Research Article - (2023) Volume 11, Issue 1
The Wound Healing Potential of Iraqi Origanum majorana L n-Hexane and Ethyl Acetate Fractions in a Rat Model of Excisional Wound Repair: Pre-Clinical Test
Sakar Dilshad Asaad1*, Thukaa Z Abdul Jalill1 and Ammar A Fadhil2
*Correspondence: Sakar Dilshad Asaad, Department of Pharmacognosy, College of Pharmacy, University of Baghdad, Baghdad, Iraq, Email:
Abstract
Origanum majorana (Majorana hortensis), an evergreen herbaceous plant belonging to the Lamiaceae family, has been well known for being used for gastrointestinal, cardiac, respiratory, rheumatologic and many other illnesses, but in wounds management hasn’t been qualified scientifically yet. The goal of the study was to evaluate the wound healing properties of sterols in n-hexane and phenols in ethyl acetate extract fractions of the Iraqi Origanum majorana L aerial parts by contrasting their wound healing abilities with those of commercially available MEBO ointment in a rat excised wound repair model. At various periods, the size of the wounds was measured and skin tissue samples were taken for histopathology. When compared to positive and negative controls, the formula's inclusion of sterols and phenols led to a significant decrease in wound diameters. According to estimates, the ethyl acetate extract fraction with the highest concentration of phenolic compounds gave the best results, as proved by qualitative phytochemical analytical screening for the Origanum majorana L extracts, giving a faster rate of wound closure.
Keywords
Origanum majorana L, MEBO ointment, Phenolic and sterols compounds, Ethyl acetate fraction, N-hexane fraction, Wound healing, Excision wound
Introduction
A wound is defined as an interruption in the cellular and anatomical structure of body tissue, including the skin, bodily fluid film, deep lying tissues or the surface of inner organs. Wounds can range from cut, slash, scraped area, cut and closed wounds like hematoma, conclusion and pulverize wounds [1,2]. Severe wounds, abnormal metabolic patterns and long term framework maladies such as diabetes and hyperglycemia can all contribute to it [3,4]. Wound recovery is actually moderate and can end up unending with a long clinical course, resulting in a continuous discharge of fiery modulators that cause agony and edema. In severe cases, persistent wounds can become infected with microorganisms, resulting in wound healing delays, septicemia, organ dejection and death [5]. Wound healing is a complicated process in which the skin or other human tissue heals and the dermis forms a protective barrier against the external environment. When the barrier is breached, a rapid cascade of biochemical exercises is initiated to repair the damage. Homeostasis, irritation, modern tissue advancement (expansion) and tissue remodeling (development), angiogenesis, collagen testimony, granulation tissue creation, epithelialization and wound withdrawal [6]. For essential healthcare, therapeutic plants play an important role in the unused time of modern pharmaceuticals. Around 80% of individuals in immature nations still depend on conventional solutions, which are for the most part based on plant and creature species. Natural product based medicines account for roughly 30% of global drug sales. This movement is based on the idea that plants have a huge potential for use as therapeutic medication [7]. The most common therapies for skin lesions are substances derived from herbs. These treatments involve applying herbs, completed herbal products and herbal preparations that have biologically active ingredients that can speed up the healing process. Today, a vast range of plants from all over the world are being studied and used to cure skin diseases [8,9]. Herbal based products are prepared as extracts, emulsions, creams and ointments. An evergreen herbaceous plant belonging to the lamiaceae family called Origanum, with numerous species, is widely used in perfumery, alcoholic beverages, food products and other products to flavor because of the spicy aroma. In addition to their commercial value, these plants have been used for a long time as seasonings and tastes in foods, including salads, soups, wieners and meats. It has recently been found to be hostile to characteristics that cause ulcers, lipidemia and hyperglycemia. Origanum majorana (Majorana hortensis (M) or as sweet marjoram) was widely used in the treatment of a variety of illnesses because of several beneficial properties, including sudorific, stomachic, emmenagogic, stimulant, sterile, expectorant, hepatoprotective and nephroprotective [10,11]. These beneficial properties are related to the presence of naturally active compounds such as thymol, carvacrol, sitosterol, tannins, cis-sapine hydrate, limonene, hydroquinone, terpinene, camphene and flavonoids such as dioscin, quercetin, luteolin and apigenin [12]. However, there are no detailed logical reports on its wound healing process available. The aerial section of an Iraqi Origanum majorana L plant was used in the display study to evaluate the wound healing movement of the ethyl acetate fraction and the n-hexane fraction.
