Research Article - (2021) Volume 9, Issue 12
The Effect of Combined Application of Tricalcium Phosphate Containing Varnish and ER, CR: YSGG Laser on Resistance of Enamel to Caries: An In Vitro Study
Hussein Sami Abdullah1* and Aisha Akram Qasim2
*Correspondence: Hussein Sami Abdullah, Researcher (Master student) College of Dentistry, University of Mosul, Mosul, Iraq, Email:
Abstract
Background: This study aims to investigate the efficacy of functionalized tri-calcium phosphate containing varnish combined with ER, CR: YSGG laser in preventing of enamel caries. Materials and Methods: Freshly extracted 50 sound upper first premolars teeth were divided into five groups, each group of ten samples. Group (C): only washed with deionized water. Group (V): functionalized tricalcium phosphate (fTCP) exclusively from Clinpro White Varnish. Group (L2): Er,Cr:YSGG laser (0.75 W, 8.5 J/cm2, 20 Hz, 11% air, 0% water) was irradiated. Group (V/L2): First Clinpro White varnish was applied then Er,Cr:YSGG laser. Group (L2/V): Er,Cr:YSGG laser was irradiated followed by Clinpro White varnish. Then, the pH cycling model was used to induce artificial caries. The effect was studied in vitro using Vickers microhardness test and the obtained results were statistically analyzed. The surface morphology was also examined by scanning electron microscopy. Results: there was a decreasing in surface microhardness in all groups due to the demineralization, but the least reduction in surface microhardness belonged to the group of Er,Cr:YSGG laser followed by Clinpro White varnish. Morphological changes of enamel surfaces were in relation with increasing of enamel resistance to demineralization. Conclusions: Er:Cr:YSGG Laser at energy density of 8.5 J/cm2 is indicated on decreasing the loss of enamel hardness with comparable effects to fTCP varnish application. There is an advantage in using laser at 8.5 J in combined with fTCP varnish over using laser alone and fTCP varnish alone with more benefit when laser was applied before application of fTCP varnish than the opposite.
Keywords
Er,Cr:YSGG Laser, Enamel demineralization, Functionalized tri-calcium phosphate, Microhardness
Introduction
Dental caries is an infectious illness; carbohydrates fermentation by acidogenic bacteria in the mouth is the basic process for caries formation, which results in the production of organic acid. The resultant acids pass into the hard tissues throughout dental plaque biofilm and dissociate into hydrogen ions. The mineral content of the tooth, calcium and phosphate in particular, can be dissolved by the action of these hydrogen ions [1]. This process known as demineralization. Preventing the progression of demineralization is possible, but when adequate treatment is not provided, progressive destruction of the teeth will occur.
Numerous preventive strategies are required for the prevention of dental caries. The efficiency of the fluoride application, whether as a topical or systemic agent, has been reported in many studies [2]. Fluoride varnishes have showed significant clinical effectiveness in dental caries reduction in addition to its easy application and high patient acceptability. On the other hand, to prevent demineralization, fluoride alone is not sufficient. To preserve the structural integrity of hydroxyapatite crystals, the existence of bioavailable calcium and phosphate in the plaque fluid is also essential. Therefore, modifications have been made on fluoride varnishes to include calcium and phosphate ions within varnish structure in an attempt to further enhance its effectiveness [3]. Functionalized tri-calcium phosphate, or fTCP, is a “smart” calcium phosphate device that permits calcium and phosphate ions delivery to the teeth and acts synergistically with the addition of fluoride to improve efficacy. Although numerous studies have stated the decline in the prevalence of dental caries worldwide, Caries continues to be one of the most prevalent chronic infectious diseases in children and adolescents worldwide. Also, Conditions such as patients with compromised motility, with poor dietary and oral hygiene, or those that use orthodontic devices still need strategies that offer a long-long-term preventive effect. One of these strategies is to enhance the resistance of enamel throughout the application of laser. The process of laser application to increase enamel resistance is a controversial protective technique which lasts for over 50 years with numerous in vitro studies on high density lasers. Erbium lasers (Er:YAG and Er, Cr:YSGG), which are hard tissue lasers, occurred more recently than other kinds of laser. Since enamel contains high amount of water and inorganic content (86% inorganic content, 12% water and 2% lipid and protein) , water and hydroxyapatite have the ability to absorb high quantities of erbium lasers (Er:YAG and Er, Cr:YSGG) which results in thermal changes in the enamel. Thermal variations can cause morphological and/or chemical changes in the structure of enamel. For caries prevention purpose, the sub ablation energy densities have been proposed with Er, Cr:YSGG laser at low fluencies and without water cooling [4].
To deliver and enhance a synergistic preventive effect that lasts for long time, combination of fluoride and laser could inhibit caries and reduce demineralization by numerous mechanisms. Laser irradiation results in microscopic spaces that could trap free fluoride ions. Additionally, it could convert the hydroxyapatite crystals into fluorapatite which increase enamel resistance against demineralization. Some controversies still exist in the literature regarding the appropriate Er, Cr: YSGG laser parameters, when used alone or in combination with fluoride component containing calcium and phosphate, and their efficacy according to the order of application in decreasing the solubility of enamel.
