Case Report - (2019) Volume 7, Issue 5
Pyoderma Gangrenosum with Extra-Cutaneous Manifestation and Elevated beta-2 Microglobulin in a Nigerian Child
Lawan Musa Tahir1*, Lokman Cam1, Ramatu Muhammed Nafiu2, Mesut Guler3, Goksel Bahadir4, Mehmet Sina Yilmaz5 and Senol Dane6
*Correspondence: Lawan Musa Tahir, Department of Pediatrics, Nizamiye Hospital, Abuja, Nigeria, Email:
Abstract
Pyoderma gangrenosum (PG) is an uncommon ulcerative cutaneous disease with unknown etiology. It is rare in children. It presents as a painful papular or papulo-pustular lesions which rapidly evolves into ulcers. Rarely, PG may present with extracutaneous manifestations. More than 50% of patients have underlying systemic diseases. Diagnosis is often delayed especially due to the absence of pathognomonic features. There are few reports of PG from Nigeria of which only one was child. We present a 10-year-old Nigerian boy who had PG with extra-cutaneous manifestation and elevated beta-2 microglobulin levels. Therefore, the elevated beta-2 microglobulin levels could be used as measures of diagnosis and improvement criteria of PG.
Keywords
Pyoderma gangrenosum, Ulcerative cutaneous disease, Extra-cutaneous manifestation, Beta-2 microglobulin
Introduction
Pyoderma gangrenosum is an uncommon ulcerative cutaneous disease with unknown etiology. It occurs mainly in young to middleaged adults but can also occur at all ages [1,2]. It is very rare in children and only about 4% of all patients are infants and children [3,4]. More than 50% of patients with PG have an underlying systemic disease like inflammatory bowel disease, rheumatoid arthritis, hematological disorder or malignancy [5]. Rarely, visceral involvements have been reported in PG, systems involved included pulmonary, cardiac, nervous, gastrointestinal, hepatic, splenic and renal systems [5].
Although the etiopathogenesis is not well understood, the involvement of immunological factors and neutrophil dysfunction may have important roles. The frequent association with autoimmune diseases, pathergy phenomenon, defective cell-mediated immune response, monoclonal or polyclonal hyperglobulinemia and involvement of immunological factors have been reported in patients with PG [1,2]. Evidence of abnormal neutrophil trafficking and aberrant integrin oscillations have been reported in the analysis of neutrophils of PG patients [1,3].
Typically, lesions of PG start as tender papules, papulo-pustules or vesicles, evolving into painful and rapidly enlarging ulcers [1,2]. Melina et al. reported that only two cases of PG pulmonary involvements which summed up the number of reported cases to 31 [6]. Pulmonary manifestations can be asymptomatic or may present with continuous fever, cough, dyspnea and hemoptysis [6]. The chest radiograph or computerized tomography (CT) shows nonspecific features. There are non-pathognomonic histological features, but typical findings are those of prominent dermal neutrophilic infiltrates with abscess formation without signs of infection and usually without significant vasculopathy [1-3]. A nonspecific perivascular mononuclear (lymphocytic) infiltration may be seen on the biopsy of the erythematous border [3].
The diagnosis is made by excluding other causes of similar-appearing cutaneous ulcerations, including infection, malignancy, vasculitis, collagen vascular diseases, diabetes, and trauma. In a process termed pathergy, new ulcerations may occur after trauma or injury to the skin in 30% of patients with pyoderma gangrenosum. Involvement of other organ systems manifests as sterile neutrophilic infiltrates. Culture-negative pulmonary infiltrates are the most common extracutaneous manifestation.
Treatment of PG involves immunosuppressive therapy, dressing of the ulcers with antiinflammatory creams and ointments. Prednisolone, azathioprine or cyclosporine are the main agents used in the treatment of PG [6,7]. The response to the treatment of the underlying disease has also a favorable impact on the prognosis of PG treatment [4].
