from the University of Ilorin Teaching Hospital’s committee on ethics was obtained before the commencement of the study.
Study population
The research population consists of 113 patients with known Chronic Kidney Disease attending the nephrology clinic of the University of Ilorin Teaching Hospital (UITH) and 113 control participants between the ages of 18-75 years recruited over three months period from June to September 2017. The participants were patients with established chronic kidney disease describe as either damage to the kidney or a decline in glomerular filtration rate (GFR) of lower than 60 ml/min/1.73m2 for not less than 3 months [15]. The GFR estimation was done using Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) [16].
Inclusion criteria
For patients
Patients with established CKD as defined by KDOQ1.
Patients with CKD who are on erythropoietin stimulating agents and or on iron supplements were also included in the study.
For controls
Controls were apparently healthy age- and sex- matched hospital staff, patient’s relatives, medical and nursing students.
Exclusion criteria
Patients with suspected CKD but did not match the criteria outlined by KDOQI.
Patients with haematological disorders e.g Sickle cell anaemia and β Thalassaemia major.
and those with an active infection such as Tuberculosis, HIV, Hepatitis.
Patients whose ages are less than 18 years, pregnant women and patients diagnosed with chronic inflammatory conditions such as chronic liver disease, malignancy and the patients that did not give consent.
For Controls
Those that did not give consent.
Established cases of Sickle cell anaemia, systemic hypertension, Diabetics Mellitus, Tuberculosis and Malignancies.
Methods
At the point of enrollment into the study, a structured questionnaire was used in obtaining pertinent information (e.g., age, gender etc.) from the patients and healthy controls.
Three millilitres (mls) of venous blood was collected from each of the patients and controls and was put into EDTA bottle for analysis of Full blood count. The Full Blood Count (haemoglobin concentration, red blood cell count, the red cell indices, as well as total white cell count and differentials, platelets count and reticulocyte count) were done within 2 hours of collection of the samples using Sysmex KX-21 automated Haematology counter according to manufacturer’s instruction.
Manual reticulocyte count
The reference method approved by the international council for standardization in haematology (ICSH) was the method used for manual counting of the reticulocyte. The procedure was conducted within 6hrs of collecting the patient’s sample in potassium EDTA-anticoagulated bottle.
Method [17]
The solution used for staining was a preparation of 1.0g of new methylene blue dissolved in 100ml of 3% trisodium citrate-saline solution (30g sodium citrate in 1L saline). The solution was then filtered before use. With two (2) drops of the staining solution put into a Pasteur pipette (plastic), 2 volumes of participant’ EDTA blood was added if Hb was normal while, 4 volumes of blood was added if participant blood was anaemic and then mixed. The resultant mixture was incubated at 37°C for 15-20mins. The red cell was gently mixed and blood film was made on glassware’s. The film was examined under the microscope when dried.
Counting reticulocytes
The counting of the number of the reticulocytes and that of the red blood cells (RBCs) was done under oil immersion, x100 objective lens. The percentage of reticulocyte counted per 100 of RBCs was calculated and the absolute and corrected reticulocyte count was obtained.
Data analysis
Statistical package for social sciences version 20. (SPSS IBM Corp. Armonk, NY) was used for inputting data and analyses. The means ± SD of the numerical variables that were normally distributed was generated while those with skewed data were expressed in the median with inter- quartile range. The differences in the means was determined by student t- test. A p-value of ≤ 0.05 was set as a test of significance.
Results
A total of 113 patients with known chronic kidney disease and 113 age-matched and sex healthy controls were recruited into our research work. The mean value of the age of the patients was 55±15.37years, while the control was 52.7±14years. (P=0.742). Table 1 shoes the age range was 19-70yrs. Hypertension 93.0 (63.7%) was the commonest underlying cause for CKD, followed by diabetes mellitus 36.0 (24.7%) and Chronic glomerulonephritis 15.0 (10.3%) (Figure 1).