Aim of study
To investigate the wound healing property of sterols in nhexane extract and phenols in ethyl acetate extract fractions of the aerial parts of Iraqi Origanum majorana L in a rat model of excisional wound repair.
Materials and Methods
Plant material collection and identification
Between February and April, the cultivated Iraqi Origanum majorana L plant fabric airborne parts (fresh takes off and lean stems) were taken from Al-Hosseinia city ranchers in Baghdad, Iraq. Dr. Khansaa Rasheed, plant and environment department/Iraqi national history investigate center and museum/University of Baghdad, identified, confirmed and authenticated these fresh parts. The plants were cleaned, dried in the shade at room temperature, crushed using an electrical technique and weighed just before extraction.
Extraction of plant material
The coarsely powdered aerial parts (100 g) were macerated in 200 mL of n-hexane for two days at room temperature and then repeated every two days for three days. The supernatants from each plant material were filtered and subsequently concentrated by solvent evaporation at room temperature [13]. The procedure yielded a dark green concentrated extract (n-hexane fraction) of 5 g. The defatted plant material was dried and extracted using a soxhlet apparatus with 85% ethanol and then the crude ethanol extract was filtered, concentrated under reduced pressure to about 6.8 g and mixed with 200 ml of distilled water, which was designated as the crude fraction. The crude portion was partitioned using the solvent ethyl acetate (2 × 100 ml) [14]. A dark brown color of 2 g of concentrated extract (ethyl acetate fraction) was obtained from the process after drying.
Formulation of herbal ointment
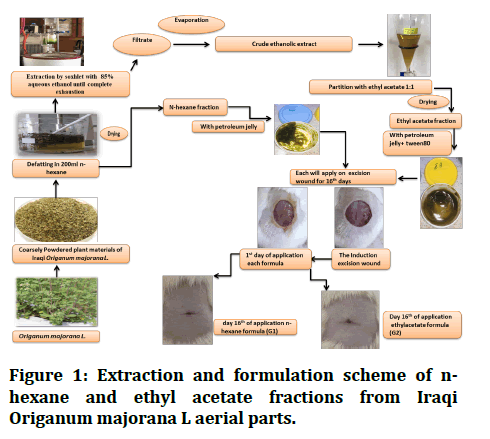
The medicated ointment was prepared by incorporating 0.4 g of each extract fraction into 40 g of petroleum jelly (vasaline) as a hydrocarbon (oleaginous) base with the addition of polysorbate 80 (tween 80) as a surfactant to enhance the solubility of ethyl acetate extract fractions in vaseline. The fusion process was used in order to create the herbal ointment. A water bath was used to melt the necessary amount of ointment base at a temperature of roughly 70°C. A homogeneous dispersion was created by adding the specified amount of the fraction to the melted base while stirring gently and constantly at 40°C in a water bath [15]. These formulations were stored in the fridge and allowed to come to room temperature for 30 minutes before they could be used for wound treatment each time (Figure 1).
Figure 1: Extraction and formulation scheme of nhexane and ethyl acetate fractions from Iraqi Origanum majorana L aerial parts.
Qualitative phytochemical analysis
The presence of steroids, fixed oils, terpenoids, tannin, phenolic compounds and flavonoids were identified by making a general test as a qualitative phytochemical screening of Iraqi Origanum majorana L aerial parts extracts following standard procedures as described by harborne [16].