Case Report
The patient was a 10-year-old boy who was referred to our hospital on the 28th of April 2019, with a diagnosis of necrotizing fasciitis with metastatic abscesses and overwhelming sepsis. He was presented to the referring hospital on the first April 2019, with severe pain on the anterolateral distal third of his left leg, following a mild trauma to the site, while playing at home. He was given syrup ibuprofen, but 2 days later a small painful nodule appeared. Then, within 24 hours, multiple vesicles and pustules appeared which developed ulcers within another 24 hours. There was a high-grade fever. He had incision and drainage and was commenced on intravenous Ceftriaxone, Augmentin, and Metronidazole. Ulcers were dressed daily with dermazine gauze. His condition continued to deteriorate, as more lesions appeared, and fever persisted with a temperature range of between 38°C to 39.9°C, and leukocytosis worsened from 42 × 103 to 72 × 103. Neutrophils were predominant (more than 75%), blood cultures on 3 occasions were negative and ulcer swab cultures were also negative on 3 occasion. He had debridement of the ulcers (Figure 1) and antibiotics were changed to Clindamycin, Ciprofloxacin, and Crystalline penicillin. He was transfused with 2 pints of fresh whole blood which raised the hematocrit from 18% to 31%. The ulcers were now irrigated with normal saline, covered with vaseline gauze and bandage soaked in Eusol. While some healing has started on the left leg, new lesions continued to erupt on both forearms and right arm (Figure 2), and fever persisted. He was commenced on Meropenem 36 hours before referral. He also had other medications including Omeprazole, Ranitidine, Gestid (antiacid suspension) and vitamin B complex orally.
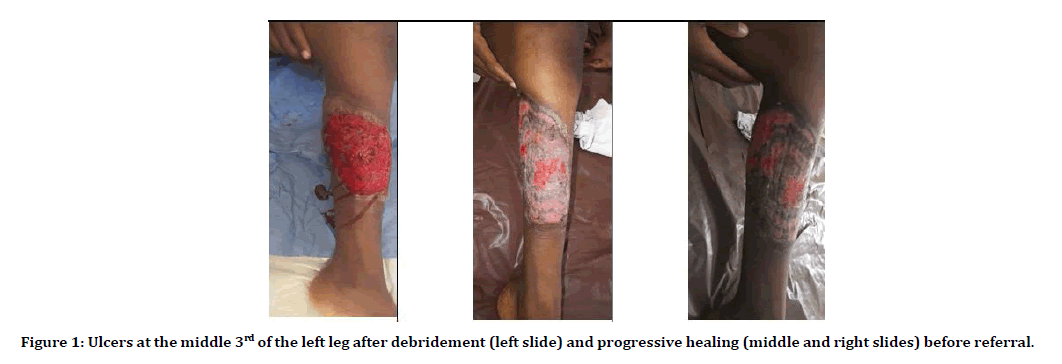
Figure 1: Ulcers at the middle 3rd of the left leg after debridement (left slide) and progressive healing (middle and right slides) before referral.
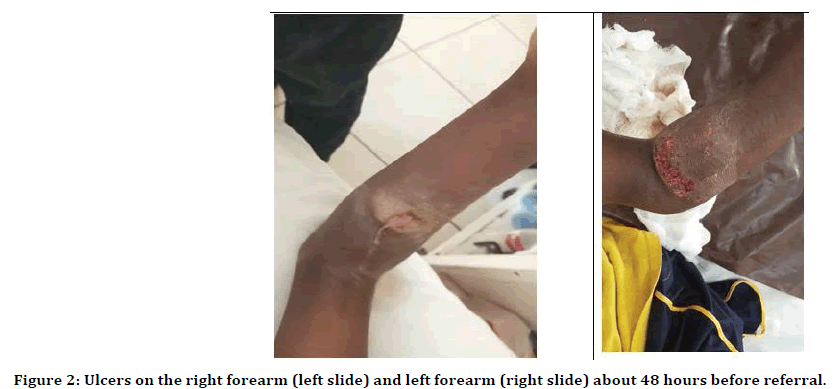
Figure 2:Ulcers on the right forearm (left slide) and left forearm (right slide) about 48 hours before referral.
On presentation at our hospital, he was found to be an ill-looking boy, in pains, mildly dehydrated and jaundiced. He was afebrile (temp 36.7°C). His vital signs were as follows: Pulse rate-112 beats per minute, respiratory rate-25 cycles per minute. He has also had bandage dressing over both arms and left leg. Further examination revealed extensive ulceration of the left mid-tibia region anteromedially, multiple tender ulcers of various shapes and sizes on the left arm and forearm; with an undermined edge. There were also multiple papulo-pustular lesions on both arms (Figure 3). He also had hepatosplenomegaly and a grade 3 pansystolic murmur.