The significance of the study is to achieve additional prevention of enamel demineralization focusing on Er,Cr:YSGG laser irradiation combined with fTCP containing varnish which are hoped to give promising synergistic results [5].
The aim of the present in vitro study was to evaluate the effect of the Er,Cr:YSGG laser, with or without use of tri-calcium phosphate containing varnish, on enamel resistance to demineralization using artificial demineralization taking in our consideration the order of treatment. The effect was studied in vitro using Vickers microhardness test and scanning electron microscopy (SEM).
Material and Method
Ethics statement
This study protocol was performed in vitro, submitted and approved by the Local Ethics Committee (UoM.Dent/ H.L.3/ 21) Research Ethics Committee of Collage of Dentistry, University of Mosul, Nineveh, Iraq.
Teeth samples preparation
A randomized controlled in vitro study design was used. The sample size estimate was calculated using G*Power version 3.1.9.2 program. The sample size of 10 specimens for each group was calculated according to Razeghi et al., (2018) study considering α=0.05, statistical power=80% and effect size=0.52. A total of 50 premolars was needed. Fifty sound upper first premolars with an intact buccal enamel surface were collected from patients aged (16-25) years old who got orthodontic treatment in private clinics in Basra. The teeth were visually examined, and only sound teeth (those with no visible signs of caries) were included in the study. The teeth with caries, restorations, discoloration, enamel developmental abnormalities, wears, cracks, fractures, hypoplasia, white spot lesions, fluorosis and subjected to bleaching agent, hydrogen peroxide or acid etching were excluded from the study. The extracted teeth were cleansed and all residues were removed, then rinsed with deionized water and stored in a 0.1 % thymol solution to prevent dehydration and the development of bacteria and fungi on the tooth surfaces until they were used in fewer than 7 days. After smoothing the teeth with non-fluoridated pumice, the remaining roots were cut 2mm below the cemento-enamel junctions with a straight diamond bur using copious irrigation to avoid harming the enamel, and the crown pulp was removed with an excavator. All specimens were inspected for cracks or enamel defects using a stereomicroscope (10 x) and then embedded in chemically cured resin in plastic rings. Each ring had a single tooth that was fixed in the center of the ring's upper surface, exposing the tooth's buccal surface. The middle third of the crown's buccal surface was marked using a caliper by measuring the distance between the cemento-enamel junction and the tip of the cusp, as well as the mesio-distal dimension. An adhesive tape of (4×2mm2) was fixed on the middle 1/3 of the crowns. Then, the crowns were painted with acid-resistant varnish (Rimmel, London, UK) except for a (4×2mm2) window of exposed enamel. After drying the samples, the tapes were removed, revealing the enamel surface. As in Figure 1.

Figure 1: specimen surface was covered with two layers of nail polish and exposing 2 × 4 mm window.
Materials and equipments
- Clinpro White Varnish that contain 5% Sodium fluoride (2.26% or 22.600 ppm of the fluoride ion) and functionalized tricalcium phosphate (fTCP) exclusively from 3M ESPE, made in USA, expire date 28/7/2022.
- Er,Cr:YSGG laser system (Waterlase i-Plus, Biolase Technologies Inc., San Clemente, CA USA) that emits photons with 78 μm wavelength With parameter of (0.75 W, 8.5 J/cm2, 20 Hz, 11% air, 0% water). The MZ6 tip was chosen for the handpiece's focal area with a beam diameter of 600 μm in order to avoid overheating of the tooth pulp. To ensure a constant spot size throughout the irradiation procedure, an endodontic file fitted with a rubber stop was attached to the fixed handpiece at 1 mm from the enamel surface. Additionally, according to Almaz et al., (2021) study, water cooling is not always necessary. The repetition rate was set to 20 Hz in accordance with the study by Ulusoy et al., (2020) . The irradiation parameters for the laser category were clearly illustrated in table (1). The samples were irradiated once for ten seconds by gently rotating the specimens horizontally by hand while holding the handpiece in its handle to guarantee adequate irradiation and coverage the whole exposed sample surface.
| Laser group | Power (W) | Energy density (J/cm2) | Repetition rate (Hz) | Irradiation time (s) | Water | Air |
|---|---|---|---|---|---|---|
| ER, CR: YSGG (L2) | 0.75 | 8.5 | 20 | 10 | NO | 11% |
Table1: ER, CR: YSGG laser irradiation parameters used in the present study.
Experimental design of the study
Each tooth was given a unique number between 1 and 50 for identification purposes, and these numbers were used to randomly assign samples to one of five study groups. Each group of ten teeth was stored at room temperature in its own beaker labeled with the group name and containing 200 ml of deionized water solution. Following that, each tooth in each group was separately placed in 10 ml deionized water in a plastic jar labeled with the tooth group's name and number and then subjected to the caries prevention approach described below:
- Group (C) (control group): no agent was applied, only washed with deionized water.
