Research - (2021) Volume 9, Issue 6
Study on Glycaemic Variability in Gestational Diabetes Mellitus (GDM) and its Maternal and Fetal Outcome
Abinaya Vijayan, V Kalavathy and K Vani*
*Correspondence: K Vani, Department of Obstetrics & Gynaecology, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, India, Email:
Abstract
A prospective, randomized study was designed to assess the glycaemic variability m Gestational Diabetes Mellitus patients. The Present study focuses on the most common maternal complication such as gestational hypertension and preeclampsia. Macrosomia, Birth trauma and jaundice were most prominent complications among neonates.
Keywords
Gestational hypertension, Preeclampsia, GDM, Complication, Trauma, Neonates
Introduction
Diabetes is a disease characterized by impairments both in the secretion and action of the insulin hormone. Glucose is the main energy substrate for intrauterine growth and is transmitted in a steady stream from mother to foetus. Glucose is produced as a result of maternal metabolism principally from carbohydrate in the diet and from the gluconeogenic amino acids.
Insulin resistance tends to increase in physiological pregnancy because of the gradually rising levels of fetoplacental hormones such as progesterone, cortisol, growth hormone, prolactin, and human placentation. In response to this imbalance, the pancreas normally compensates with a higher insulin secretion. When this compensatory mechanism fails, glucose intolerance develops and, in pregnancy, this is called gestational diabetes mellitus (GDM).
Gestational diabetes mellitus 1s a condition of pregnant woman where glucose intolerance is found during pregnancy, complicates 2 to 5% of pregnancies and is associated with both neonatal and obstetric complications. The relationship between the onset of GDM and complications in pregnancy and increased perinatal morbidity and mortality has been well documented. It has recently been suggested that glucose variability in GDM, plays a major role in both neonatal morbidity and obstetric complications.
Glycemic variability (GV) means swings, 1n blood glucose level around an average. Diminished or absent glycemic auto regulation or short falls of insulin availability are hypothesized to be the etiological factors for this glycemic variability [1]. Glycemic variability is a complex phenomenon that includes both intraday and interday variability. The intraday component corresponds to the within-day vertical glycemic fluctuations. The interday component is defined as day-to-day glucose variations, i.e., glycemic variability along a time-dependent horizontal axis. As a consequence , glycemic variability in patients with GDM is a composite of the vertical and horizontal components. If GDM is not properly treated, there is an increased risk of adverse maternal (preeclampsia, pregnancy induced hypertension, recurrent vulva-vaginal infections, increased incidence of operative deliveries, obstructed labour and development of diabetes mellitus later in life), fetal (macrosomia, polyhydromnios, preterm labour, respiratory distress, unexplained intrauterine fetal death, traumatic delivery) and neonatal complications (hypoglycaemia, jaundice, polycythaemia, tetany, hypocalcaemia, hypomagnesaemia). Glucose variability is still a poorly understood factor in GDM patients, especially its link with maternal and fetal complications. It is with this union; the objective of this prospective study was to assess the glycemic variability in GDM patients and its association with maternal and fetal outcome in India especially at Chennai. Diabetes is characterized by the development of specific microvascular complications and a high incidence of accelerated atherosclerosis. Although a large number of studies have investigated and compared the roles of the many factors involved in diabetic vascular complications, an accurate assessment of their respective contributions 1s still difficult . However, as demonstrated by many trials, microvascular and macrovascular complications are mainly or partly dependent on deglycation, which has two components: chronic sustained hyperglycaemia and acute glycemic fluctuations from peaks to nadirs. Both components lead to diabetes complications through two main mechanisms: Cessive protein glycation and activation of oxidative stress. A few years ago, these two mechanisms were unified in an elegant theory that suggested that the glycemic disorders observed in diabetic patients result in an activation of oxidative stress with an overproduction of superoxide by the mitochondrial electron- transfer chain. This activation in turn produces a cascade of such deleterious metabolic events as enhanced polyol activity, increased formation of advanced glycation end products, activation of protein kinase C and nuclear factor B, and increased hexosamine pathway flux. It is now well established that hyperglycemia both at fasting and during postprandial periods results in exaggerated and accelerated glycation. For instance, all the studies conducted in type 1 and type 2 diabetes have clearly shown a strong positive relationship between Al C levels and plasma glucose levels at fasting and over postprandial periods with the strongest correlation being observed between A1C and mean plasma glucose levels.The latter relationship was considered sufficiently demonstrative to serve as a reference in the recent standard lf medical care in diabetes that are published every year by the American Diabetes Association.
