Case Report - (2019) Volume 7, Issue 3
Peripheral Giant Cell Granuloma: A Case Report
Prathusha Subramanian, Darimeka Kharbuli and Swarga Jyoti Das*
*Correspondence: Swarga Jyoti Das, Department of Periodontics and Oral Implantology, Regional Dental College and Hospital, India, Email:
Abstract
Peripheral giant cell granuloma appears as a solitary purplish-red nodule, having vascular or hemorrhagic appearance, with or without surface ulceration. It may be a sessile or pedunculated lesion on the gingiva or the alveolar crest, common in relation to incisors and first molars. It occurs in response to local irritation from the connective tissue of the gingiva, periodontal ligament or periosteum of the alveolar ridge and may occur in periimplant tissues and edentulous arches. They are more common in females than males, more frequently seen in mandible than maxilla. Their growth potential is very high, and may penetrate interdentally to involve the adjacent cortical bone. An accurate diagnosis based on the histological observation is vital for its management. Considering its rapid and penetrating nature of its growth, surgical excision including its base is recommended to prevent the recurrence.
Keywords
Solitary, Gingival overgrowth, Periimplant tissues, Sessile, Excision
Introduction
Localized gingival overgrowth is a common finding in clinical practice and may often present a diagnostic challenge due to similar clinical presentation. Various types of localized gingival overgrowth are identified, amongst which, the most prevalent one is peripheral fibroma (56 - 61%), followed by pyogenic granuloma (19%-27%), peripheral ossifying fibroma (10%-18%) and peripheral giant cell granuloma (1.5%-7%) [1,2]. These occur as reactive response of the gingiva to chronic, lowgrade irritation caused by plaque and calculus, periodontal disease, poor dental restorations, ill-fitting dental appliances or extractions [3].
Clinically, peripheral giant cell granuloma appears as a solitary purplish-red nodule, having vascular or hemorrhagic appearance, with or without surface ulceration. It may be a sessile or pedunculated lesion on the gingiva or the alveolar crest, common in relation to incisors and first molars. It occurs in response to local irritation from the connective tissue of the gingiva, periodontal ligament or periosteum of the alveolar ridge. It may even be reported in periimplant tissues and edentulous arches [3,4]. They occur commonly in females, at fourth to sixth decades of life, more frequently in mandible [5]. Their growth potential is very high, and may penetrate interdentally to involve the adjacent cortical bone resulting in separation of the adjacent teeth [6]. Radiographically it appears as ‘cuffing’ [3].
The diagnosis is confirmed histologically based on the presence of multinucleated giant cells embedded in a highly fibrillar connective tissue stroma containing large number of ovoid or spindle shaped young fibroblasts and spicules of bone or osteoid. Complete surgical excision following removal of the local irritants is the preferred treatment modality in order to reduce the recurrence (10%-15%) [3].
Case Report
A 38 year old male reported with a localised painless purplish-red, sessile, bi-lobulated gingival overgrowth in relation to 14 and 15, extending from the interdental papilla on the buccal aspect to the palatal aspect, the size being 11 mm × 15 mm (Figure 1A and Figure 1B). The growth started as a small swelling approximately 6 months back. It was resilient in consistency, with no blanching on digital pressure. Surface ulceration was seen on the palatal aspect and revealed blood on aspiration. 14 were found to be non-vital on electric pulp testing and Grade II mobile. The growth was asymptomatic, though tended to bleed during brushing or mastication, if accidentally bitten on. No history of trauma to that area was reported. Medical history was non-contributory. Extra-oral examination revealed no facial asymmetry or lymphadenopathy.
Intra-oral periapical radiograph revealed widening of the PDL space in relation to 14 and 15 with erosion of interdental bone and external root resorption on the distal aspect of 14 (Figure 1C). Routine blood investigations were within normal range. Based on the history and clinical and radiographic findings, provisional diagnosis was made as Peripheral giant cell granuloma. Pyogenic granuloma and peripheral fibroma were considered for differential diagnosis due to the similarity of their clinical presentation.
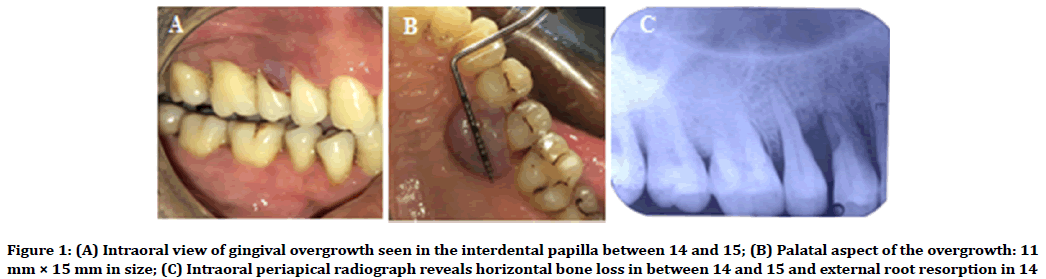
Figure 1. (A) Intraoral view of gingival overgrowth seen in the interdental papilla between 14 and 15; (B) Palatal aspect of the overgrowth: 11 mm × 15 mm in size; (C) Intraoral periapical radiograph reveals horizontal bone loss in between 14 and 15 and external root resorption in 14
Phase I periodontal therapy followed by extraction of 14 and surgical excision of the growth was planned. Written consent was obtained from the patient. Accordingly, scaling and root planing was performed. 14 were extracted and growth was excised along with the surrounding normal tissues using Kirkland ’ s gingivectomy knife, and interdental bone was curetted under local anaesthesia (Figure 2A and Figure 2B). Hemostatic gel-sponge was placed into the socket and flap margins were approximated with interrupted sutures. Periodontal dressing (Coe-pack) was placed and the patient was recalled after 7 days for suture removal. The excised tissue was sent for histopathological examination. The patient was followed up regularly upto 4 months post-surgically and no evidence of recurrence was observed (Figure 2C).
