Research - (2021) Volume 9, Issue 11
Vitamin C and E Down Regulates the Expression of C-JNK, IKKB, NF-kB in Adipose Tissue of PCB-Exposed Rats
Dev Arora, Gayathri R*, J Selvaraj, V Vishnu Priya and Kavitha S
*Correspondence: Gayathri R, Department of Biochemistry, Saveetha Institute of Medical and Technical Sciences, Saveetha University, India, Email:
Abstract
Introduction: PCBs have an adverse effect on the environment and animals. They are present in paints, coolants, rubber based products, and plastic based products, transformers, capacitors. PCBs cause endocrine disruption, inhibit reproductive functions, and can increase oxidative stress which may lead to diabetes. Aim: To analyse if Vitamin C and Vitamin E helps in down-regulation of certain markers such as NF-kB, IKKB, c-JNK. Methods: For this study, adult male wistar rats weighing between 180-200g were obtained for further study. Animals were divided into three groups (Control, Test and Treatment group). Total RNA isolation followed by conversion of RNA to cDNA with the help of reverse transcriptase and the mRNA expression level was analysed in RT-PCR. The data was analysed statistically by one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test was used to see the statistical significance among the groups. The results with the p<0.05 level were considered to be statistically significant. Results: Vitamin C and E led to significant decrease in expression of C-JNK, IKKB and NF-kB in Adipose Tissue of PCB exposed rats when compared to control groups. On the mediation of IKKB and NF-kB; PCB induced group of male wistar rats showed significant increase and when treated with Vitamin C and E there was a significant decrease. Conclusion: Vitamin C and E have a prospective antioxidant and antidiabetic potential by regulating the levels of NF-kB, IKKB expression which is involved in the process of oxidative stress and inflammation.
Keywords
PCB, diabetes, NF-kB, IKKB, C-JNK, PCBs, Vitamin C, Vitamin E, Adipose Tissue, Innovative technology, Novel method
Introduction
PCBs or polychlorinated biphenyls are synthetic organic compounds. They are used in electric equipment such as transformers, paints, plastics, rubber products, and coolants. PCBs consist of 209 aromatic chlorinated hydrocarbons. PCBs bioaccumulate in the food chain [1]. PCBs cause endocrine disruption [2] which may lead to diabetes, inhibit reproductive function, decreases sperm motility [3], causes early menarche [4], and alters sex ratio in humans. PCBs mediated cell dysfunction leads to an increase in level of oxidative stress.
The imbalance between free radicals and antioxidants is oxidative stress. It is produced due to diabetic conditions, possibly due to low levels of antioxidants. Oxidative stress activates the C-JNK pathway, which helps in suppression of insulin gene expression i.e. they are involved in diabetes [5].Oxidative stress leads to activation of JNK pathway [6].
C-JNK or C- Jun- N- terminal kinases are a family of kinases that are signalling pathways used in gene expression, cell death, and cellular senescence [7]. C-JNK helps in the regulation of proinflammatory cytokines [8], insulin resistance. Suppression of JNK pathway in diabetic mice regulates insulin resistance.
Vitamin C (ascorbic acid) and Vitamin E (alphatocopherol) are antioxidants. Vitamin E is lipid soluble whereas Vitamin C is water soluble. Vitamin E prevents lipid peroxidation chain reactions. Antioxidant property of Vitamin E can protect cell damage [9]. Vitamin C prevents oxidative damage by interacting with free radicals. These Vitamin C and E supplementation improves oxidative stress associated with aging, work output. Vitamin C is shown to block IL-1 by blocking IKB kinase activation [10].
Vitamin C is required in the diet of humans as a terminal enzyme in many pathways. It is involved in many functions such as iron transport [11]. Recommended diet for Vitamin C is 60mg/day. Vitamin C has both preventive and therapeutic roles in cancer, atherosclerosis, viral infections etc. [12]; it is also capable of inhibiting TNF induction of IL-8.
NF-kB or Nuclear Factor k-B is also called ‘cellular sensor’ for oxidative stress [13, 14]. In the proper NF-Kb activation pathway used by cytokine; activation occurs through IKK signalosomes which have three major components IKKA, IKKB, IKKC [21]. IKKB is an inhibitory protein that dissociates from NF-kB and is an inhibitor of NF-kB kinase. NF-kB depends on expression of proinflammatory factors. Our team has extensive knowledge and research experience that has translate into high quality publications [15-33].