- Group (V) (functionalized tricalcium phosphate (fTCP) containing varnish): exclusively from Clinpro White Varnish was applied on the window of specimens’ surface for four minutes according to the manufacturer’s instructions. The specimens then were stored in deionized water for 4 minutes, and then the varnish layer was removed by rubber cap and low-speed handpiece.
- Group (L2) (Er,Cr:YSGG laser with parameter of (0.75 W, 8.5 J/cm2, 20 Hz, 11% air, 0% water): was irradiated as explained previously on the window of specimens’ surface in a scanning style.
- Group (V/L2): First Clinpro White containing functionalized tricalcium phosphate (fTCP) varnish was applied as described for group (V). After 24 hours, the varnish layer was removed. Then, Er,Cr:YSGG laser laser was irradiated as explained previously.
- Group (L2/V): Er,Cr:YSGG laser was irradiated similarly to group (L2) followed by application of Clinpro White containing functionalized tricalcium phosphate (fTCP) varnish with the same detail of group (V).
Then, using the pH cycling model, we used a cycle of demineralization and remineralization to produce artificial caries in all treatment groups. Additionally, all treatments were done by a skilled operator, and the evaluation was done blindly.
PH-Cycling
By subjecting the teeth samples to PH cycling, artificial caries was formed. For three hours, the teeth samples were immersed in a Demineralizing solution containing (CaCl2 2.2mM, NaH2PO4 2.2mM, and acetic acid 0.05M, PH 4.5, adjusted with KOH 1M), followed by twenty hours in a Remineralizing solution containing (CaCl2 1.5mM, NaHPO4 0.9mM, and KCl 0.15mM, PH 7.0). Teeth samples were briefly rinsed with deionized water in between solutions and immersed in artificial saliva composed of (NaCl 0.40, KCl 0.40, CaCL2.2H2O 0.79, NaH2PO4.2H2O 0.78, NaS9.H2O 0.005, CO(NH2)2 Urea 0.1, in 1000 ml distilled water, PH 7 (concentration G \L) for 30 minutes at the end of the demineralization process and 30 minutes at the end of the remineralization process. Ten cycles were performed on the teeth, with each cycle was lasting one day (24 hours). The demineralizing and remineralizing solutions were replaced daily, and the artificial saliva was replaced following each treatment. Preparation of the chemical solutions was done by the researcher at the Marine Science Centre in University of Basra.
Microhardness assessment
All specimens were assessed for their microhardness on two stages: first before the treatments (baseline), and then at the end of the pH cycling stage (post-treatment). A Vickers hardness tester (Jinan Kason Testing Equipment Co., Ltd., China) with the load of 500 gm for 15 seconds was used (according to the instruction of the machine) for calculating the microhardness, as in figure (2). The load and time were constant for all samples. Three indentations were made on each sample's exposed labial enamel surface, and the average value was recorded as the specimen's microhardness. This test was conducted at University of Basra, Collage of Engineering, Department of Mechanic.
VHN= (kg \mm2) = 1.854 × P \d2
P= the testing load in grams.
d= the length of the diagonal line across the indentation in microns.
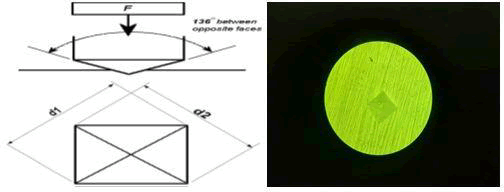
Figure 2: A schematic illustration of Vickers technique (Vickers microhardness machine utilizes a 136° diamond pyramid indenter that forms a square indent on the surface (30). B) An image taken by the researcher of a tetra pyramidal indentation in Vickers technique that creates a clear, measurable indentation in the field as diagonals.
Surface morphology evaluation by Scan Electron Microscope (SEM)
Two specimens from each group that was not subjected to PH cycle were examined under SEM to observe morphological changes on enamel. teeth were mounted on the SEM holder using removable adhesive and their treated surfaces were positioned so that they would face upwards. Under vacuum conditions, each tooth was coated with gold–palladium layer using a sputter coater (Leice EM ce 600 High Vacuum Sputter Coater) and adjusted to be observed under 8 kV accelerating voltage using a SEM (Nova Nano SEM 450) at University of Basra, Collage of Sciences, Department of Physics. The surface morphology of the teeth was examined, and representative photomicrographs were captured and digitally stored.
Statistical analysis
Data were analyzed using the Statistical Package for Social Sciences software program (IBM SPSS Statistics 26). Shapiro–Wilks test was applied to find out the distribution type of experimental measurement data. As the data were normally distributed, the one-way ANOVA test and Duncana test enabled us to decide whether there are any statistically significant differences between the groups or not. A p-value of less than 0.01 was considered statistically significant.