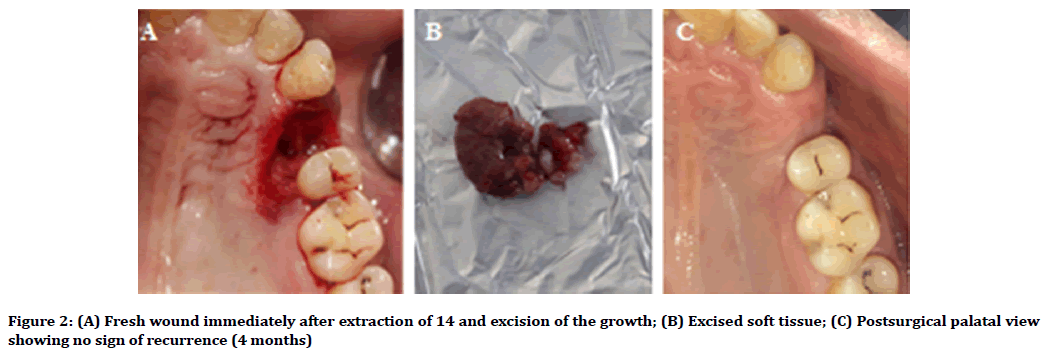
Figure 2. (A) Fresh wound immediately after extraction of 14 and excision of the growth; (B) Excised soft tissue; (C) Postsurgical palatal view showing no sign of recurrence (4 months)
Histopathological examination of the tissue section revealed ulcerated, hyperplastic stratified squamous epithelium. Underlying connective tissue stroma was moderately collagenous, with numerous young fibroblasts and scattered giant cells of varying sizes along with diffuse chronic inflammatory cells (Figure 3).
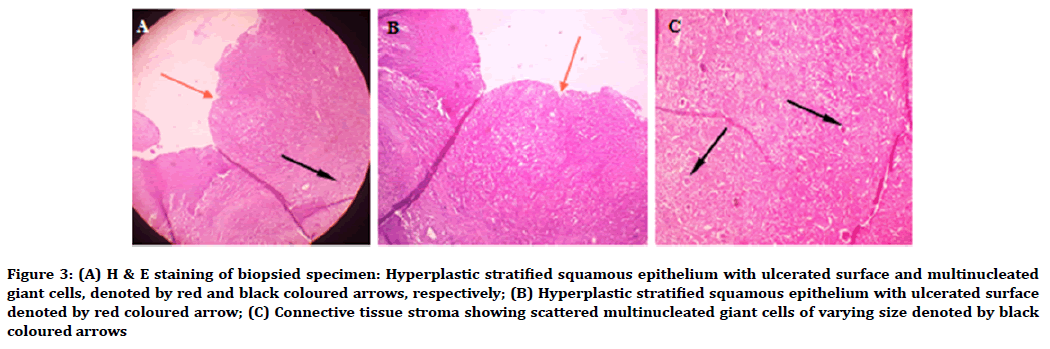
Figure 3. (A) H & E staining of biopsied specimen: Hyperplastic stratified squamous epithelium with ulcerated surface and multinucleated giant cells, denoted by red and black coloured arrows, respectively; (B) Hyperplastic stratified squamous epithelium with ulcerated surface denoted by red coloured arrow; (C) Connective tissue stroma showing scattered multinucleated giant cells of varying size denoted by black coloured arrows
Discussion
Peripheral giant cell granuloma is a benign exophytic lesion, initially thought to be of reparative nature, and was named Giant cell reparative granuloma by Jaffe [7]. In contrast, Waldron et al. described that histologically this lesion showed features of benign giant cell tumors of bone with no reparative characteristics [8]. Bhaskar et al. subdivided giant cell granuloma into central and peripheral. Central giant cell granuloma occurs within the bone, while peripheral giant cell granuloma originates in gingiva or edentulous alveolar processes [9].
The central variant may occur as slowly growing asymptomatic swelling or an aggressive lesion that manifests with pain, local destruction of bone, root resorption or displacement of teeth and usually associated with Cherubism, Noonan Syndrome, Jaffe- Campanacci Syndrome, Neurofibromatosis Type 1, or Ramon Syndrome. Radiographically, it may appear as a large, unilocular or multilocular radiolucency with welldefined or ill-defined margins and varying degrees of expansion of cortical plates [10,11].