Experimental animals
Twenty four healthy adults Wister albino rats of either sex with (120 g-140 g) body weight were used, matured between (2) months, obtained from the animal house of the college of pharmacy/University of Baghdad. For about two weeks prior to the experiment, animals are accommodated in a same location and individual cages under standardized temp, humidity and light/dark cycle conditions. All experimental animals were nourished a standard rodent pellet diet and given unlimited access to water. The research protocol was approved by the local ethical committee at the University of Baghdad's college of pharmacy.
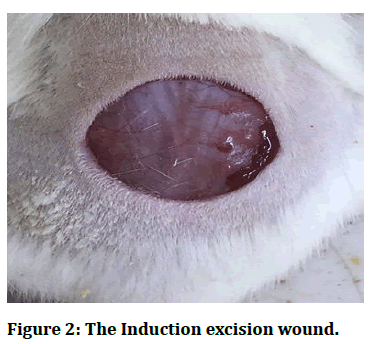
Excision wound model: On the first day, each rodent was anesthetized by utilizing diethyl ether. At that point, the rats were kept clean and shaved (1.5 cm × 1.5 cm). By evacuating a fix of skin, a full thickness open extraction wound was created (Figure 2). At that point, the therapeutic treatments were connected topically once every day for 15 days. All wounds were uncovered for discussion and checkout day by day. The estimation zones of the extraction wound were taken on the 1st, 4th, 8th, 12th and 16th days [17].
Figure 2: The Induction excision wound.
Grouping and dosing of animals: Animals were randomly divided into four groups (6 rats in each group) as following:
• Group 1: Wounds were treated with topical 1% nhexane fraction formula.
• Group 2: Wounds were treated with topical 1% ethyl acetate fraction formula.
• Group 3: Wounds were treated with a simple ointment base (negative control).
• Group 4: Wounds were treated with 0.25% MEBO ointment (positive control).
Wound healing evaluation
Measurement of wound area: Photography’s of the wound area were obtained regularly every 2 days by a camera phone to observe wound surface areas healing and constriction. Also, the wound area was measured every four days by manually tracing the wound. The wound shapes and surface areas were both measured. The wound contraction was expressed as a percentage reduction in the original wound size. The percentage of wound contraction was calculated using the equation below [18]:
% Wound contraction=(Current wound area/Wound area at the beginning) × 100
Histological examination: On day 16, rats' skin specimens from wound healed areas were sacrificed and the healed skin was assessed by taking a 5 mm2 section and removing it. The sections were then fixed in 10% buffered formalin and processed through a paraffin tissue processing machine before being dyed with hematoxylin and eosin for examination under light microscopy [19,20].
Statistical analysis: All data were presented as means with Standard Deviations (SD). Two way repeated measure Analysis of Variance (ANOVA) was used to analyze the data [21]. A statistically significant difference was defined as a p value less than 0.05.
Results and Discussion
Herbal medicines are valued and frequently used in the process of healing wounds. The bioactive substances isolated from plant materials (leaves, roots, bark, fruit, etc.) have been clinically demonstrated to aid in wound healing, in addition to being affordable and readily available. On wound excision models, this study examined the topical preparation of Origanum majorana aerial parts extract fractions' capacity to cure wounds. The universal solvent 85% ethanol is well known for its ability to attract both polar and non-polar molecules. Oily medicated compositions were recommended for therapeutic or aesthetic purposes. Vaseline is also an oily base, which supports the absorption of active ingredients into the target tissue while preserving skin hydration. The benefit of this oily foundation affects the amount of time the ointment's active component is in touch with the target area.
Qualitative phytochemistry
A qualitative phytochemical study of the fractions revealed the presence of many phyto compounds (Table 1). Steroids and terpenoids were found mainly in the nhexane fraction, while tannins, phenolic compounds and flavonoids were more in the ethyl acetate fraction.
| Fractions | N-hexane | Ethyl acetate |
|---|---|---|
| Phyto constituents | ||
| Phenolic compounds and flavonoids | - | + |
| Steroids | + | - |
| Tannins | - | + |
| Terpenoids | + | - |
| The key +: Present, â??:Absent | ||
Table 1: Phytochemical constituents of n-hexane and ethyl acetate fractions of Origanum majorana L aerial parts.