Glycemic variability (GV) means swings in blood glucose level. Diminished or absent glycemic auto regulation or short falls of insulin availability are hypothesized to be the etiological factors for these glycemic bumps. Intermittent high blood glucose exposure rather than constant high blood glucose exposure has been shown to have deleterious effect in experimental studies. In present era of targeting optimum glycemic control, it is also important to focus on GV as an additional goal point along with the traditionally followed parameters. Variations in HbA1c were proposed to contribute to development of microvascular complications like retinopathy and nephropathy. In the event of new therapeutics in the management of type 2 diabetes mellitus by glucagon like peptide-I (GLP-1) analogs and dihydropeptidyl peptidase-IV (DPP-IV) inhibitors through incretin mimetic effect, studying GV in an individual to achieve glycemic control is promising.
Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study was to clarify the risk of adverse outcome associated with degrees of glucose intolerance during pregnancies that are less severe than overt diabetes. Glucose tolerance was measured by a 75-g 2-h oral glucose tolerance test (OGTT) m a large, heterogeneous , multinational, ethnically divers e cohort of women at 24-32 (mean 28) weeks gestation with medical caregivers blinded to status of glucose tolerance (except when predefined thresholds were met). Associations between maternal glycemia and increased size at birth, delivery by caesarean section, development of neonatal hypoglycaemia, and the presence of fetal hyperinsulinemia were the predefined primary outcomes of the study. Results of the study showing continuous relationships of maternal glucose 1evels below those diagnostic of diabetes with each of the primary outcomes have been reported. Associations of maternal glucose and birth weight 90th percentile and fetal hyperinsulinemia .Weaker associations were found with caesarean delivery and clinical neonatal hypoglycaemia .Pedersen postulated that maternal hyperglycemia was transmitted to the foetus, which, in turn, produced and released large amounts of insulin, with the resultant fetal hyperinsulinemia being the cause of various aspects of diabetic fetopathy, including deposition of large amounts of body fat, which gave the infant its characteristic appearance. Pedersen documented increased body weight in infants of diabetic mothers compared with control subjects. Fetal hyperinsulinemia, in the absence of maternal diabetes, has been demonstrated to cause "diabetes-like" fetopathy in rhesus monkey offspring. At least some of the increased fetal weight has been shown to be attributable to increased fat accretion. Sparks reported that body fat more specifically represents effects of the in utero environment, whereas lean body mass represents more of the genetic component of growth. Glucose is the mam energy substrate for intrauterine growth and is transmitted in a steady stream from mother to foetus [2-15].
Materials and Methods
This prospective study was carried out in Sree Balaji Medical College & Hospital, Chrompet, Chennai Department of Obstetrics & Gynaecology.
This study was spread over a period of 18 months from December 2013 - May 2015.The study population consisted of 100 GDM patients.
Inclusion criteria
Diagnosis of GDM.
Exclusion criteria
• Diabetic before pregnancy.
• Pre gestational BMI > 35kg/m2.
• HbAlC > 8%.
• Patient having asthma epilepsy.
• Known hypertension heart problems those who received blood, plasma or immunoglobulin within the last 3 months of participation those diagnosed with malignancy or mmunodeficiency diseases patients with severe medical or psychological co morbidity.
Case definition and data collection
World health organization (WHO) guidelines were adopted for selection of GDM cases defined as any glucose intolerance with onset or first recognition during pregnancy. A proforma containing following information was obtained from each patient who included name, age, date of birth, residence, occupation, family history, menstrual history, presenting illness, date of GDM diagnosis, obstetric score, examination on each trimester, and other complications. An informedconsent was obtained from each patient and as per ICMR guidelines on biomedical research, human ethical clearance was obtained from Sree Balaji Medical College and Hospital, Chennai, India (Human Ethical No: 00 2/SBMC/IHE C/ 2013-175).