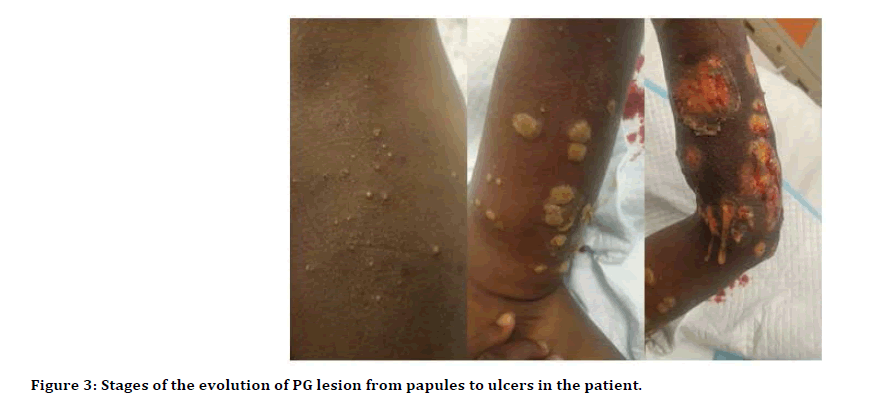
Figure 3:Stages of the evolution of PG lesion from papules to ulcers in the patient.
A preliminary diagnosis of septicemia with extensive cellulitis to rule out PG was made. The patient was continued to have Meropenem, Vancomycin intravenously, ulcers were dressed and some investigations were ordered (Table 1). A central line was inserted; the corrections in fluid and electrolyte balances were commenced.
| Date | Test requested | Result | Comment |
| 28-04-2019 | Full Blood Count (FBC) | WBC=44.76 × 103/µL | Leucocytosis with neutrophilia |
| Neut=95.8%, Lymph=1.3%, Mon=2.0%, Eos=0.8% | |||
| PLT=311 × 103/µL | |||
| Htc=28.8%, Hb=10.4 g/dL, | |||
| RBC=3.34 × 106/µL | |||
| 28-04-2019 | Viral screening tests | HIV, HBsAg, HCV were non-reactive | - |
| 28-04-2019 | Clotting profile | PT=21.4 (6.5-13.1) seconds | |
| APTT=41.3 (26-41) seconds | |||
| INR=1.93 (0.6-1.2) | |||
| 28-04-2019 | C-Reactive Protein (CRP) | CRP=31.31 (0.0 – 0.5) mg/dL | Elevated |
| 28-04-2019 | Liver Function Test (LFT) | AST=25.9 (0 - 40) U/L | - |
| ALT=26.8 ( 0 – 41) U/L | |||
| ALP=468 ( 40 – 129) U/L | |||
| Albumin=2.45 (3.5 – 5.2) g/dl | |||
| Total bilirubin=17.8 (0 - 1) g/dl | |||
| 28-04-2019 | Electrolytes, urea and creatinine | Na+=130 (136-145) mmol/l | |
| K+=4.36 (3.5-5.1) mmol/l | |||
| Cl-=94.1(98-107 ) mmol/l | |||
| Mg2+ = 2.21(1.6 – 2.6) mmol/ | |||
| 28-04-2019 | Blood and wound cultures | No growth | |
| 28-04-2019 | Immunological studies | IgA=254 (53-204) mg/dl | |
| IgE=1088 (˂ 150) kUI/L | |||
| IgG=1739 (698-1560) mg/dl | |||
| IgM=138 (31-179) mg/dl | |||
| 10-05-2019 | Antinuclear Antibody (ANA) | Negative | |
| 20-05-2019 | - | Protein C=107 | |
| Protein S=103 | |||
| 01-05-2019 | Rheumatoid factor | Negative | |
| 21-05-2019 | Beta-2 microglobulin | 7776 (0.004 - 4) mg/L repeat test on 20/06/19 was 1964 mg/L |
Table 1: Initial investigations.