Results
Indentation surface microhardness test results
According to the obtained measurements of this study, table (2) showed the descriptive statistics including means, standard deviations, in addition to the numbers of the samples of tested groups at the baseline and after PH cycle. Based on the means values for tested groups after PH cycle, ER,CR:YSGG laser followed by functionalized tri-calcium phosphate varnish (L2V) had the least reduction in the surface microhardness mean value and thus considered the most effective preventive agent when compared with other tested groups.
| Groups | Variables | Baseline | After PH Cycle |
|---|---|---|---|
| Deionized water (C) | Mean | 310.427 | 190.115 |
| Std. Deviation | 11.77794 | 7.80915 | |
| N | 10 | 10 | |
| functionalized tri-calcium phosphate varnish (V) | Mean | 307.429 | 251.022 |
| Std. Deviation | 7.75936 | 11.98158 | |
| N | 10 | 10 | |
| Er,Cr:YSGG Laser (0.75 W) (L2) | Mean | 311.235 | 245.132 |
| Std. Deviation | 8.19641 | 7.96299 | |
| N | 10 | 10 | |
| fTCP varnish + Er,Cr:YSGG Laser (0.75 W) (VL2) | Mean | 304.757 | 274.735 |
| Std. Deviation | 12.611 | 8.57071 | |
| N | 10 | 10 | |
| Er,Cr:YSGG Laser (0.75 W) + fTCP varnish (L2V) | Mean | 312.711 | 292.18 |
| Std. Deviation | 9.13205 | 7.50193 | |
| N | 10 | 10 |
Table2: Descriptive statistics of microhardness measurements among tested groups at baseline and after PH cycle.
Table (3) ANOVA test explains that there was no significant difference for the surface microhardness readings existed among the tested groups at the baseline at p≤0.01, while after PH cycle, there was a high significant difference among tested groups at p≤ 0.01.
| Microhardness | Sum of Squares | Df | Mean Square | F | Sig. | |
|---|---|---|---|---|---|---|
| Baseline | Between Groups | 407.881 | 4 | 101.97 | 1.003 | 0.416 |
| Within Groups | 4576.864 | 45 | 101.708 | |||
| Total | 4984.744 | 49 | ||||
| After PH Cycle | Between Groups | 59999.002 | 4 | 14999.75 | 188.588 | 0 |
| Within Groups | 3579.177 | 45 | 79.537 | |||
| Total | 63578.179 | 49 | ||||
*highly significant difference at p≤ 0.01
Table3: ANOVA test between tested groups at baseline and after PH cycle respectively at p≤ 0.01.
Duncana multiple analysis range test was done to further explaining that there was no significant difference existed before PH cycle at p≤ 0.01, as all groups were arranged in a homogenous subset of data representing the surface microhardness means values for each group before PH cycle, table (4).
| Groups | N | Subset for alpha = 0.01 |
|---|---|---|
| 1 | ||
| VL2 | 10 | 304.757 |
| V | 10 | 307.429 |
| C | 10 | 310.427 |
| L2 | 10 | 311.235 |
| L2V | 10 | 312.711 |
| Sig. | 0.122 |
Table4: Duncana Multiple Analysis Range test for tested groups before PH cycle.
Duncana multiple analysis range test was done to further explain that there was a high significant difference of microhardness values for groups after PH cycle existed at p≤ 0.01. All groups were arranged in nonhomogeneous subsets of data representing the surface microhardness means values of each group after PH cycle at which L2V group had a highly significant resistance against microhardness loss followed by VL2 group and there is no significant difference between L2 group and V group in surface microhardness mean values while the least value of surface microhardness belonged to the control group where no preventive agent was applied as demonstrated in table (5).
*highly significant difference at p≤ 0.01
Table5: Duncana Multiple Analysis Range test for tested groups after PH cycle.
| Groups | N | Subset for alpha = 0.01 | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| C | 10 | 190.115 | |||
| L2 | 10 | 245.132 | |||
| V | 10 | 251.022 | |||
| VL2 | 10 | 274.735 | |||
| L2V | 10 | 292.18 | |||
| Sig. | 1 | 0.147 | 1 | 1 | |
Table (6) showed that the percentage of surface microhardness loss (SML%) for all groups was calculated according to equation:
SML%=SMH2 –SMh2 / SMh2 × 100 (SML%: the percentage of microhardness loss, SMh2: surface microhardness at baseline, SMH2: surface microhardness after PH cycle).
| Groups | SML% |
|---|---|
| De-ionized water | 38.75% |
| fTCP varnish | 18.34% |
| Er,Cr:YSGG Laser (0.75 W) | 21.23% |
| fTCP varnish + Er,Cr:YSGG Laser (0.75 W) | 9.85% |
| Er,Cr:YSGG Laser (0.75 W) + fTCP varnish | 6.56% |
Table6: The percentage of surface microhardness loss of all tested groups.
Table (6) was further explained in Figure (3) showing that the percentage of loss of surface microhardness for Er,Cr:YSGG Laser (0.75 W) + fTCP varnish group was less than fTCP varnish + Er,Cr:YSGG Laser (0.75 W) , fTCP varnish , Er,Cr:YSGG Laser (0.75 W) and control group of deionized water and thus considers better preventive agent than other groups.