Patient recruitment and blood sample collection
A total of 123 GDM patients were recruited over a period of 18 months from Sree Balaji Medical College and Hospital, Chennai, India who were diagnosed as a case of GDM. Among 123 GDM case, 100 (81.3%) patients involved in this study and following 23 (18.6%) patients were excluded because, refused to participate (n=9), not interested (n=4), social issues (n=3), work commitments (n=2), previous stillbirth (n=l), new to area (n= l ), problems with transport (n=l ) and discontinuation in between (n=2) Based on the selection criteria, one hundred gestational diabetic women were registered for the study. Blood samples were collected in each trimester to till delivery to monitor continues glucose level. Blood was collected for diagnostic analysis as part of GDM investigations; this value was used for this study.
Statistical analysis
Values are reported as the median with range when normally distributed. The differences in the results between two groups were analysed by means of student T-test, non parametric Mann-Whitney U test. Difference between more than two groups was analysed by means of One-way ANOVA, non-parametric Krusl-1-Wall is test. A p<0.05 was statistically significant. For all the above analyses, a GraphPad Prism 6, version 6.01 was used.
Results
After collection of data from 100 pregnant women with GDM, analysis was done on the following.
Age was measured as a continuous variable in this study and for the purpose of analysis; it was categorized into 25 years and above 25 years (Table 1 and Figure 1). Minimum, median, and maximum age was 18, 27 and 36 years, respectively. Half of the subjects were above 25 years (51%). The percentages for nullipara (no child), multipara (1-5 children) and grand multipara (>5 children) women were 25%, 72% and 3% respectively (Table 2 and Figure 2). 89% were educated up to primary level and, 69%, 23% and 8% belonged to low, middle, and upper socioeconomic status respectively (Table 3 and Figure 3). In the present study, 31% patients had family history of diabetes mellitus (Table 4 and Figure 4).
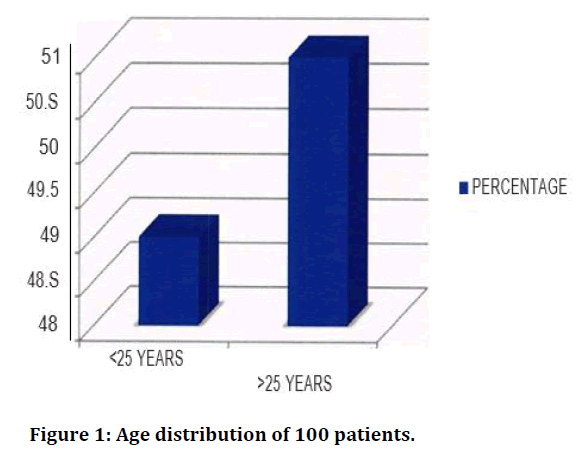
Figure 1. Age distribution of 100 patients.
| Age | <25 Years | >25 Years |
|---|---|---|
| Percentage | 49 | 51 |
Table 1: Age distribution of 100 patients.
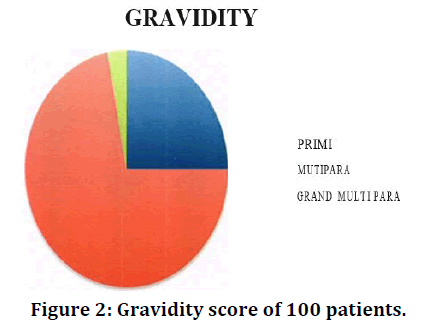
Figure 2. Gravidity score of 100 patients.
| Parity | Primi | Multi para | Grand Multi para |
|---|---|---|---|
| Percentage | 25 | 72 | 3 |
Table 2: Gravidity score of 100 patients.
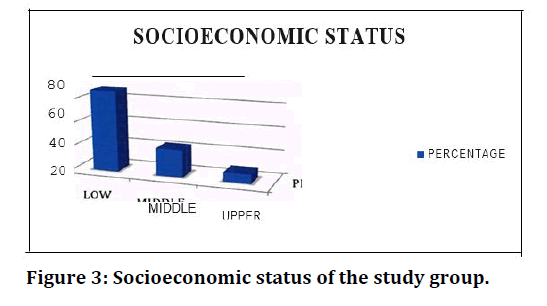
Figure 3. Socioeconomic status of the study group.
| Socioeconomic status | Percentage |
|---|---|
| Low | 69 |
| Middle | 23 |
| Upper | 8 |
Table 3: Socioeconomic status of the study group.