Within the first week of admission, he continued to have intermittent high-grade fever; new lesions appeared especially on the left upper limb. The boy also complained of cough and chest pain. Results of blood cultures were negative but one of the wound cultures yielded Staphylococcus aureus sensitive to only fusidic acid. After dressing with fusidic acid, ulcers commenced the improvement. Fungal and mycobacterial wound cultures were negative. The chest radiograph (Figure 4) showed an area of consolidation in the left basal segments consistent with pneumonia and a chest CT also showed an area of consolidation and para-pneumonic effusion in the left basal segments consistent with pneumonia. Abdominal CT showed non-specific hepatosplenomegaly with mild free peritoneal fluid. He had slightly elevated IgA and IgG, severely elevated IgE and normal level of IgM. Pathergy test was positive. The occult blood in the stool test was negative. The hematocrit had dropped and needed a debridement (Figure 5) which necessitated another blood transfusion. The diagnosis of pyoderma gangrenosum was strengthened at this stage and corticosteroid therapy, prednisolone 2 mg/kg/day, was started.
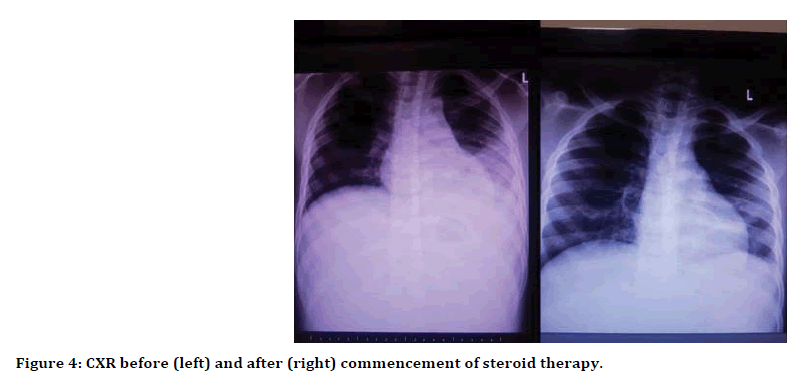
Figure 4:Left arm and forearm during debridement (first 2 pictures) and progressive healing stages (last 2 pictures).
In the second week, a remarkable improvement was noticed. No new lesion was erupting and the ulcers began to heal. Leucocytosis began to improve and CRP levels were decreasing, apatite improved and fever resolved. By the 3rd week, most of the ulcers have healed with the cribriform appearance and the patient was actively ambulating and playing. Antibiotics were stopped towards the end of the 2nd week (10 days) and stepping down of steroid (25% of the total dose weekly) commenced by the beginning of 3rd week. The central line was removed on the 15th day of insertion. Beta-2 microglobulin was found to be extremely elevated, leukemia was rule out, the antinuclear antibody was negative and the chest radiograph was now normal. The patient was discharged on the 22nd day of admission and was followed up. By the 3rd week of follow-up, all ulcers have almost healed (Figure 5) and most laboratory parameters (Table 2) had normalized and steroid was discontinued by the end of the 5th week.
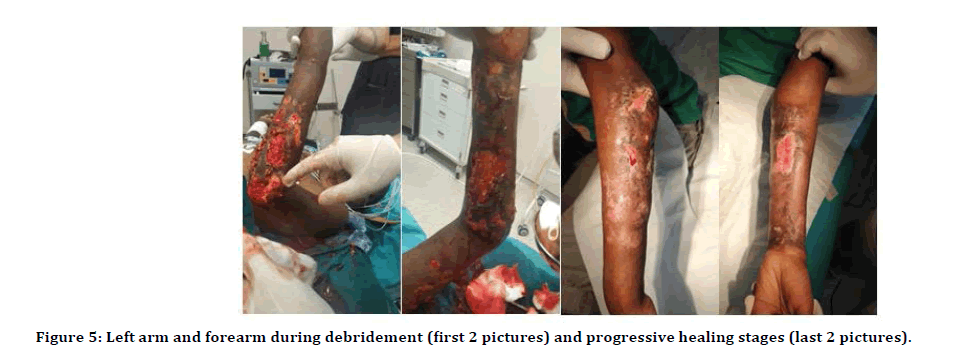
Figure 5:Cribriform scars in all the limbs, left arm (first and middle-upper pictures) right forearm (right upper picture) and left leg (lower picture).