Evaluation of n-hexane and ethyl acetate fractions pharmacological properties on wound healing
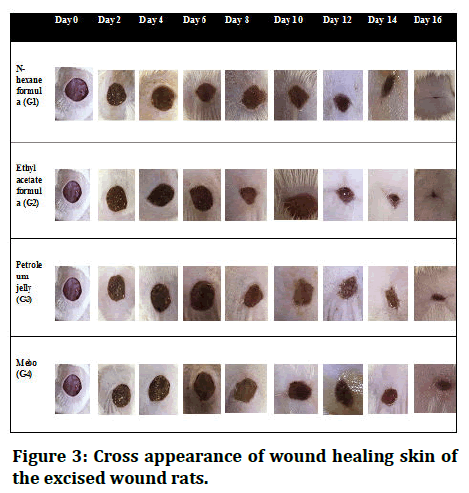
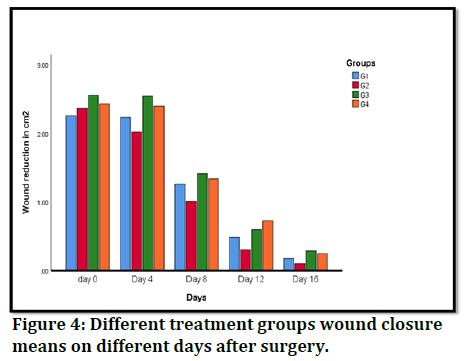
Wound contraction rate: In order to evaluate the effects of n-hexane and ethyl acetate extract fractions on excision wound healing, wound size must be measured at various time intervals. n-hexane and ethyl acetate fractions dramatically decrease wound estimates starting on day 3 following surgery as compared to the negative control group. Additionally, ethyl acetate fraction therapy had considerably greater wound healing mobility than nhexane treatment and MEBO treatment at all-time points examined. The effects of n-hexane on wound compression rate on days 8 and 12 following surgery were considerably superior to MEBO, according to the comparison of wound estimates between the n-hexane treated group and the MEBO treated group. n-hexane performed better on day 3. However, on day 16, the nhexane and ethyl acetate fractions had a higher impact on the rate of wound closure than the standard and negative controls. Figure 3 depicts clearly discernible wound healing patterns, whereas Table 2 and Figure 4 present pertinent factual findings.
Figure 3: Cross appearance of wound healing skin of the excised wound rats.
| G1 | G2 | G3 | G4 | |
|---|---|---|---|---|
| Mean of wound area Cm2 ± SD | Mean of wound area Cm2 ± SD | Mean of wound area Cm2 ± SD | Mean of wound area Cm2 ± SD | |
| Day 0 | 2.260333 ± 0.138566 | 2.366667 ± 0.287796 | 2.561667 ± 0.118561 | 2.433333 ± 0.248891 |
| Day 4 | 2.236667 ± 0.153058 | 2.021667 ± 0.588776 | 2.548333 ± 0.126082 | 2.401667 ± 0.25119 |
| Day 8 | 1.265 ± 0.305794 | 1.006667 ± 0.202846 | 1.415 ± 0.265387 | 1.343333 ± 0.548039 |
| Day 12 | 0.483333 ± 0.220151 | 0.306667 ± 0.099532 | 0.596667 ± 0.126122 | 0.726667 ± 0.398631 |
| Day 16 | 0.178333 ± 0.096833 | 0.103667 ± 0.037554 | 0.29 ± 0.044721 | 0.248333 ± 0.095586 |
| Values are expressed in Mean ± SEM of 6 animals. | ||||
Table 2: The effect of extract fractions of Origanum majorana L on excised wounds in rats.
Figure 4: Different treatment groups wound closure means on different days after surgery.