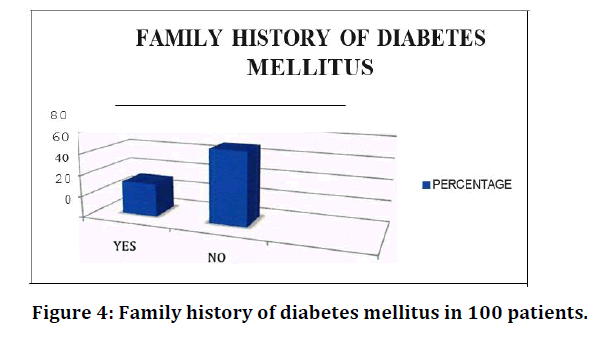
Figure 4. Family history of diabetes mellitus in 100 patients.
| Family history of diabetes mellitus | Percentage |
|---|---|
| Yes | 31 |
| No | 69 |
Table 4: Family history of diabetes mellitus in 100 patients.
Management of GDM patients
GDM of 86% was controlled by insulin and on the other hand remaining 14% were managed normoglycemia by diabetic diets only (Table 5 and Figure 5). It was observed that despite good glycaemic control by diabetic diets, 9% patients had no complication, while the remaining (5%) did and multiple complications were observed in 16% patients who had good glycaemic control by insulin.
Figure 5. GDM of 86% was controlled by insulin and 14% were managed by diabetic diets.
| Management | Insulin | Medical nutrition therapy |
|---|---|---|
| Percentage | 86 | 14 |
| Complication | 16% | 5% |
Table 5: Management of GDM patients.
Discussion
Gestational diabetes mellitus (GDM) initially defined as glucose tolerance presenting in pregnancy but remitting thereafter, is currently defined as any glucose intolerance with onset or first recognition during pregnancy. Gestational diabetes mellitus (GDM) was initially defined by O'Sullivan and Mahan m 1964 as "transient abnormalities of glucose tolerance that develop during pregnancy" and resolve post-partum. This newer definition removes the need for knowledge of normal pre pregnancy glucose tolerance, as well as the need to define reversion to normal glucose tole- nee post-partum. The timing and tests used for screening and diagnosis of GDM in pregnancy remain subject to controversy. The initial screening test proposed by O'Sullivan was conducted in the second trimester and involved a non-fasting 50g glucose "challenge" followed by measurement of the plasma glucose one hour later. A positive result was a one-hour glucose >7.8 mmol/ L. This test was typically offered at 24-28 weeks' gestation and, if equivocal, was followed by a confirmatory standardized three-hour 100g OGTT to establish the diagnosis. This "two-step approach" is still endorsed by the American College of Obstetricians and Gynaecologists (ACOG). These new guidelines recommend first trimester screening for highrisk women. The diagnosis of pre-gestational diabetes is made either by finding glycated hemoglobin level 6.5% (47.5 mmol/mol) and/or fasting blood glucose (FBG)? 7 mmol/L, or a positive 75 -g, two-hour oral glucose tolerance test (OGTT) using standard (non-pregnancy) thresholds (i.e., fasting glucose 7mmo/L or 2hr glucose? 11mmol/L). The associations, American College of Obstetricians and Gynecologists (ACOG) advocates a screening glucose challenge for all women in the second trimester, whereas the American Diabetes Association (ADA) and the International Association of Diabetes and Pregnancy Study Group (IADPSG) recommend first trimester screening for women at high risk of diabetes, followed by universal glucose tolerance testing at 24-28 weeks. Clinical evidence has confirmed that the main goal of glycemic management in GDM is to keep the mother's blood glucose levels as close as possible to the normal range. It is with this notion, our objective of this prospective study was to assess the glycemic variability in GDM patients based on ACOG, ADA and it Is association with maternal and fetal outcome m India especially at Chennai.
The studies have documented that, increasing maternal age was associated with higher frequency of GDM, showing that carbohydrate tolerance deteriorates progressively with age especially in females.