The aim of the study is to find out the role of Vitamin C and E plays in down regulation of certain pathways like C-JNK, IKKB, NF-kB in adipose tissue of PCB exposed rats and also to evaluate the role of Vitamins in oxidative stress and diabetes.
Materials and Methods
Chemicals
All chemicals and reagents used in this study were purchased from Sigma Chemical Company St. Louis, MO, USA; Invitrogen, USA; Eurofins Genomics India Pvt Ltd, Bangalore, India; New England Biolabs (NEB), USA; Promega, USA. PCB was procured from Sigma Chemical Company St. Louis, MO, USA; Total RNA isolation reagent (TRIR) was purchased from Invitrogen, USA. The reversetranscriptase enzyme (MMuLv) was purchased from Genet Bio, South Korea purchased from Promega, USA. Interleukin-1ß and ß-actin primers were purchased from Euro fins Genomics India Pvt Ltd, Bangalore, India and.
Animals
The present experimental study was approved by the institutional animal ethics committee (IAEC no.: BRULAC/SDCH/SIMATS/IAEC/12.2019/048). Adult male Wistar albino rats, weighing 180–200g, were obtained and maintained in clean propylene cages at the Biomedical Research Unit and Laboratory Animal Centre (BRULAC), Saveetha Dental College and Hospitals, Saveetha University, India) in an air-conditioned animal house, fed with standard rat pelleted diet (Lipton India Ltd., Mumbai, India), and clean drinking water was made available ad libitum. Rats were divided into 3 groups, each consisting of 6 animals.
Experimental Design
Experimental Design • Group 1: Control (Vehicle control, rats were intraperitoneally (i.p.) administered with the vehicle (corn oil) for 30 days.
• Group 2: Rats received PCB (PCB was dissolved in corn oil at a dose of 2mg/kg body • Weight (b.wt) intraperitoneally daily at 10:00 a.m. for 30 days.
• Group 3: PCB and vitamin E (dissolved in olive oil at a dose of 50 mg/kg body weight), and vitamin C treated (100 mg/kg body weight dissolved in distilled water daily at 10 AM through gastric intubation for 30 days).
At the end of treatment, animals were anesthetized with sodium thiopental (5 mg/kg, i.p), and 20 ml of normal saline was perfused through the left ventricle, to clear blood from the liver, and other organs. Visceral adipose tissue was dissected out and used for the assay of various parameters.
Gene Expression Analysis by Real Time PCR Analysis
Isolation of total RNA
Total RNA was isolated from control and experimental samples using TRIR (total RNA isolation reagent) kit. Briefly, 100 mg fresh tissue was homogenized with 1 ml TRIR and the homogenate was transferred immediately to a microfuge tube and kept at -80°C for 60 min to permit the complete dissociation of nucleoprotein complexes. Then, 0.2 ml of chloroform was added, vortexed for 1 min and placed on ice at 4°C for 5 min. The homogenates were centrifuged at 12,000 x g for 15 min at 4°C. The aqueous phase was carefully transferred to a fresh microfuge tube and an equal volume of isopropanol was added, vortexed for 15 sec and placed on ice at 4°C for 10 min. The samples were centrifuged at 12,000 x g for 10 min at 4°C. The supernatant was discarded and RNA pellet was washed with 1 ml of 75% ethanol by vortexing and subsequent centrifugation for 5min at 7,500 x g (4°C). The supernatant was removed and RNA pellets were mixed with 50 µl of autoclaved Milli-Q water and dissolved by heating in a water bath for 10 min at 60°C.
Quantification of RNA
Diluted RNA samples were quantified spectrophotometrically by measuring the absorbance (A) at 260/280 nm. 40 µg of RNA in 1 ml gives one absorbance at 260 nm. Therefore, the concentration of RNA in the given sample can be determined by multiplying its A260 by 40 and dilution factor. The purity of RNA preparation can be calculated using the ratio between its absorbance at 260 and 280 nm. A ratio of absorbance at 260/280 nm >1.8 is generally considered as good quality RNA. The purity of RNA obtained was 1.8.