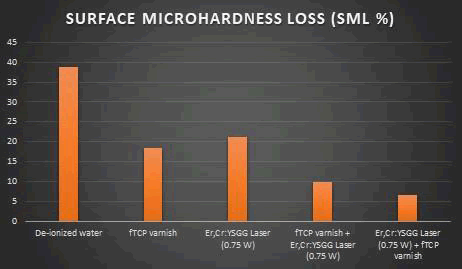
Figure3: The percentage of surface micro hardness loss among tested groups.
Surface morphology by SEM evaluation



Figure4: SEM images of all the groups.
SEM examination of the enamel surface of control (C) group treated with deionized water shows a flattened smooth surface with multiple enamel prism ends and the absence of cracks and surface deposits in the polished untreated enamel. The SEM image of the enamel surface after treatment with functionalized tricalcium phosphate varnish (V) group reveals the formation of a dense surface coating of numerous granular particles and amorphous crystals on flat enamel rods, implying the formation of CaF2-like products. Additionally, no intercrystallite gaps are seen.
The SEM image of the enamel surface treated with ER,CR:YSGG laser irradiation (L2) group reveals a rough surface promoted by the laser irradiation due to partial thermal degeneration and the typical melting appearance can appear with evidence of fissures, cracks, micro ablation areas, conical craters and sharp enamel projections leading to a surface devoid of the typical enamel prism ends and has irregular undulated surface (figure 5). Additionally, globules of CaF2-like content on rough surfaces can be noticed at higher magnifications of 65000 x in (figure 4).

Figure5: SEM images of enamel surface of ER, CR: YSGG laser 0.75 W sample (L2). Original magnification: 8000 x.
The SEM image of the enamel surface treated with functionalized tricalcium phosphate varnish followed by ER, CR: YSGG laser irradiation (VL2) group displays a relatively smooth, homogenous surface in compared to the laser alone and fTCP varnish alone. There are fewer cracks on the glazed enamel surface, which is most likely related to the application of fTCP varnish before laser irradiation. This is associated with the extensive incidence of granular and spherical crystals on the enamel surface.
The SEM image of the enamel surface of ER, CR: YSGG laser irradiation followed by functionalized tri-calcium phosphate varnish (L2V) group demonstrates the formation of large agglomerates and clusters in the irregularities produced by laser irradiation as in figure (4). However, a greater number and concentration of globules and crystals produce primarily in ablated areas, as observed in figure (6).

Figure6: SEM images of enamel surface of ER, CR:YSGG laser 0.75 W combined with fTCP varnish sample (L2V). Rough, irregular and porous surfaces promoted by laser irradiation, which can form and retain the CaF2 -like crystals and globules inside the porous as it can be seen in Original magnification: 10000 x.
Finally, figure (7) shows the result of the energy-dispersive X-ray spectroscopy (EDS microanalysis) along with the morphological evaluation using SEM of enamel surface of ER,CR:YSGG laser (0.75W) followed by functionalized tri-calcium phosphate varnish (L2V) specimen. The distributions of the Ca, P, and F mass percentages on enamel are shown on the enamel surface. The evidence of the fluoride signal (red line) may suggest that these globules could be globules of CaF2 like material.
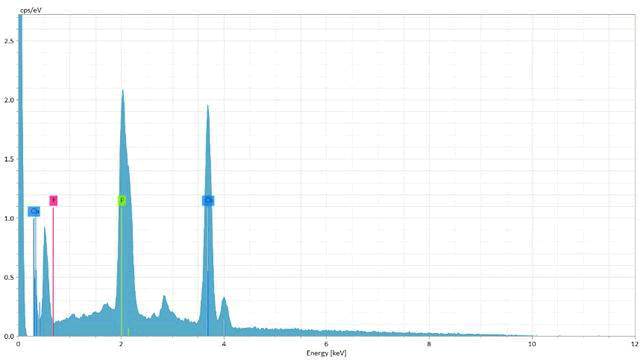
Figure7: EDS analysis graph of a specimen in ER, CR: YSGG laser (0.75W) followed by functionalized tri-calcium phosphate varnish (L2V) group. The distributions of the Ca, P, and F mass percentages on enamel are shown on the enamel surface. The evidencing of the fluoride signal (red line) may suggest that these globules could be globules of CaF2 -like material. EDS, energy-dispersive X-ray.
Discussion
The prevalence of dental caries is ranging from 49 % to 83 % according to the latest Global Oral Health Data Bank report and dental cavities affect almost all age groups. This behavior and prevalence characterize the dental caries assessments and stresses the importance of the interprofessional team in dental caries prevention. The cavities prevention effect of fluoride is owing to the development of acid-resistant fluorapatite crystals, which inhibit demineralization and enhance tooth structure remineralization. Various approaches incorporating the topical use of calcium and phosphate in order to increase effectiveness, including varnish-containing functionalized tri-calcium phosphate (fTCP).