| Investigation | Date and results | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 28-04-2019 | 30-04-2019 | 02-05-2019 | 06-05-2019 | 08-05-2019 | 11-05-2019 | 14-05-2019 | 18-05-2019 | 27-05-2019 | |
| WBC (× 103/µL) | 44.76 | 57.72 | 57.58 | 72.43 | 41.23 | 12.56 | 9.18 | 10.88 | 9.13 |
| NEUT (in %) | 95.8 | 96.9 | 96.1 | 97 | 94.5 | 85 | 84.2 | 69.4 | 73.6 |
| PLT (× 103/µL) | 311 | 316 | 217 | 399 | 485 | 540 | 712 | 405 | 324 |
| HCT (%) | 28.8 | 23.8 | 17.1 | 18.9 | 24.4 | 33.8 | 37 | 38.6 | 12.1 |
| CRP (mg/dl) | 31.306 | 19.047 | 17.096 | 25.17 | 12.459 | 2.637 | 0.381 | 9.393 | 0.132 |
| AST (U/L) | 25.9 | - | - | - | - | 12.7 | 21.8 | 27.1 | 20.8 |
| ALT (U/L) | 26.8 | - | - | - | - | 33.3 | 50.2 | 69.7 | 43.2 |
| Bilirubin (mg/dL) | 17.8 | 16.9 | 20.4 | 12.8 | 5.1 | 4 | - | 3.1 | 2.1 |
| Albumin (g/dL) | 2.45 | - | 2.29 | 3.3 | - | 3.83 | - | 4.65 | 5.1 |
| Investigation | Dates and results | ||||||||
| 30-04-2019 | 11-05-2019 | 14-05-2019 | 18-05-2019 | 20-05-2019 | 27-05-2019 | 20-06-2019 | |||
| ESR (mm/hr) | 75 | 29 | 67 | 22 | - | 7 | - | ||
| Beta-2 microglobulin (mg/L) | - | - | - | - | 7776 | - | 1968.0 (0.004 - 4) | ||
Table 2: Summary of some investigations.
Discussion
PG is a rare inflammatory neutrophilic dermatosis that occurs predominantly in patients 25 to 54 years of age [1-4]. PG presents with painful, sterile ulcers. PG may have many different etiologies. Drug-induced PG is a rare condition; propylthiouracil, granulocyte colony-stimulating factor, and sunitinib are drugs that have been implicated to date. A recent article presented a case of PG associated with sunitinib [8]. In addition to, a case of pyoderma gangrenosum induced by insulin was reported [9]. Also, PG may be a rare postoperative complication of enterostomy [10].
There were 3 reports of PG from Nigeria consisting of 7 patients out of which only one was a child (12 years old) [11-13]. Our case was a 10-year-old Nigerian boy. He presented with a typical history of a tender papule that rapidly progressed to an ulcer. The preceding mild trauma suggests pathergy which was strengthened by the positive pathergy test. The pattern of distribution of the lesions in adults is said to be more on the lower extremities; whereas in children, the lesions are also seen on the head and face, buttocks, and perianal and genital regions [14]. In our patient, there was only one lesion on the buttock and the others were on the left leg and both arms. Our case had the typical cribriform scars in all the sites of the healed lesions (Figure 6).