Reverse Transcriptase – Polymerase Chain Reaction (RT – PCR)
RT-PCR is an approach for converting and amplifying a single stranded RNA template to yield abundant double stranded DNA products. 1. First strand reaction: Complementary DNA (cDNA) is made from the mRNA template using Oligo dT, dNTPs & reverse transcriptase. 2. Second strand reaction: After the reverse transcriptase reaction is complete, standard PCR (called the “second strand reaction”) is initiated. Principle RT-PCR is a method used to amplify cDNA copies of RNA. It is the enzymatic conversion of mRNA into a single cDNA template. A specific oligodeoxynucleotide primer hybridizes to the mRNA and is then extended by an RNAdependent DNA polymerase to create a cDNA copy. First strand DNA synthesis The RT kit was purchased from Eurogentec (Seraing, Belgium). Reagents 1. 10X RT buffer: One vial containing 1.4 ml of 10X RT buffer. 2. Euro Script reverse transcriptase: One tube containing 75 µl of Moloney Murine leukaemia virus reverse transcriptase (3750 U at 50 U/µl).
Quantitative Real Time PCR Principle
The purpose of a PCR (Polymerase Chain Reaction) is to make a huge number of copies of a gene. There are three major steps in a PCR, which are as follows: Denaturation at 94°C for 3 min: During the denaturation at 94°C for 2-5 min, the double strand melts open to single stranded DNA, all enzymatic reactions stop. Annealing at 54°C-65°C for 30 sec: Ionic bonds are constantly formed and broken between primer and the single stranded template to ensure the extension process. Extension at 72°C for 30 sec: Primers that are in positions with no exact match get loose again (because of the higher temperature) and don't give an extension of the fragment. The bases (complementary to the template) are coupled to the primer on the 3' side (the polymerase adds dNTP from 5' to 3', reading the template from 3' to 5' side; bases are added complementary to the template). Because both strands are copied during PCR, there is an exponential increase of the number of copies of the gene.
Reagents
• 2X Reaction buffer: The PCR master mix kit was purchased from Takara Bio Inc., Japan. Contains TaKaRa Ex Taq HS (a hot start PCR enzyme) dNTP Mixture, Mg2+, Tli RNase H (a heat-resistant RNase H that minimizes PCR inhibition by residual mRNA), and SYBR Green I.
• Forward primer (10µM).
• Reverse primer (10µM).
• cDNA- Template.
• Autoclaved milli Q water.
• Primers: The following gene specific oligonucleotide primers were used.
Details of primers used in the present study
Rat IKKB
FW- 5’-AGCTCTGGAACCTCCTGAAGA – 3’
RW- 5’–AGCTCCAGTCTAGGGTCGTGA – 3’
Rat c-JNK FW: 5’-AGACGCTTGAGTTGAGAGCC-3’
RW: 5’-CTTCAGTGTGCGGCTTAGGA-3’
Rat-NfkB
FW: 5’-GCTTTGCAAACCTGG GAATA-3’
RW: 5’-CAAGGTCAGAAT GCACCAGA-3’
Rat ß-actin
FW–5’-TACAGCTTCACCACCACAGC - 3’
RW–5’- TCTCCAGGGAGGAAGAGGAT - 3’
Procedures
Procedure Real Time PCR was carried out on CFX 96 Real Time system (Bio-Rad). The reaction mix (10 µl) was prepared by adding 5 µl of 2X reaction buffer, 0.1 µl of sense and antisense primer, 1 µl of cDNA and 3.8 µl of sterile water. The thermal cycler protocol was as follows: Initial denaturation at 95°C for 3 min, followed by 40 cycles of PCR, denaturation at 95°C for 10 sec, annealing at 60°C for 20 sec and extension at 72°C for 20 sec. All reactions were performed in triplicate along with no template control (NTC). Melt curve analysis was performed using the thermal cycling programmed at 50-95°C for each sample to determine the presence of multiple amplicons, non-specific products and contaminants. The results were analysed using CFX 96 Real Time system software (Bio-Rad). As an invariant control, the present study used rat ß-actin.
Statistical analysis
The triplicate analysis results of the experiments performed on control and treated rats were expressed as mean ± SEM. Results were analysed statistically by oneway analysis of variance (ANOVA) and significant differences between the mean values were measured using Duncan's multiple range test using Graph Pad Prism version 5. The results with p<0.05 level were considered to be statistically significant.