Laser induced enamel resistance is a novel technique for caries prevention. laser photothermic effect will melt and fuse crystals from hydroxyapatite so that more acid resistant enamel will be generated.
However, it is necessary to investigate the clinical efficacy of professional functionalized tri-calcium phosphate containing varnish combined with ER, CR: YSGG laser in preventing dental caries. In addition, a comparison between the preventive effects of these techniques has not been clearly identified before on the teeth of people in the city of Basra. Therefore, evaluating the effectiveness of Er,Cr:YSGG laser’s alone in increasing caries resistance to demineralization with power of 0.75 W and in combination with functionalized tri-calcium phosphate containing varnish (fTCP) in pretreatment and post treatment strategy is the aim of our study.
Er,Cr:YSGG (wavelength: 2.78 μm) has a large hydroxyapatite absorption. Controversial topics are present considering the optimum energy range for caries prevention application of erbium lasers. Some studies claimed that caries prevention should include the sub-ablative energy density. Whereas other researchers have suggested that greater caries resistance can be achieved with higher degree of laser power. The value of ablation threshold depends on laser pulse length, tissue absorption depth through laser pulses and heat diffusion.
In the current study, Er,Cr:YSGG laser beams were applied at energy density of 8.5 J/cm2 accordance. we used 600 μm diameter laser tips to achieve greater energy density with low laser power. The laser frequency (Hz) should be established at a level that can be clinically effective and allow sufficient cooling between pulses at the same time. The quantity of pulses should be adequate, but the total energy transferred must be held to a minimum to prevent pulpal damage.While the frequency value for erbium lasers is not agreed, 20 Hz in this study was used as in the previous studies. More investigations focusing on the need for cooling waters to improve enamel resistance are required as studies using variable cooling water are minimal. In some previous studies, the low energy intensity of laser as well as the use without water would avoid the ablation of the target tissue. Therefore, we chose laser irradiation without cooling of water in our research. As of now, the findings show the inverse relationship between exposure time and caries inhibition degree. This could be clarified because, after 10 seconds, enamel irradiation could result in an enamel surface being heated up to a temperature higher than 650˚C, resulting in chemical changes leading to more soluble, crystal with less resistance to acid. That's why in this study we use the 10 second period in according to Razeghi et al., (2018) study.
The effect of Er,Cr:YSGG laser and fTCP varnish on human permanent teeth enamel’s resistance to demineralization was studied in vitro using Vickers microhardness test, scanning electron microscopy (SEM).
Hardness is a mechanical property that allows the degree of mineralization of the tooth to be determined. It depends directly on the mineral content and the enamel prism crystalline. As enamel softening is a clinical characteristic of caries, laser induced caries prevention can also be assessed by means of a microhardness measurement. Therefore, microhardness test has been used in this analysis because it is known as a straightforward, more precise and less exhausting tool. The overall protective effects of various preventive regimens can be assessed in the test of surface microhardness.
According to the current study, the descriptive statistics include means, standard deviations and the numbers of all study groups at baseline and after PH cycle. Based on the means values, the preventive effect of combination of Er,Cr:YSGG laser irradiation then functionalized tri-calcium phosphate (fTCP ) varnish was better than fTCP varnish alone and laser alone while both of them were beneficial to protect the microhardness of enamel surface of permanent teeth in comparison with the control group of deionized water.
ANOVA Test demonstrated the difference among the tested groups at baseline and after PH cycle and noticed that there was no significant difference at baseline, while after the addition of the test materials and introduction in to the PH cycle, there was a high significant difference among tested groups as further noticed at Duncana, a Multiple Analysis Range Test for test groups after PH cycle, which illustrated that combination of Er,Cr:YSGG laser irradiation then functionalized tri-calcium phosphate varnish (L2V) group had a minimum reduction in the microhardness mean value followed by combination of functionalized tri-calcium phosphate varnish then Er,Cr:YSGG laser irradiation (VL2) group. While there was no significant reference between L2 group and V group while both of them were beneficial to protect the microhardness of enamel surface of permanent teeth in comparison with the control group of deionized water.
In fTCP varnish Group, there was 18.34% loss in the percentage of microhardness as compared to the control group of deionized water which has 38.75 % loss in the percentage of microhardness after ph. cycle. Calcium fluoride (CaF2) has been developed on enamel surfaces and fluoride was released to fluid levels after a professional application of fluoride with calcium and phosphate has been performed. This effect helps to reduce the demineralization of enamel. In addition, the dose-response effect between enamel mineralization and fluoride released through reserve concentrations of CaF2, and subsequent inhibition of enamel demineralization was observed [48]. The findings of present study have shown that topical application of fTCP varnish was effective in the prevention of demineralization process. The research of Alamoudi et al., (2013) revealed that fTCP containing varnish had significantly higher SMH than unprotected teeth, and hence the protective effect was substantially higher. The current study favored the use of fTCP varnish to enhance the time of contact between fluoride and tooth surface to make the tooth more caries resistant.