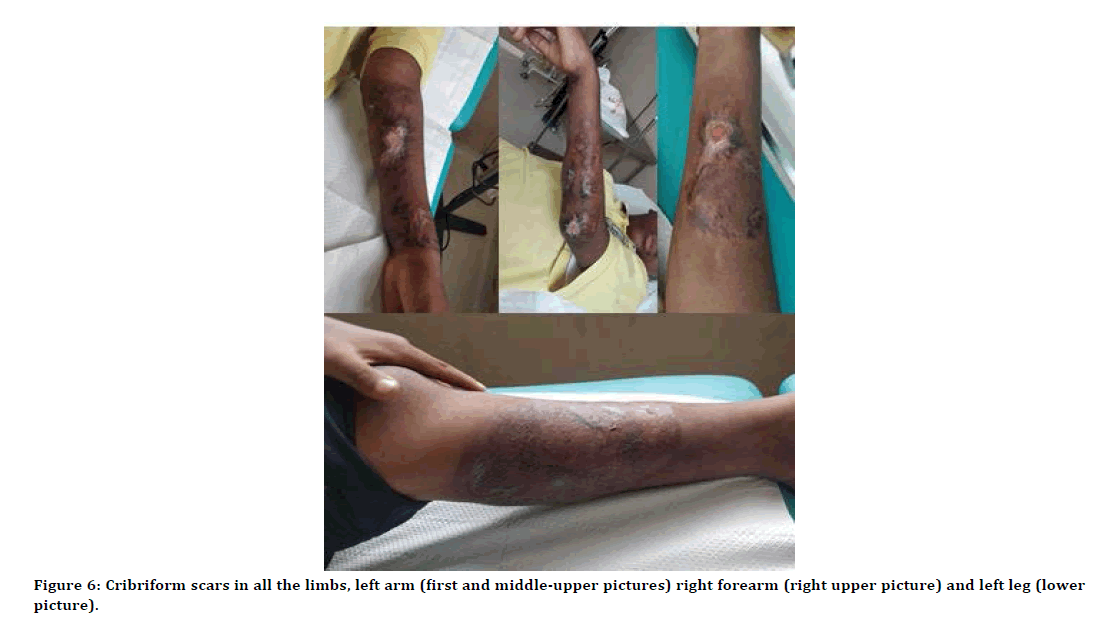
Figure 6:Cribriform scars in all the limbs, left arm (first and middle-upper pictures) right forearm (right upper picture) and left leg (lower picture).
Our patient had extra-cutaneous, pulmonary and hepatosplenic manifestations. The cough, chest pain and radiologic findings occurred while he was still on antibiotic therapy and they resolved only after commencing steroid therapy. This may suggest that the respiratory features were part of the PG processes. Pulmonary involvement in PG has been reported simultaneously with cutaneous lesions or from a few weeks up to several years after the diagnosis of cutaneous PG [6]. The deranged liver function tests and hepatosplenomegaly resolved gradually after commencing the steroid therapy. Although there are no specific laboratory findings that confirm PG, we noticed common findings reported in PG patients, such as elevated IgA and IgM; leukocytosis with neutrophilia, elevated CRP and elevated ESR. The histological findings were edematous dermis with extravasated red cells, fibrin, lymphocytes, neutrophils, and eosinophils. No vasculitis or giant cells were seen (Figure 7) [7].
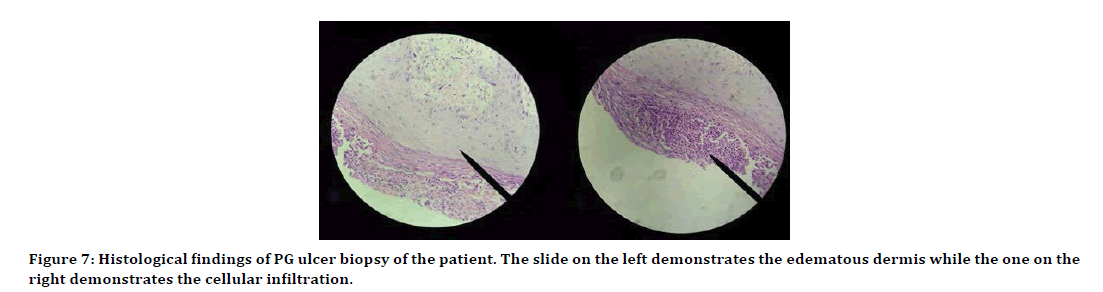
Figure 7:Histological findings of PG ulcer biopsy of the patient. The slide on the left demonstrates the edematous dermis while the one on the right demonstrates the cellular infiltration.
We have attempted to find a possibility associated underlying disease. He had no significant gastrointestinal symptoms. Occult blood in the stool test was negative and another stool analysis was essentially normal and abdominal ultrasound, as well as CT abdomen, reported non-specific hepatosplenomegaly only, which gradually resolved. There was no history suggestive of arthritis in the patient; rheumatoid factor and antinuclear antibody were negative. A very high level of beta-2 microglobulin was observed, but a hematologic review ruled out malignancy. The beta-2 microglobulin significantly reduced to lower levels after 4 weeks and was thought to be part of the immune response in PG. Therefore, the elevated beta-2 microglobulin levels can be used as a measure of diagnosis and improvement criteria of PG.