| Variable | Subject | Control | Total | χ2 | p value |
|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | |||
| Age | |||||
| ≤ 25 | 8 (7.1) | 7 (6.2) | 15 (6.6) | 2.727 | 0.742 |
| 26 – 35 | 10 (8.8) | 8 (7.1) | 18 (8.0) | ||
| 36 – 45 | 8 (7.1) | 13 (11.5) | 21 (9.3) | ||
| 46 – 55 | 29 (25.7) | 31 (27.4) | 60 (26.5) | ||
| 56 – 65 | 31 (27.4) | 34 (30.1) | 65 (28.8) | ||
| > 65 | 27 (23.9) | 20 (17.7) | 47 (20.8) | ||
| Mean ± SD | 55.00±15.37 | 52.73±13.59 | 1.178t | 0.24 | |
| Gender | |||||
| Male | 82 (72.6) | 84 (74.3) | 166(73.5) | 0.091 | 0.763 |
| Female | 31 (27.4) | 29 (25.7) | 60(26.5) | ||
| χ2: Chi square test | |||||
Table 1: Age and gender distribution of the study participants.
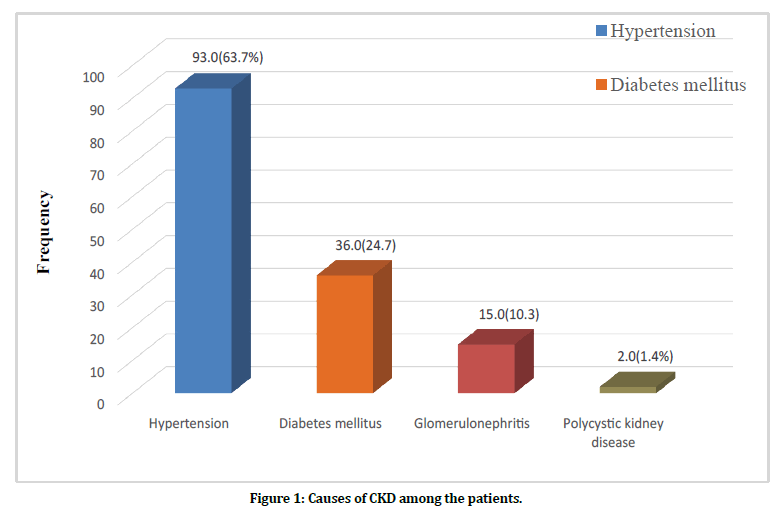
Figure 1. Causes of CKD among the patients.
A greater proportion of the patients with established CKD were in late stages of Renal disease, 47 (41.6%) stage 4 and 44 (38.9%) stage 5, both accounting for 91 (80.5%) of patients with CKD while 22.0 (19.3%) were in stage 3.
The haematological profile of the participants in our study is presented in Table 2. The mean haemoglobin concentration, total RBC count, reticulocyte count, corrected reticulocyte count and absolute reticulocyte count were reduced among the patients with CKD when contrasted with the control participants (p-value <0.001). The mean MCV, MCH, MCHC of both groups revealed no significant difference. (P values=0.148, 0.141, 0.201 respectively). However, significantly higher values of the mean WBC and platelet count were found in the patients when compared with the control participants (p-value <0.001).
| Haematological parameters | Patients (Mean ± Sd) | Control (Mean ± Sd) | T | P value |
|---|---|---|---|---|
| Hb (g/dL) | 9.69 ± 2.25 | 13.62 ± 1.45 | -15.608 | <0.001* |
| PCV (%) | 29.02 ± 6.12 | 40.33 ± 3.89 | -16.573 | <0.001* |
| RBC (×1012/L) | 3.62 ± 0.92 | 4.94 ± 0.62 | -12.627 | <0.001* |
| MCV (fl) | 80.44 ± 6.65 | 81.56 ± 4.82 | -1.452 | 0.148 |
| MCH (pg) | 27.73 ±2.24 | 28.18 ± 2.30 | -1.478 | 0.141 |
| MCHC (g/dL) | 33.26 ± 2.89 | 33.66 ± 1.59 | -1.283 | 0.201 |
| WBC (× 109/L) | 6.54 ± 2.19 | 5.66 ± 1.64 | 3.417 | 0.001* |
| PLT count (×109/L) | 263.67 ± 115.45 | 206.28 ± 70.13 | 4.516 | <0.001* |
| Reticulocyte count (%) | 0.47 ± 0.23 | 1.26 ± 0.36 | -19.649 | <0.001* |
| Corrected reticulocyte (%) | 0.32 ± 0.22 | 1.14 ± 0.35 | -20.98 | <0.001* |
| Absolute reticulocyte (/L) | 1.79 ± 1.26 | 6.16 ± 2.17 | -18.443 | <0.001* |
Table 2: Haematological profile of the patients and the control participants.