Results
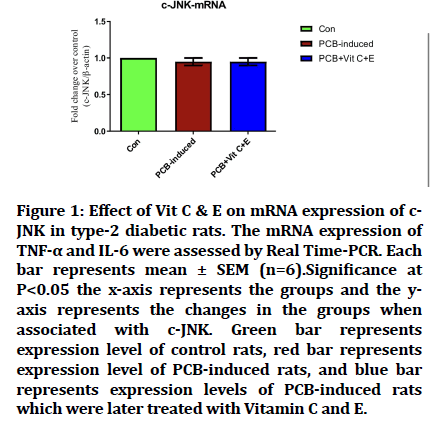
Effect of PCB on cJNK-mRNA
In the relation to mRNA expression of C-JNK, there is no significant change in the level in PCB induced group and no significant change with the group treated with Vitamin C and Vitamin E when compared to the Control group (Figure 1).
Figure 1: Effect of Vit C & E on mRNA expression of c- JNK in type-2 diabetic rats. The mRNA expression of TNF-a and IL-6 were assessed by Real Time-PCR. Each bar represents mean ± SEM (n=6).Significance at P<0.05 the x-axis represents the groups and the yaxis represents the changes in the groups when associated with c-JNK. Green bar represents expression level of control rats, red bar represents expression level of PCB-induced rats, and blue bar represents expression levels of PCB-induced rats which were later treated with Vitamin C and E.
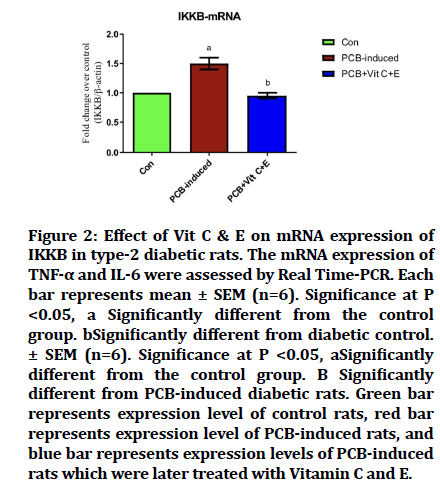
Effect of PCB on IKKB-mRNA
mRNA of IKKB was studied, there was a significant increase in the level in the PCB induced group, but when treated with Vitamin C and Vitamin E, there was a significant decrease in the expression, which became equivalent to the control group (Figure 2).
Figure 2: Effect of Vit C & E on mRNA expression of IKKB in type-2 diabetic rats. The mRNA expression of TNF-a and IL-6 were assessed by Real Time-PCR. Each bar represents mean ± SEM (n=6). Significance at P <0.05, a Significantly different from the control group. bSignificantly different from diabetic control. ± SEM (n=6). Significance at P <0.05, aSignificantly different from the control group. B Significantly different from PCB-induced diabetic rats. Green bar represents expression level of control rats, red bar represents expression level of PCB-induced rats, and blue bar represents expression levels of PCB-induced rats which were later treated with Vitamin C and E.
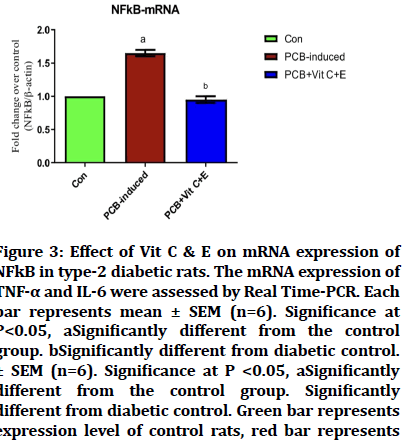
Effect of PCB on NF-kB-mRNA
The mRNA expression of NF-kB was analysed, there was a significant increase in the levels in the PCB induced group, but when treated with Vitamin C and Vitamin E, there was a significant decrease in the level which became equivalent to the control group (Figure 3).
Figure 3: Effect of Vit C & E on mRNA expression of NFkB in type-2 diabetic rats. The mRNA expression of TNF-a and IL-6 were assessed by Real Time-PCR. Each bar represents mean ± SEM (n=6). Significance at P<0.05, aSignificantly different from the control group. bSignificantly different from diabetic control. ± SEM (n=6). Significance at P <0.05, aSignificantly different from the control group. Significantly different from diabetic control. Green bar represents expression level of control rats, red bar represents expression level of PCB-induced rats, and blue bar represents expression levels of PCB-induced rats which were later treated with Vitamin C and E..
On the mediation of IKKB and NF-kB; PCB induced group of male wistar rats showed significant increase and when treated with Vitamin C and E there was a significant decrease.