Prednisolone has been the mainstay of treatment and typically, a rapid response is observed within days to weeks [6]. Other immune-modulating agents such as cyclosporine and dapsone are also used as monotherapy or in combination with systemic steroids. We used prednisolone and noticed a significant response with the first week of commencement of therapy.
Conclusion
We presented a case with PG, a rare disease, with pulmonary and hepatosplenic manifestations. He was a 10-year-old Nigerian boy. He had high levels of beta-2 microglobulin before treatment and it recovered after treatment. It can be used as a measure of diagnosis and improvement criteria of PG.
References
- Powell FC, Hackett BC. Pyoderma Gangrenosum. In: Wolff K, Goldsmith LA, Katz SL, Gilchrist BA, Paller AS, Leffell DJ, Fitzpatrick's Dermatology in General Medicine. 7th Edn. New York: McGraw Hill 2007; 1:296–302.
- Wollina U. Pyoderma gangrenosum-A review. Orphanet J Rare Dis 2007; 2:19.
- Bhat RM, Nandakishore B, Sequeira FF, et al. Pyoderma gangrenosum: An Indian perspective. Clin Exp Dermatol 2011; 36:242-247.
- Powell FC, Su WP, Perry HO. Pyoderma gangrenosum: Classification and management. J Am Acad Dermatol 1996; 34:395-409.
- Bennett ML, Jackson JM, Jorizzo JL, et al. Pyoderma gangrenosum. A comparison of typical and atypical forms with an emphasis on time to remission. Case review of 86 patients from 2 institutions. Medicine (Baltimore) 2000; 79:37-46.
- Melina G, Frej S, Anne KA, et al. Pulmonary manifestations of pyoderma gangrenosum: 2 cases and a review of the literature. Respir Med 2015; 109:443-450.
- Dabade TS, Davis MD. Diagnosis and treatment of the neutrophilic dermatoses (pyoderma gangrenosum, Sweet’ssyndrome). Dermatol Ther 2011; 24:273-284.
- Sevimli Dikicier B. Pyoderma gangrenosum associated with sunitinib: A case report. Adv Skin Wound Care 2019; 32:427-429.
- Noronha M, Arora S, Pai K, et al. A case of pyoderma gangrenosum induced by insulin. Int Wound J 2019; in press.
- Yu YM, Lai FJ, Feng C, et al. Pyoderma gangrenosum around an ileostoma: A case report. Medicine (Baltimore) 2018; 97:13415.
- Obasi OE. Pyoderma gangrenosum and malignant pyoderma in Nigeria. Clin Exp Dermatol 1991; 16:34-37.
- Ahamefule NC, Olumide YM, Akinkugbe AO, et al. Pyoderma gangrenosum: A case report. Nigerian Med Practiti 2006; 49:85-87.
- Alese OB, Irabor DO. Pyoderma gangrenosum and ulcerative colitis in the tropics. Rev Soc Bras Med Trop 2008; 41:664-667.
- Graham JA, Hansen KK, Rabinowitz LG, et al. Pyoderma gangrenosum in infants and children. Pediatr Dermatol 1994; 11:10-17.
Author Info
Lawan Musa Tahir1*, Lokman Cam1, Ramatu Muhammed Nafiu2, Mesut Guler3, Goksel Bahadir4, Mehmet Sina Yilmaz5 and Senol Dane6
1Department of Pediatrics, Nizamiye Hospital, Abuja, Nigeria2Department of Pediatrics, State Service Hospital, Abuja, Nigeria
3Departments of Orthopedia, Nizamiye Hospital, Abuja, Nigeria
4Departments of Anaesthesia, Nizamiye Hospital, Abuja, Nigeria
5Departments of Radiology, Nizamiye Hospital, Abuja, Nigeria
6Department of Physiology, College of Health Sciences, Nile University of Nigeria, Abuja, Nigeria
Citation: Lawan Musa Tahir, Lokman Cam, Ramatu Muhammed Nafiu, Mesut Guler, Goksel Bahadir, Mehmet Sina Yilmaz, Senol Dane, Pyoderma Gangrenosum with Extra-cutaneous Manifestation and Elevated Beta-2 Microglobulin in a Nigerian Child, J Res Med Dent Sci, 2019, 7(5):67-73.
Received: 19-Jul-2019 Accepted: 24-Sep-2019