The mean haematological parameters in all 3 stages of CKD showed no verifiable difference P-value>0.05 (Table 3). Although, mean values of most of the parameters were highest in stage 3, while stage 5 had the lowest values. Anaemia in patients with CKD was more prevalent 100 (88.5%) compared with control participants 24 (21.2%), with a statistically significantly statistical difference (P-value<0.001). The prevalence of mild, moderate, and severe anaemia was more common among the male participants when compared to female participants. P=0.002 (Table 4).
| Haematological parameters | Stage 3 (Mean ± Sd) | Stage 4 (Mean ± Sd) | Stage 5 (Mean ± Sd) | F | p value |
|---|---|---|---|---|---|
| Hb (g/dL) | 10.35 ± 2.35 | 9.67 ± 2.18 | 9.69 ± 2.26 | 1.404 | 0.25 |
| PCV (%) | 31.11 ± 6.33 | 29.14 ± 5.91 | 27.85 ± 6.06 | 2.147 | 0.122 |
| RBC (×1012/L) | 3.85 ± 0.78 | 3.66 ± 1.03 | 3.47 ± 0.84 | 1.35 | 0.264 |
| MCV (fl) | 80.44 ± 4.97 | 81.22 ± 6.82 | 79.60 ± 7.22 | 0.665 | 0.516 |
| MCH (pg) | 28.00 ± 2.18 | 28.07 ± 2.48 | 27.24 ± 2.24 | 1.805 | 0.169 |
| MCHC (g/dL) | 33.59 ± 2.22 | 33.03 ± 3.33 | 33.35 ± 2.73 | 0.302 | 0.74 |
| WBC (× 109/L) | 6.30 ± 2.28 | 3.67 ± 2.61 | 6.53 ± 1.63 | 0.208 | 0.813 |
| PLT count (× 109/L) | 258.23 ± 99.68 | 264.83 ± 118.04 | 265.16 ± 112.23 | 0.03 | 0.971 |
| Reticulocyte count (%) | 0.54 ± 0.28 | 0.47 ± 0.20 | 0.43 ± 0.22 | 1.848 | 0.162 |
| Corrected reticulocyte count (%) | 0.40 ± 0.27 | 0.33 ± 0.22 | 0.27 ± 0.19 | 2.478 | 0.089 |
| Absolute reticulocyte(/L) | 2.13 ± 1.46 | 1.88 ± 1.32 | 1.54 ± 1.05 | 1.879 | 0.158 |
Table 3: Haematological parameters of patients with CKD at different stages.
| Gender | Total N (%) | χ2 | p value | ||
|---|---|---|---|---|---|
| Severity of anaemia (N=100) | Male N (%) | Female N (%) | |||
| Mild | 23 (31.9) | 1 (3.6) | 24 (24.0) | 12.574 | 0.002* |
| Moderate | 28 (38.9) | 21 (75.0) | 49 (49.0) | ||
| Severe | 21 (29.2) | 6 (21.4) | 27 (27.0) | ||
Table 4: Severity of anaemia among the patients with CKD based on gender distribution.
The proportion of patients with anaemia varied among stages of CKD with 18 (81.8%) in stage 3, 42 (89.4%) in stage 4 and 40 (90.4%) in stage 5. The most frequent morphological features among 100 anaemic patients with CKD were normocytic normochromic 67 (67%) followed with microcytic hypochromic anaemia 32 (32%). One patient with CKD had macrocytosis 1 (1%).
Discussion
The findings in our study revealed that the patients with known -CKD had low levels of Hb, RBC count as well as reduced corrected reticulocyte count and this is in keeping with the reports of similar studies done in the past [1,8]. A progressive decline in Hb concentration, PCV, Red cell count, corrected reticulocyte count was observed in patients with CKD as the disease progressed from stage 3 to stage 5 CKD. This is similar to reports of the study done by Shittu et al [8] and Iyawe et al. [18] The aetiology of reduced RBC count as well as low levels of Hb seen in patients with CKD is reduced erythropoietin production, low response to erythropoietin use, iron deficiencies, infection, uraemic inhibitors of erythropoiesis and shortened red cell survival [11].