Discussion
Previous studies have reported that there is an association found between PCB and diabetes [34]. PCBs promote urea formation, rather than glucose synthesis [35]. PCBs also caused liver glycogen islets to disappear [36]. In this study, there was a significant decrease in level of PCB induced group on the expression of mRNA with C-JNK and no change was found when treated with Vitamin C and Vitamin E. These results were in line with the previous report in which there was a significant decrease when Vitamin C was added to the C-JNK pathway from the control group.
In this research, the PCB induced group showed a significant increase in the levels of mRNA expression with IKKB and NF-kB in type 2 diabetic rats but when treated with Vitamin C and Vitamin E, it showed a decrease in the level. In a previous study, Vitamin C significantly decreased IKKB and inhibited NF-kB [37].
In this study, there was a significant decrease in the levels of PCB due to Vitamin C and Vitamin E. The previous studies only showed the correlation with Vitamin C, but in this study Vitamin E also decreased the oxidative stress. In accordance with the previous study [38], reported that PCB exposure to adult rats induces glucose/lipid metabolic disorder via the activation of HNF1b/ROS/NF-kB/IL-1 and IL-6 mediated increase in inflammation in liver and adipose tissue and down regulated the antioxidant enzymes such as glutathione peroxidase. The increased level of ROS activates NF-kB expression which in turn stimulates the proinflammatory cytokines such as IL-6 that leads to the development of metabolic disorders such as type 2 diabetes. Hence, in the present study also PCB induced group increased expression of NF-kB, c-JNK and IKKB -mRNA might be due to the increased levels of ROS as a result of decreased antioxidant enzymes such as SOD, GPX that results in activation of NF-kB, c-JUN and IKKB leads to development of diabetes. However, treatment with Vitamin C and Vitamin E were able to reduce the expression of the same to that of the control level due to its potential antioxidant properties. In supporting these findings, Williams et al (2019) reported that PCB exposed detrimental changes in insulin signalling was reversed by the supplementation of lycopene.
Exposure to PCBs can lead to type-2 diabetes by altering the proinflammatory markers which increases the insulin resistance [39]. Another study [40] stated that supplementation of Vitamin C and E prevented oxidation. SIT also inhibited obesity induced insulin resistance ameliorating the inflammatory events in the adipose tissue through the down regulation of IKKB/NF-kB and c- JNK signalling pathway [41].
Conclusion
According to the study, it is evident that exposure to PCB induces oxidative stress and inflammation which can give rise to various disorders like diabetes, cancer etc. Vitamin C and E could reverse the expression of inflammatory signalling molecules in PCB-induced rats. Hence, Vitamin C and E can be considered as potential drugs for the management of diabetes.
Acknowledgement
The authors would like to express their gratitude to Saveetha Dental College & Hospitals for supporting and for successful completion of this project.
Conflict of Interest
All the authors declare that there was no conflict of interest in the present study.
Author Contribution
Dev Arora: Contributed in designing the study, execution of the project, statistical analysis, manuscript drafting.
Gayathri R: Contributed in study design, guiding the research work, manuscript correction
J. Selvaraj, V Vishnu priya, Kavitha S: Study design, statistical analysis, manuscript proofreading and correction.
Funding
The present study was supported by the following agencies:
• Saveetha Dental College and Hospitals.
• Saveetha Institute of Medical and Technical Science.
• Saveetha University.
• Arora Multispecialty Dental Hospital and Implant Centre.
References
- Ivanciuc T, Ivanciuc O, Klein DJ. Modeling the bioconcentration factors and bioaccumulation factors of polychlorinated biphenyls with posetic quantitative super-structure/activity relationships (QSSAR). Mol Diver. 2006; 10:133-45.
- McGovern V. PCBs are endocrine disruptors: Mixture affects reproductive development in female mice. Environ Health Perspect 2006; 114:A368–A369.
- Meeker JD, Hauser R. Exposure to polychlorinated biphenyls (PCBs) and male reproduction. Syst Biol Reprod Med 2010; 56:122-31.
- Denham M, Schell LM, Deane G, et al. Relationship of lead, mercury, mirex, dichlorodiphenyldichloroethylene, hexachlorobenzene, and polychlorinated biphenyls to timing of menarche among Akwesasne Mohawk girls. Pediatrics. 2005;115:e127–34.