Earlier studies found that the increase in laser power at sub-ablative threshold (8-13 J/cm2) would increase the superficial temperature and produce more morphologically and crystallographic modified enamel including alpha-phase formation of tricalcium phosphate, beta- phase tricalcium phosphate and tetra calcium phosphate. These changes may be the cause of improved overall demineralization resistance of irradiated enamel identified in previous studies [51]. In other words, Researchers have also attributed improvements in surface resistance to the photo-chemical effect of a laser by lowering the carbonate contents or partial decomposition of the organic matrix. These results of previous studies were similar to the present study , so, The microhardness of samples with laser power density of 8.5 J/cm2 have increased compared with control samples and there was 21.23% loss in the percentage of microhardness in L2 group after ph. cycle as compared to control group of deionized water with 38.75%. Although, Apel et al., (2004) indicated that 8 J/cm2 energy density was not able to cause changes in surface hardness following an in-situ cariogenic challenge. The difference may be related to sample differences including total mass, hardness, thickness, hydration and other characteristics can lead to different results when the specimen interacts with lasers. Pulse duration, laser fluence (efficient beam diameter on sample) and repetition rate are other significant factors. Laser equipment/manufacturer and irradiation requirements must also be taken into consideration in predicting laser effects of enamel in addition to the sample characteristics.
In the present study the advantages achieved by laser radiation alone are not superior to those achieved by topical application of fTCP varnish, whereas laser at power density of 8.5 J/cm2 had statistically similar effects to fTCP varnish.
The percentage of surface microhardness loss of fTCP varnish group was 18.34% in compared with L2 group which had 21.23% of surface microhardness loss. This differences could be due to the laser radiation effect formed in the production of fine irregularities and cracks in enamel surfaces that favored enamel brittleness as seen in SEM laser group images , as in figure 4, which revealed that the enamel surface was melted and fused with the formation of cracks. Enamel surface cracks were not seen in the fTCP varnish group micrographs and were covered with amorphous crystals and globular deposits. These cracks serve as the base for acid attacks and cause enamel to be fragile, resulting in a reduction of SMH. These findings were in agreement with Ana et al., (2012) results which indicated that the advantages achieved with laser radiation alone are not higher than those with topically treated fluoride treatment but the laser that applied at 8.5 J/cm2 had similar effects to acidulated fluoride.
A few studies have tested the effects of combination of fluoride application and Er,Cr:YSGG laser irradiation on demineralization prevention of permanent teeth enamel. In the literature, some inconsistencies regarding the order of fluoride application and laser irradiation can be found. Tagomori and Morioka (1989) found that laser irradiation with a subsequent application of fluoride can lead to greater intake of fluoride in enamel than when reversing the order. Alternatively, laser irradiation of the fluoride covered enamel could cause the enamel’s outer layer to melt and fluoride ions to bind to the hydroxyapatite crystals with the formation of fluorapatite crystals that are more resistant to acid than hydroxyapatite with improvement in mechanical properties of the calcium fluoride-like deposits. Vieira et al., (2015) have the same findings. The authors found that it was possible for hydroxyapatite crystals to convert directly into fluorapatite crystals using laser irradiation in the presence of fluoride. In contrast, Bedini et al., (2010) stated that laser irradiation of the enamel combined with fluoride (fluoride varnish and/or APF) did not show better results than in case of control groups, and caries activity that resulted in the formation of cavities occurred after just 6 months, indicating no synergistic impact [55]. The conflicting results are most likely due to a large number of variables such as laser type, energy, pulse frequency and radiation duration. Razeghi et al., (2018) findings revealed that combination of Er,Cr:YSGG laser irradiation and application of topical fluoride have no superiority over either laser irradiation or fluoride application alone in increasing microhardness of primary teeth enamel, regardless of order of treatments or laser power. He worked on primary teeth while caries progression pattern and its prevention are not similar in primary and permanent teeth.
In the present study, the combination of Er,Cr:YSGG laser irradiation and functionalized tri-calcium phosphate varnish in L2V group and VL2 groups was more resistance to demineralization than laser alone and fTCP varnish alone. Also, the combination of Er,Cr:YSGG laser irradiation then functionalized tri-calicum phosphate varnish (L2V) group has higher enamel resistance to demineralization compared with the combination of functionalized tri-calicum phosphate varnish then Er,Cr:YSGG laser irradiation (VL2) group. There was 6.56% loss in the percentage of microhardness in L2V group and 9.85% loss in the percentage of microhardness in VL2 group as compared to the control group of deionized water which has 38.75 % loss in the percentage of microhardness after ph. Cycle. It may be due to the amount of fluoride entrapment inside the pores created by laser, so the fluoride has the ability to increase enamel resistance against demineralization. By mean of laser irradiation, microscopic spaces can be created and trapped the free fluoride ion. In addition, hydroxyapatite can become fluorapatite, making it safer against demineralization. In terms of the creation and persistence of CaF2-like material, laser irradiation improved the production of CaF2-like material at an energy density of 8.5 J/cm2 followed by topical application of fluoride which make the enamel more resistance to demineralization.