Our research study revealed that the prevalence of anaemia observed in patients with CKD was 88.5% and this was significantly higher than the controls (21.2%). A finding like this was observed in other studies done in Nigeria [8,9,18]. Despite the increased use of erythropoietin stimulating agents and iron supplements in our center, the prevalence rate of anaemia in patients with CKD appears not to have significantly reduced. Previous studies done some years ago at our center showed a similar prevalence of anaemia [8]. Thus, the response to the use of erythropoietin in patients with CKD still needs further evaluation more especially at the late stages of the disease.
The prevalence of anaemia in CKD is higher among patients in developing countries compared to developed countries [6,19]. This may partly be due to the presence of factors that are not directly due to kidney disease which include a high burden of infections and malnutrition. Other contributory factors are the aetiology of the disease and the time of presentation of these patients to the nephrologists.
Anaemia prevalence increases as the clinical stage of CKD progresses, from 81.8% at stage 3 to 90.9% at stage 5, this is like the study done by Chinwuba et al. [20] in Enugu Nigeria. This is consistent with the fact that as the eGFR declines, there is a corresponding decline in Hb concentration, which is mostly due to the reduced synthesis of erythropoietin.
In this study, normocytic normochromic blood picture was seen in 67% of patients with CKD, 32% had microcytic hypochromic blood picture while 1% had macrocytic blood picture. These findings are like other studies done in Nigeria [8]. The total WBC of the patients was within the normal range of WBC count. This finding is consistent with other similar studies done in Nigeria [5,21].
The finding of a statistically significantly higher platelet count among the patients compared with control participants is consistent with the report of a previous study done in India by George et al. [22]. A mildly elevated levels of platelets counts were observed in a category of patients with known CKD who had treatment with erythropoietin [23]. This may partly explain the raised platelet count found in our study as a significant number of our patients with CKD in this study were on ESA.
Conclusion
The prevalence rate of anaemia among the patients with established Chronic kidney disease as observed in our study is high, and it is pronounced in later stages. This may have a role in increasing the rate of patient’s mortality and morbidity. A significant proportion of our patients had microcytic hypochromic anaemia (32%). Careful evaluation of the cause of anaemia and urgent interventions are needed in the management of these patients.
References
- Arogundade F, Barsoum R. CKD prevention in sub-saharan Africa: A call for governmental, nongovernmental and community support. Am J Kidney Dis 2008; 51:515-523.
- Xue JL, Ma JZ, Louis TA, et al. Forecast of the number of patients with end stage renal disease in the United States to the year 2010. J Am Soc Nephrol 2001; 12:2753-2758.
- Afolabi MO, Abioye-kuteyi AE, Arogundade FA, et al. Prevalence of chronic kidney disease in a Nigeria practice population. SA Fam Pract 2009; 51:132-137.
- Olanrewaju TO, Aderibigbe A, Chijioke A, et al. Prevalence of chronic kidney disease in Nigeria population-analysis of aggregate data from seven communities in Kwara State, Nigeria. 2015. Abstract presented at world congress of nephrology 2015. Cape Town International.
- Akinsola A, Durosinmi MO, Akinola NO. The haematological profile of Nigerians with chronic renal failure. Afr J Med Sci 2000; 29:13-16.
- Babitt JL, Lin HL. Mechanisms of anaemia in CKD. J Am Soc Nephrol 2012; 23:1631-1634.
- Astor BC, Muntner P, Levin A, et al. Association of kidney function with anaemia. The third national health and nutrition examination survey. Arch Intern Med 2002; 162:1401-1408.
- Shittu A, Chijioke A, Biliaminu S, et al. Haematological profile of patients with chronic kidney disease in Nigeria JNRT 2013; 5:2.
- Ulasi II, Ijoma CK, Onodugo OD, et al. Towards prevention of chronic kidney disease in Nigeria: a community-based study in Southeast Nigeria. Kidney Int Suppl 2013; 3:195-201.
- Oluboyode OA, Williams A. Serum ferritin and other indices in Nigerians with chronic renal failure-A review of management of anaemia. Afri J Med Sci 1995; 24:231-238.
- Teehan G, Benz RL. An update on the controversies in anaemia management in chronic kidney disease: Lessons learned and lost. Anaemia 2011; 2011.
- Uzma M, Robert DT. Anaemia, diabetes, and chronic kidney disease. Diabetics Care 2009; 32:1320-1326.