Furthermore, statistically the results show that there is shrinkage in the percentage of wound surface area when dealing with partial depth excised wound in rats (Table 3).
| G1 | G2 | G3 | G4 | |
|---|---|---|---|---|
| % of wound area contraction (Mean ± SEM) | % of wound area contraction (Mean ± SEM) | % of wound area contraction (Mean ± SEM) | % of wound area contraction (Mean ± SEM) | |
| Day 0 | 0 | 0 | 0 | 0 |
| Day 4 | 98.91762 ± 0.337434 | 84.50264 ± 7.07199 | 99.46784 ± 0.2927 | 98.68193 ± 0.355682 |
| Day 8 | 55.52942 ± 4.320804 | 43.48014 ± 5.03702 | 54.963 ± 3.487508 | 54.40173 ± 7.300998 |
| Day 12 | 21.01319 ± 3.536653 | 13.02427 ± 1.669769 | 23.18834 ± 1.710164 | 29.61474 ± 6.233834 |
| Day 16 | 7.755 ± 1.64504 | 4.428836 ± 0.639573 | 11.31158 ± 0.648541 | 10.25466 ± 1.662919 |
Table 3: The effect of extract fractions of Origanum majorana L on the percent of wound contraction with respect to the zero day of excised wound in rats was studied.
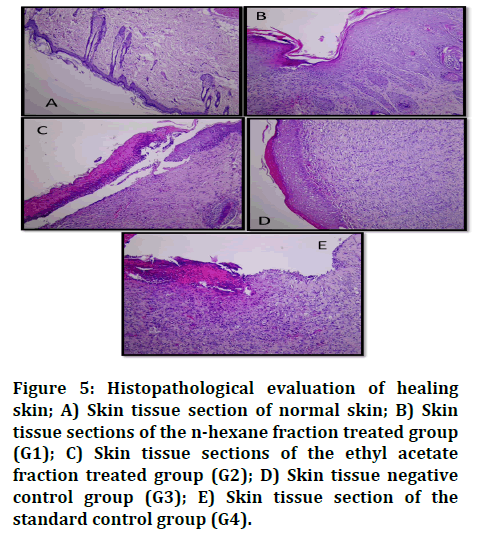
Histopathological analysis: The wound biopsies were studied on day 16 post excision and the results revealed that the skin tissue section of normal skin consisted of stratified epithelium and dermis layer with the insertion of hair shift and sebaceous glands (Figure 5A). The nhexane fraction treated group (G1) showed reepithelialization of stratified epithelium and the presence of granulation tissue in the upper dermis (Figure 5B). Ethyl acetate fraction treated group (G2) showed reepithelization of stratified epithelium with inflammatory cell infiltration in the keratin layer and the presence of granulation tissue in the upper dermis (Figure 5C). Negative control group (G3) re-epithelization of stratified epithelium and formation of granulation tissue with abundant fibrous tissue formation (Figure 5D). The standard control group (G4) section still had ulceration of the stratified epithelium and a heavy inflammatory cell reaction with scab formation on the surface and abundant granulation tissue (Figure 5E). In all sections, granulation tissue formation and collagen deposition are found. The result from histological data found wound healing in rats treated with ethyl acetate fraction was better than the n-hexane fraction treated group, the negative control group and the reference groups due to the healing of wounds with less complications.
Figure 5: Histopathological evaluation of healing skin; A) Skin tissue section of normal skin; B) Skin tissue sections of the n-hexane fraction treated group (G1); C) Skin tissue sections of the ethyl acetate fraction treated group (G2); D) Skin tissue negative control group (G3); E) Skin tissue section of the standard control group (G4).