Overall, the diversity of results from different studies is likely due to variations in irradiation parameters, laser settings and demineralization measurement methods used in various studies. However, variations in results could be due to the fact that their fluoride agent and demineralization solutions were not similar to those used in the current study.
The surface morphology of enamel was viewed using Scanning electron microscope examination. The scanning electron micrographs obtained in the control study showed the typical design of the regular enamel that is fairly smooth with shallow depression and enamel prisms. The smooth and rough eroded surfaces were observed on Er,Cr:YSGG laser group which can retain the CaF2 -like material globules. There was a slight melting and thermal degeneration of the surface, giving lava molten like appearance and an irregular structure. However, Laser caused deeper and wider craters with significant margins suggesting micro explosions and melting [58]. The overall effect of the Er,Cr:YSGG laser was not homogeneous; the effect was found only in regions that were in direct contact with the laser. Molten lava-like regions were found in previous laser-induced demineralization prevention (LIPD) studies due to thermal degeneration and lack of cooling. Similar detrimental effects were found in the SEM examinations. The resistance of the enamel achieved at lower temperatures was therefore argued to be caused by decomposition of the organic matrix protein and blocking of interprismatic spaces. The morphologic alterations of the irradiated enamel surface increase its resistance to demineralization.
The SEM micrographs of both the lasing + varnish and that of varnish +lasing group were in accordance with the large surface granules, globules and crystals adhering in L2V group, compared with VL2 group, were present. The crystals are heavily packed crystals emerging from porosities that was formed by laser action. The morphological modifications on the irradiated surfaces of enamel make it more resistant with regard to demineralization. The surface granular and globular material coatings present may provide a reservoir of mineral phases during a cariogenic attack. Surface layer contains higher quantity of fluoride in the form of calcium fluoride achieved by reaction of fTCP varnish with enamel surface making it more resistant to caries and increasing the microhardness and make it resistant to caries. In the present study EDS analysis showed fluoride red signal in L2V group sample and confirmed these coating may be calcium fluoride crystals in the surface of enamel.
Additional research is necessary to completely characterize the surface and chemical changes in enamel following laser and/or fTCP varnish treatment. The experimental findings of this study showed promising opportunities with several clinical limitations. The combined application of laser and fTCP varnish treatment was done only once followed by high reduction in the percentage of enamel mineral loss expressed by the values of enamel microhardness. This could give benefits for patient’s treatment as it would not depend on frequent use of mouth rinses. However, future studies require to be conducted in order to assess the long-term predictability of this innovative demineralization and caries prevention of combined therapy. Other studies may also be required to evaluate the amount of mineral loss and assessment the long-time effect of the CaF2 -like material globule formation on enamel surface.
Conclusion
The Er:Cr:YSGG Laser at energy density of 8.5 J/cm2 is indicated on decreasing the loss of enamel hardness with comparable effects to fTCP varnish application. The impacts on hardness loss suggest that there is an advantage in using laser at 8.5 J in combined with fTCP varnish over using laser alone and fTCP varnish alone with more benefit when laser was applied before topical application of fTCP varnish than the opposite.
References
- Almaz M, Ulusoy NB, Akbay Oba A et al. Thermal, morphological, and spectral changes after Er, Cr:YSGG laser irradiation at low fluences on primary teeth for caries prevention. Microsc Res Tech. 2021; 84: 150– 159.
- Shen P, Bagheri R, Walker GD et al. Effect of calcium phosphate addition to fluoride containing dental varnishes on enamel demineralization. Aust Dent J. 2016;61(3):357-65.
- Toumba, K. J., Twetman, S., Splieth, C et al. Guidelines on the use of fluoride for caries prevention in children: an updated EAPD policy document. Eur Arch Paediatr Dent. 2019; 20: 507–516.
- Chen KJ Gao SS, Duangthip D. The caries-arresting effect of incorporating functionalized tricalcium phosphate into fluoride varnish applied following application of silver nitrate solution in preschool children: study protocol for a randomized, double-blind clinical trial. Trials. 2018;19:352.
- Zamataro CB, Ana PA, Benetti C et al. Influence of Er,Cr:YSGG laser on CaF2-like products formation because of professional acidulated fluoride or to domestic dentifrice application. Microsc Res Tech 2013; 76:704–713.
Author Info
Hussein Sami Abdullah1* and Aisha Akram Qasim2
1Researcher (Master student) College of Dentistry, University of Mosul, Mosul, Iraq2Department of Pedo. Ortho. Preventive Dentistry, College of Dentistry, University of Mosul, Mosul, Iraq
Citation: The Effect of Combined Application of Tricalcium Phosphate Containing Varnish and ER, CR: YSGG Laser on Resistance of Enamel to Caries: An In Vitro Study, J Res Med Dent Sci, 2021, 9(12): 558-568
Received: 01-Dec-2021 Accepted: 15-Dec-2021 Published: 22-Dec-2021