- Macdougall IC, Hutton RD, Cavill I, et al. Poor response to treatment of renal anaemia with erythropoietin corrected by iron given intravenously. BMJ 1989; 299:1781-788.
- KDOQ1 National kidney foundation: KDIQ1 clinical practice guidelines and clinical practice. Recommendation for anaemia in chronic kidney disease. Am J kidney Dis 2006; 47:11-45.
- Levey AS, Coresh J, Balk E, et al. National practice guidelines for chronic kidney disease: evaluation, classification, stratification. Ann intern Med 2003; 139:137-147.
- Levey AS, Stevens LA. Estimating GFR using the CKD epidemiology collaboration (CKD-EPI) creatinine equation: More accurate GFR estimates, lower CKD prevalence estimates, and better risk predictions. Am J Kidney Dis 2010; 55:622.
- Briggs C, Bain BJ. Basic haematologiacal tecniques In: Lewis SM, Bain BJ, Bates I, et al. Dacie and Lewis. 11th Edn London: Churchill Livingstone 2012; 33-36.
- Iyawe OI, Adejumo OA. Haematological profile of predialysis chronic kidney disease patients in a tertiary hospital in southern Nigeria. J Med Trop 2018; 20:36-41.
- Ijioma C, Ulasi I, Ijeomo U, et al. High prevalence of anaemia in predialysis patients in Enugu, Nigeria. Nephrol Rev 2010; 2.
- Chinwuba I, Ulasi I, Ifebunandu N. High prevalence of anaemia in predialysis patients in Enugu, Nigeria. Nephrology Reviews 2010; 2:e14.
- Abdu A, Arogundade F, Adamu B, et al. Anaemia and its response to treatment with recombinant human erythropoietin in chronic kidney disease patients. WAJM 2009; 28:295-299.
- George SV, Pullockara JK, Sailesh KS, et al. A study to assess changes in the haematological profile in chronic kidney disease. Pharm J 2015; 4:1-3.
- Sharpe P, Desai Z, Morris T. Increase in mean platelet volume in patients with chronic renal failure treated with erythropoietin. J Clin Path 1994; 47:159-161.
Author Info
Emmanuel Oladipo Sanni1*, Hannah Oluwayemisi Olawumi2, Idayat Adenike Durotoye2, Abiola Samuel Babatunde2, Olasunkanmi Akeem Shittu AO2, Sikiru Abayomi Biliaminu3, Khadijat Olaitan Omokanye KO4, Mutiat Kehinde Ogunfemi4, Olabisi Omolara Akinwumi OO5, Dayo Oyedepo6 and Senol Dane7
1Department of Haematology, Faculty of Basic Clinical Sciences, Nile University of Nigeria, Abuja, Nigeria2Department of Haematology, Faculty of Basic Medical Sciences, University of Ilorin, P.M.B 1515, Ilorin, Nigeria
3Department of Chemical Pathology and Immunology, Faculty of Basic Medical Sciences, University of Ilorin, P.M.B 1515, Ilorin, Nigeria
4Department of Haematology and Blood Transfusion, University of Ilorin Teaching Hospital, P.M.B 1459, Ilorin, Nigeria
5Department of Haematology and Blood Transfusion, Kwara state general Hospital, Ilorin, Nigeria
6Department of Nephrology, Internal Medicine, University of Ilorin Teaching Hospital, P.M.B 1459, Ilorin, Nigeria
7Department of Physiology, Faculty of Basic Medical Sciences, College of Health Sciences, Nile University of Nigeria, Abuja, Nigeria
Citation: Emmanuel Oladipo Sanni, Hannah Oluwayemisi Olawumi, Idayat Adenike Durotoye, Abiola Samuel Babatunde, Olasunkanmi Akeem Shittu AO, Sikiru Abayomi Biliaminu, Khadijat Olaitan Omokanye KO, Mutiat Kehinde Ogunfemi, Olabisi Omolara Akinwumi OO, Dayo Oyedepo, Pattern of Haematological Changes in Patients with Chronic Kidney Disease Attending Nephrology Clinic in a Tertiary Hospital in North Central Nigeria, J Res Med Dent Sci, 2020, 8 (6): 170-175.
Received: 27-Aug-2020 Accepted: 21-Sep-2020