Terpenoids, sterols, glycosides, tannins and flavonoids were found in the extract fractions after a phytochemical examination. Typically, these metabolites are accountable for the pharmacological effects of medicinal plants [22]. According to reports, flavonoids have the ability to promote wound healing. Terpenoids are well recognized for accelerating the healing process of wounds, particularly because of their astringent and antibacterial properties, that seem to be the cause of wound contraction and accelerated epithelialization. It is well known that flavonoids and their derivatives reduce lipid peroxidation via enhancing vascularity, which slows down cell necrosis. Additionally, phlobatannins have been shown to be involved in the healing of wounds [23]. Therefore, the contributions of several phytoconstituents may be used to explain the ability of Origanum majorana to cure wounds. Phlobatannins and flavonoids may have had a significant role in the extract fraction's overall ability to heal wounds, as evidenced by the considerable activity seen in the ethyl acetate fraction. Origanum majorana has been said to have the combined actions of antibiotics, anti-inflammatory drugs, astringents and antipyretics, which are more or less necessary for conventional wound therapy. As a result, the extracted fractions can be utilized to heal wounds.
Conclusion
Wound healing is a complex phenomenon that encompasses several different phases, concentrating on wound contraction and epithelization, which are separate and run concurrently. The ethyl acetate and n-hexane extract fractions of Origanum majorana L aerial parts exhibit wound healing properties in vivo in albino wister rats. The results strongly suggest that wound healing promotion and acceleration can be achieved based on faster healing when compared to negative and standard control wounds as contraction, epithelization and tensile strength were improved. By using plainly visible, morphometric and histological data, Origanum majorana extracts were more potent than standard commercially available remedies for clinical use, while the recovery time was 14 days (two days shorter than the standard), giving significant benefit over the control and reference groups.
Acknowledgement
Thanks to the university of Baghdad, college of pharmacy, professors of the pharmacology toxicology and of the pharmacognosy and medicinal plants branch for providing necessary facilities and assistance.
References
- Sowjanya K, Spandana U, Manjula RR, et al. Wound healing activity and anti-ulcer activity of ethanolic extract of peels of Ananas comosus. European J Pharm Med Res 2016; 3:417-422.
- Sharma Y, Jeyabalan G, Singh R. Potential wound healing agent. Pharmacologia 2013; 4:349–358, 2013. [Crossref]
- Gushiken LFS, Beserra FP, Bastos JK, et al. Cutaneous wound healing: An update from physiopathology to current therapies. Life 2021; 11:665. [Crossref][Googlescholar][Indexed]
- Kazerouni A, Kazerouni O, Pazyar N. Effects of ginger (Zingiber officinale) on skin conditions: A non-quantitative review article. J Turk Acad Dermatol 2013; 7:10-13.[Crossref][Googlescholar][Indexed]
- Adiele LC, Adiele RC, Enye JC. Wound healing effect of methanolic leaf extract of Napoleona vogelii (Family: Lecythidaceae) in rats. Asian Pac J Trop Med 2014; 7:620–624. [Crossref][Googlescholar][Indexed]
- Variji Z, Aghazadeh N, Hasanzadeh H, et al. Extracorporeal shock wave therapy in the treatment of non-healing diabetic ulcer: A pilot study. J Clin Exp Dermatol Res 2015; 6:2. [Crossref][Googlescholar][Indexed]
- Sabale P, Bhimani B, Prajapati C, et al. An overview of medicinal plants as wound healers. J Appl Pharm Sci 2012; 2:143–150. [Crossref][Googlescholar][Indexed]
- Manjula RR, Uppuluri S, Anand TJ, et al. Wound healing effect of methanolic flower extract of Bauhinia tomentosa Linn with emu oil in rats. J Pharmacogn Phytochem 2017; 6:247–253.
- “Technical Publications of the South African National,” 2009; 39:107, 2009.