Increasing parity as an associated risk factor for GDM was well demonstrated m this study, where 72% of the patients were multiparous and this correlates well with other studies in which 80% and 76 % of patients with GDM were multiparous.
Presence of illiteracy anqverty adversely affect the outcomes; in the present study, 69% patients had no formal education and belonged to lower socioeconomic class. Minor abnormalities in carbohydrate metabolism during pregnancy can adversely affect pregnancy outcomes. Glucose intolerance increases as pregnancy advances. This trend was also demonstrated by our study, where all the patients were diagnosed as cases of GDM in the late first trimesters, results which can be compared with those of a study conducted at the civil hospital, Karachi, Pakistan.
Management of gestational diabetes is one of the most rewarding clinical experiences. Current management advocates outpatient care. An effective treatment regimen consists of dietary therapy, self-blood glucose monitoring and the administration of insulin if the target blood glucose values are not met with the diet alone. Approximately 15% of women with GDM require insulin therapy. Another study carried out at Jinnah hospital; diabetes require insulin. In this study, 86% patients were on insulin for glycemic normalization. Such a high number in the present study was due to illiteracy and lack of awareness. Despite good glycemic control by insulin 16% patients had multiple complications.
These results support our hypothesis that increasing glucose concentration less severe than diabetes 1s associated with fetal overgrowth, specifically adiposity. Data presented here show a strong and continuous association between neonatal fat content and maternal glycemia and with fetal insulin levels as measured by cord C-peptide concentrations. These relationships were present for each maternal glucose measurement and cord C-peptide. Relationships persisted even when potential confounding variables such as field centre, BMI, height, MAP, gestational age , smoking status, and alcohol use were taken into account. This pattern is like that reported for maternal glucose and birth weight 90th percentile and was also seen for the association with fat free mass, a parameter derived by subtracting fat mass from total body weight.
Significant interactions for 1-h plasma glucose and age m relation to sum of skin folds 90th percentile and triceps skin fold 90th percentile, which indicated stronger associations with increasing age, may be a chance finding due to the large number of interactions examined. The findings reported here, however, are not proof of causality. Fetal insulin, stimulated by maternal glucose transport from mother to fetus across the placenta, may act on a variety of nutrients m addition to glucose, resulting m fetal overgrowth and adiposity [16-24].
Conclusion
Measurement of glycemic variability in GDM will remain challenging to prevent maternal and fetal complications. The scientific community is just attained the requisite level of experience to resolve the methodological issues, identify common pitfalls, and optimize methods for data collection, analysis, and display in GDM and its outcome. Both maternal and neonatal complications of GDM can be reduced by dietary and lifestyle advice during pregnancy. There is increased independent association between caesarean section rates, prematurity, and macrosomic infants born to mothers. Most of the children were healthy, but there are still increased congenital anomalies. Internationally agreed criteria are highly desirable to define standards and goals, and to enable meaningful comparisons of data from different sources. Gestational diabetes mellitus (GDM) is a condition of pregnant woman where glucose intolerance is found during pregnancy, and it is associated with increased risk of serious perinatal morbidities and mortalities, as well maternal morbidities. With this notion our study was aimed to calculate the glycemic variability and to compare the maternal and fetal outcome of gestational diabetes mellitus. Selection of gestational diabetes mellitus cases WHO guidelines were adopted. An informed consent, proforma and human ethical clearance for sample collection was obtained. Our results indicate strong, continuous increased associations of maternal glucose levels leads to maternal and fetal complications. Strict Glycemic control is required during pregnancy to minimize complications. Glycemic Variability–Future target for optimum glycemic control.
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgments
The encouragement and support from Bharath Institute of Higher Education and Research, Chennai is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- Abell DA, Beischer NA, Papas AJ , et al. The association between abnormal glucose tolerance (hyperglycemia and hypoglycemia) and estriol excretion in pregnancy. Am J Obstet Gynecol 1976; 124:388-92.
- Aberg A, Rydhstroem H, Frid A. Impaired glucose tolerance associated with adverse pregnancy outcome: A population-based study in southern Swed en . Am J Obstet Gynecol 2001; 184 :77-83.
- ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Obstet Gynecol 2001; 98:525-38.
- Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001; 414:813
- Buckingham B, Block J, Wilson DM. Continuous glucose monitoring. Curr Opin Endocrinol Diabetes 2005; 12:273-279.
- Caruso A, Paradisi G, Ferrazzani S, et al. Effect of maternal carbohydrate metabolism on fetal growth. Obstet Gynecol 1998; 92:8.
- Catalano PM, Vargo KM, Bernstein IM, et al. Incidence and risk factors associated with abnormal postpartum glucose tolerance in women with gestational diabetes. Am J Obstet Gynecol 1991; 165:914-19.
- Khan A, Jaffarey SN. Screening for gestational diabetes. Medical Channel 1997; 3:8-12.
- Perveen N, Saeed M. Gestational diabetes and pregnancy outcome: Experience at Shaikh Zayed Hospital. Mother Child 1996; 34:83-8.
- Jovanovic-Peterson L, Peterson CM , Reed GF , et al. Maternal postprandial glucose le vels and infant birth weight: The diabetes in early pregnancy study. The national institute of child health and human development-diabetes in early pregnancy study. Am J Obstet Gynecol 1991; 164:103-11.
- Gillmer MDG, Hurley PA. Diabetes and endocrine disorders in pregnancy. In: Edmonds DK. Dewhurst's textbook of obstetrics and gynaecology for postgraduates. 6th Edn. Oxford: Blackwell Science 1999; 197-209.
- Cavalot F, Petrelli A, Traversa M, et al. Postprandial blood glucose is a stronger predictor of cardiovascular events than fasting blood glucose in type 2 diabetes mellitus, particularly in women: lessons from the San Luigi Gonzaga Diabetes Stud y. J Clin Endocrinol Metab 2006; 91:813- 9.
- Ceriello A, Colagiuri S, Gerich J, et al. Guideline for management of postmeal glucose. Nutr Metab Cardiovasc Dis 2008; 18:S17-33.
- Ceriello A, Quagliaro L, Catone B, et al. Role of hyperglycemia in nitrotyrosine postprandial generation. Diabetes Care 2002; 25:1439-43.
- Hanefeld M, Leiter L, Monnier L, et al. Postprandial glucoseregulation and diabetic complications. Arch Int Med 2004; 164:90-5.
- Farooq MU, Ayaz A, Ali Bahoo L, et al. Maternal and neonatal outcomes in gestational diabetes mellitus. Int J Endocrinol Metab 2007; 3:109-115.
- Cavalot F, Pagliarino A, Valle M, et al. Postprandial blood glucose predicts cardiovascular events and all-cause mortality in type 2 diabetes in a 14-year follow-up: Lessons from the san luigi gonzaga diabetes study. Diabetes Care 2011; 34:2237-2243.
- Catalano P. Management of obesity in pregnancy. Obstet Gynecol 200 7; 109:419- 433.
- Catalano PM, Ehrenberg HM . The shortand long-term implications of maternal obesity on the mother and her offspring. BJOG 2006; 113:1126-1133.
- Barbieri M, RizzoMR , Marfella R, et al. Decreased carotid atherosclerotic process by control of daily acute glucose fluctuations in diabetic patients treated by DPP-IV in hibitors . Atherosclerosis 2013; 17.
- Boden G. Fuel metabolism in pregnancy and in gestational diabetes mellitus. Obstet Gynecol Clin North Am 1996; 23:1-10.
- Boney CM, Verma A, Tucker R, et al. Metabolic syndrome in childhood: association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 2005; 115:e290-e296.
- Randhawa MS, Moin S, Shoaib F. Diabetes mellitus during pregnancy: A study of fifty cases. Pakistan J Med Sci 2003; 19.
- Perveen N, Saeed M. Gestational diabetes and pregnancy outcome: Experience at Shaikh Zayed Hospital. Mother Child 1996; 34:83-88.
Author Info
Abinaya Vijayan, V Kalavathy and K Vani*
Department of Obstetrics & Gynaecology, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Abinaya Vijayan, V Kalavathy, K Vani, Study on Glycaemic Variability in Gestational Diabetes Mellitus (GDM) and its Maternal and Fetal Outcome, J Res Med Dent Sci, 2021, 9(6): 212-218
Received: 08-May-2021 Accepted: 18-Jun-2021