Peripheral giant cell granuloma also known as peripheral giant cell tumor, osteoclastoma, reparative giant cell granuloma, giant cell epulis and giant cell hyperplasia [10]. As the periodontium responds to similar irritants in a different way, it is postulated that peripheral giant cell granuloma occurs as a more exaggerated response of periosteum to these factors than that associated with the formation of pyogenic granuloma, which is a more common lesion.
In addition, peripheral giant cell granuloma has found to be associated with xerostomia, which may be attributed to the reduced cleansing action of saliva that facilitates accumulation of local irritants [6].
Peripheral giant cell granuloma is also reported as an oral manifestation of hyperparathyroidism, X-linked hypophosphatemic rickets, though very rare [12-14]. Here, external root resorption is seen, which is commonly seen in central giant cell granuloma.
Surgical excision along with its base is the treatment of choice. Extraction of the adjacent teeth may be necessary to ensure complete resection in some cases [15].
Improper eradication of underlying source of irritation [2] or lack of inclusion of the periosteum or periodontal ligament during excision may lead to recurrence [16] which demands further re-excision [3,17].
Conclusion
An accurate diagnosis of gingival overgrowth through clinical, radiographic and histopathological examination is vital for its management. Due to the rapid growth pattern and tendency to resorb bone with resultant tooth movement, the treatment should include the surgical excision of the growth including its base in addition to elimination of etiologic factors. Post-surgical regular follow-up is essential to prevent recurrence of the growth.
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this manuscript.
References
- Zhang W, Chen Y, An Z, et al. Reactive gingival lesions: A retrospective study of 2,439 cases. Quinessence Int 2007; 38:103-10.
- Kfir Y, Buchner A, Hansen LS. Reactive lesions of the gingiva: A clinicopathological study of 741 cases. J Periodontol 1980; 51:655-61.
- Shafer WG, Hine MK, Levy BM. A textbook of oral pathology. 6th ed. Philadelphia: WB Saunders Co 1983.
- Hernandez G, Lopez-Pintor RM, Torres J, et al. Clinical outcomes of peri-implant peripheral giant cell granuloma: A report of three cases. J Periodontol 2009; 80:1184-91.
- Katsikeris N, Kakarantza-Angelopoulou E, Angelopoulos AP. Peripheral giant cell granuloma. Clinicopathologic study of 224 new cases and review of 956 reported cases. Int J Oral Maxillofac Surg 1988; 17:94-9.
- Bodner L, Peist M, Gatot A, et al. Growth potential of peripheral giant cell granuloma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1997; 83:548-51.
- Jaffe HL. Giant-cell reparative granuloma, traumatic bone cyst, and fibrous (fibro-oseous) dysplasia of the jawbones. Oral Surg Oral Med Oral Pathol 1953; 6:159-75.
- Waldron CA, Shafer WG. The central giant cell reparative granuloma of the jaws. An analysis of 38 cases. Am J Clin Pathol 1966; 45:437-47.
- Bhaskar SN, Bernier JL, Godby F. Aneurysmal bone cyst and other giant cell lesions of the jaws: Report of 104 cases. J Oral Surg Anesth Hosp Dent Serv 1959; 17:30-41.
- Chuong R, Kaban LB, Kozakewich H, et al. Central giant cell lesions of the jaws: A clinicopathologic study. J Oral Maxillofac Surg 1986; 44:708-13.
- Valentine JC, Nelson BL. Central giant cell lesion. Head and Neck Pathol 2011; 5:385-88.
- Chaparro-Avendanño AV, Berini-Aytés L, Gay-Escoda C. Peripheral giant cell granuloma. A report of five cases and review of the literature. Med Oral Patol Oral Cir Bucal 2005; 10:48-52.
- Flaitz CM. Peripheral giant cell granuloma: A potentially aggressive lesion in children. Pediatr Dent 2000; 22:232-3.
- Parbatani R, Tinsley GF, Danford MH. Primary hyperparathyroidism presenting as a giant-cell epulis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998; 85:282-4.
- Patil KP, Kalele KP, Kanakdande VD. Peripheral giant cell granuloma: A comprehensive review of an ambiguous lesion. J Int Clin Dent Res Organ 2014; 6:118-25.
- Regezi JA, Sciubba J, Jordan RC. Oral pathology: Clinical pathologic correlations. Elsevier Health Sciences 2016.
- Neville BW, Damm DD, Allen CM, et al. Oral and maxillofacial pathology. Elsevier Health Sciences 2015.
Author Info
Prathusha Subramanian, Darimeka Kharbuli and Swarga Jyoti Das*
Department of Periodontics and Oral Implantology, Regional Dental College and Hospital, Bhangagarh, Guwahati, Assam, IndiaCitation: Prathusha Subramanian, Darimeka Kharbuli, Swarga Jyoti Das, Peripheral Giant Cell Granuloma: A Case Report, J Res Med Dent Sci, 2019, 7(3): 169-172.
Received: 06-May-2019 Accepted: 30-Jun-2019