- Mnif W, Dhifi W, Jelali N, et al. Characterization of leaves essential oil of Pelargonium graveolens originating from Tunisia: Chemical composition, antioxidant and biological activities. J Essent Oil Bear Pl 2011; 14:761-769. [Crossref][Googlescholar][Indexed]
- Pimple BP, Patel AN, Kadam PV, et al. Microscopic evaluation and physicochemical analysis of Origanum majorana Linn leaves. Asian Pacific J Trop Dis 2012; 2:S897-S903. [Crossref][Googlescholar][Indexed]
- Altemimi A, Lakhssassi N, Baharlouei A, et al. Phytochemicals: Extraction, isolation and identification of bioactive compounds from plant extracts. Plants 2017; 6:42. [Crossref][Googlescholar][Indexed]
- Walia H, Kumar S, Arora S. Comparative antioxidant analysis of hexane extracts of Terminalia chebula retz. prepared by maceration and sequential extraction method. J Med Plants Res 2011; 5:2608-2616. [Googlescholar][Indexed]
- Lama-Munoz A, Espinola F, Moya M, et al. Content of phenolic compounds and mannitol in olive leaves extracts from six Spanish cultivars: Extraction with the Soxhlet method and pressurized liquids. Food Chem 2020; 320:126626. [Crossref][Googlescholar][Indexed]
- Laut M, Ndaong NA, Utami T. Cutaneous wound healing activity of herbal ointment containing the leaf extract of Acalypha indica L. on mice (Mus musculus). J Phys Conf Ser 2019; 1146:012025. [Crossref][Googlescholar][Indexed]
- Harbone, “Harborne” B Phytochem 1987.
- Dons T, Soosairaj S. Evaluation of wound healing effect of herbal lotion in albino rats and its antibacterial activities. Clin Phytoscience 2018; 4:0–6. [Crossref][Googlescholar][Indexed]
- Okur ME, Ayla S, Cicek polat D, et al. Novel insight into wound healing properties of methanol extract of Capparis ovata Desf. var. Palaestina Zohary fruits. J Pharm Pharmacol 2018; 70:1401-1413. [Crossref][Googlescholar][Indexed]
- Ayla S, Gunal YM, Sayin S, et al. Effects of Prunus spinosa L. fruits on experimental wound healing. Medeni Med J 2017; 32:152–158. [Crossref][Googlescholar][Indexed]
- Najim SM, Fadhil AA, Abdullah MN, et al. Estimation of the healing effects of the topical use of MEBO and hyaluronic acid gel in the burned rats. J Adv Pharm Educ Res 2022; 12:91–97. [Crossref]
- Shaygan S, Fakhri S, Bahrami G, et al. Wound healing potential of Cucurbita moschata duchesne fruit peel extract in a rat model of excision wound repair. Adv Pharmacol Pharm Sci 2021; 2021:6697174. [Crossref][Googlescholar][Indexed]
- Umeh V, Ilodigwe E, Ajaghaku D, et al. Wound healing activity of the aqueous leaf extract and fractions of Ficus exasperate (moraceae) and its safety evaluation on albino rats. J Tradit Complement Med 2014; 4:246–252. [Crossref][Googlescholar][Indexed]
- Jain PS, Bari SB. Evaluation of wound healing effect of petroleum ether and methanolic extract of Abelmoschus manihot (L.) Medik, Malvaceae and Wrightia tinctoria R. Br apocynaceae in rats. Rev Bras Farmacogn 2010; 20:756–761. [Crossref][Googlescholar][Indexed]
Author Info
Sakar Dilshad Asaad1*, Thukaa Z Abdul Jalill1 and Ammar A Fadhil2
1Department of Pharmacognosy, College of Pharmacy, University of Baghdad, Baghdad, Iraq2Department of Pharmacology and Toxicology, College of Pharmacy, University of Baghdad, Baghdad, Iraq
Citation: Sakar Dilshad Asaad, Thukaa Z Abdul Jalill, Ammar A Fadhil, The Wound Healing Potential of Iraqi Origanum majorana L n-Hexane and Ethyl Acetate Fractions in a Rat Model of Excisional Wound Repair: Pre-Clinical Test, J Res Med Dent Sci, 2023, 11 (01):237-242
Received: 25-Oct-2022, Manuscript No. JRMDS-22-72951; , Pre QC No. JRMDS-22-72951 (PQ); Editor assigned: 28-Oct-2022, Pre QC No. JRMDS-22-72951 (PQ); Reviewed: 11-Nov-2022, QC No. JRMDS-22-72951; Revised: 26-Dec-2022, Manuscript No. JRMDS-22-72951 (R); Published: 24-Jan-2023