- Kaneto H, Kawamori D, Nakatani Y, et al. Oxidative stress and the JNK pathway as a potential therapeutic target for diabetes. Drug News Perspect 2004; 17:447–53.
- Kaneto H, Matsuoka TA, Nakatani Y, et al. Oxidative stress, ER stress, and the JNK pathway in type 2 diabetes. J Mol Med 2005; 83:429-39.
- Yarza R, Vela S, Solas M, et al. c-Jun N-terminal kinase (JNK) signaling as a therapeutic target for Alzheimer’s disease. Frontiers in Pharmacol 2016; 6:321.
- Lee HM, Kim KS, Kim J. A comparative study of the effects of inhibitory cytokines on human natural killer cells and the mechanistic features of transforming growth factor-beta. Cell Immunol. 2014 ; 290:52–61.
- Hennig B, Hammock BD, Slim R, et al. PCB-induced oxidative stress in endothelial cells: modulation by nutrients. Int J Hyg Environ Health 2002; 205:95–102.
- Bowie AG, O’Neill LA. Vitamin C inhibits NF-kappa B activation by TNF via the activation of p38 mitogen-activated protein kinase. J Immunol 2000; 165:7180–8.
- Hallberg L, Brune M. Is there a physiological role of vitamin C in iron absorption?. Ann NY Acad Sci 1987; 498:324-32.
- Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad Sci U S A. 1971; 68:2678–81.
- Wu F, Zhu J, Li G, et al. Biologically synthesized green gold nanoparticles from Siberian ginseng induce growth-inhibitory effect on melanoma cells (B16). Artif Cells Nanomed Biotechnol 2019; 47:3297–305.
- Chen F, Tang Y, Sun Y, et al. 6-shogaol, a active constiuents of ginger prevents UVB radiation mediated inflammation and oxidative stress through modulating NrF2 signaling in human epidermal keratinocytes (HaCaT cells). J Photochem Photobiol Biol 2019; 197:111518.
- Li Z, Veeraraghavan VP, Mohan SK, et al. Apoptotic induction and anti-metastatic activity of eugenol encapsulated chitosan nanopolymer on rat glioma C6 cells via alleviating the MMP signaling pathway. J Photochem Photobiol Biol 2020; 203:111773.
- Babu S, Jayaraman S. An update on ß-sitosterol: A potential herbal nutraceutical for diabetic management. Biomed Pharmacother 2020; 131:110702.
- Malaikolundhan H, Mookkan G, Krishnamoorthi G, et al. Anticarcinogenic effect of gold nanoparticles synthesized from Albizia lebbeck on HCT-116 colon cancer cell lines. Artif Cells Nanomed Biotechnol 2020; 48:1206–13.
- Han X, Jiang X, Guo L,et al. Anticarcinogenic potential of gold nanoparticles synthesized from Trichosanthes kirilowii in colon cancer cells through the induction of apoptotic pathway. Artif Cells Nanomed Biotechnol 2019; 47:3577–3584.
- Gothai S, Muniandy K, Gnanaraj C, et al. Pharmacological insights into antioxidants against colorectal cancer: A detailed review of the possible mechanisms. Biomed Pharmacother 2018; 107:1514–22.
- Veeraraghavan VP, Hussain S, Papayya Balakrishna J, et al. A comprehensive and critical review on ethnopharmacological importance of desert truffles: Terfezia claveryi, Terfezia boudieri, and Tirmania nivea. Food Rev Int 2021; 1-20.
- C Tobon-Velasco J, Cuevas E, A Torres-Ramos M. Receptor for AGEs (RAGE) as mediator of NF-kB pathway activation in neuroinflammation and oxidative stress. CNS Neurol Disord Drug Targets 2014; 13:1615-626.
- Sathya S, Ragul V, Veeraraghavan VP, et al. An in vitro study on hexavalent chromium [Cr(VI)] remediation using iron oxide nanoparticles based beads. Environ. Nanotechnol Monit Manag 2020; 14:100333.
- Yang Z, Pu M, Dong X, et al. Piperine loaded zinc oxide nanocomposite inhibits the PI3K/AKT/mTOR signaling pathway via attenuating the development of gastric carcinoma: In vitroandin vivostudies. Arab J Chem 2020; 13:5501–16.
- Rajendran P, Alzahrani AM, Rengarajan T, et al. Consumption of reused vegetable oil intensifies BRCA1 mutations. Crit Rev Food Sci Nutr 2020; 1–8.
- Barma MD, Muthupandiyan I, Samuel SR, et al. Inhibition of Streptococcus mutans, antioxidant property and cytotoxicity of novel nano-zinc oxide varnish. Arch Oral Biol 2021; 126:105132.
- Samuel SR. Can 5-year-olds sensibly self-report the impact of developmental enamel defects on their quality of life? Int J Paediatr Dent 2021; 31:285–6.
- Samuel SR, Kuduruthullah S, Khair AMB, et al. Dental pain, parental SARS-CoV-2 fear and distress on quality of life of 2 to 6 year-old children during COVID-19. Int J Paediatr Dent 2021; 31:436–41.
- Bigham A, Salehi AO, Rafienia M, et al. Zn-substituted Mg2SiO4 nanoparticles-incorporated PCL-silk fibroin composite scaffold: A multifunctional platform towards bone tissue regeneration. Mater Sci Eng 2021; 112242.
- Yin Z, Yang Y, Guo T, et al. Potential chemotherapeutic effect of betalain against human non-small cell lung cancer through PI3K/Akt/mTOR signaling pathway. Environ Toxicol 2021; 36:1011–20.
- Veeraraghavan VP, Periadurai ND, Karunakaran T, et al. Green synthesis of silver nanoparticles from aqueous extract of Scutellaria barbata and coating on the cotton fabric for antimicrobial applications and wound healing activity in fibroblast cells (L929). Saudi J Biol Sci 2021; 28:3633–3640.
- Mickymaray S, Alfaiz FA, Paramasivam A, et al. Rhaponticin suppresses osteosarcoma through the inhibition of PI3K-Akt-mTOR pathway. Saudi J Biol Sci 2021; 28:3641–3649.
- Teja KV, Ramesh S. Is a filled lateral canal–A sign of superiority?. J Dent Sci 2020; 15:562.
- Kadanakuppe S, Hiremath S. Social and behavioural factors associated with dental caries experience among adolescent school children in Bengaluru City, India. Br J Med Med Res 2016; 14:1–10.
- Silverstone AE, Rosenbaum PF, Weinstock RS, et al. Polychlorinated biphenyl (PCB) exposure and diabetes: results from the Anniston Community Health Survey. Environ Health Perspect 2012; 120:727–32.
- Ebner KV, Bergen WG, Braselton Jr WE. Nitrogen deficits in aroclor 1254-treated rats. Toxicol. Sci. 1987 ;8:89-96.
- Thomé JP, Roelandt L, Goffmet G, et al. Cytotoxic effects of aroclor 1254 on ultrastructure and biochemical parameters in cultured foetal rat hepatocytes. Toxicology 1995; 98:83-94.
- Cárcamo JM, Pedraza A, Bórquez-Ojeda O, et al. Vitamin C is a kinase inhibitor: Dehydroascorbic acid inhibits I?Ba kinase ß. Mol Cell Biol 2004; 24:6645-52.
- Wu H, Yu W, Meng F, et al. Polychlorinated biphenyls-153 induces metabolic dysfunction through activation of ROS/NF-?B signaling via downregulation of HNF1b. Redox Biol 2017; 12:300–310.
- Weinhold B. PCBs and diabetes: Pinning down mechanisms. Environ Health Perspect 2013; 121:A32.
- Rajesh P, Sathish S, Srinivasan C, et al. Phthalate is associated with insulin resistance in adipose tissue of male rat: Role of antioxidant vitamins. J Cell Biochem 2013; 114:558–569.
- Jayaraman S, Devarajan N, Rajagopal P, et al. ß-Sitosterol circumvents obesity induced inflammation and insulin resistance by down-regulating IKKß/NF-?B and JNK signaling pathway in adipocytes of type 2 diabetic rats. Molecules 2021; 26:2101
Author Info
Dev Arora, Gayathri R*, J Selvaraj, V Vishnu Priya and Kavitha S
Department of Biochemistry, Saveetha Institute of Medical and Technical Sciences, Saveetha University, IndiaCitation: Dev Arora1, Gayathri R, J Selvaraj, V Vishnu Priya, Kavitha S, Vitamin C and E Down Regulates the Expression of C-JNK, IKKB, NF-kB in Adipose Tissue of PCB-Exposed Rats, J Res Med Dent Sci, 2021, 9(11): 39-44
Received: 09-Sep-2021 Accepted: 22-Oct-2021