Original Article - (2019) Volume 7, Issue 2
Comparison of Candida albicans Adhesion and Colonization on All-metal and Metal-ceramic Crowns
Farimah Sardari1* and Fazeh Khosravi2
*Correspondence: Farimah Sardari, Oral Medicine Department, Dental School, Rafsanjan University of Medical Sciences, Iran, Email:
Abstract
Introduction: Candidiasis is responsible for 85% of fungal infections of the oral cavity in humans. There is little data available on the adhesion of C. albicans to a variety of crowns. This study aimed to investigate the adhesion and colonization of C. albicans to two conventional crowns.
Materials and Methods: In this in-vitro study, first 36 circular disks of two crowns were created: all-metal and metalceramic (18 for each crown). The disks were transferred to plates containing the suspensions 1 × 105 cfu/ml of C. albicans colonies. After 40, 80, and 120 minutes, the disks were rinsed in sterile normal saline and shaken at low speed to separate yeast cells. This suspension was cultured on Sabouraud dextrose agar culture medium with pour plate method and was then counted. Differences were considered significant when pË0.05, using computer software SPSS version 18 for windows.
Results: In all-metal and metal-ceramic samples, adhesion and colonization of C. albicans significantly increased during time (pË0.001). The lowest adhesion and colonization of C. albicans was observed in metal-ceramic crowns after 40 minutes, and the highest adhesion and colonization of C. albicans in metal-ceramic crowns after 120 minutes.
Conclusion: The results showed that adhesion of C. albicans to all-metal crowns was significantly less than metal-ceramic crowns after 80 minutes and 120 minutes.
Keywords
Candida albicans, Crown, All-metal, Metal-ceramic, In-vitro
Introduction
Several microorganisms may exist in the oral cavity, some of which may cause severe diseases. Oral candidiasis, the most common human fungal infection, is one of the important infections of the oral cavity, caused by Candida [1,2]. Among different species of Candida, the most common (more than 80%) of oral infections are caused by three species: C. albicans, C. glabrata, and C. tropicalis [3]. The prevalence of C. albicans in mouth is reported to range from 3% to 48% with a higher prevalence in healthy children and young adults [4].
C. albicans invades the epithelium and specific glycoproteins, and co-agglutination reactions can have a higher affinity for epithelial cell adhesion [5]. The infection is controlled in healthy people by antimicrobial proteins in the saliva and oral microflora [6]. The use of dentures, drug use, hormonal factors, radiation therapy, diabetes mellitus, acquired immunodeficiency syndrome, and salivary disorder like Sjogren’s syndromes can predispose the patients to C. albicans [1,4,6].
C. albicans can be colonized in different surfaces of the oral cavity, including the tongue, palate, dental plaques, and restorations [7]. Different restorations have been suggested to cover the teeth’s lost crown; including allmetal, all-ceramic, and porcelain-fused-to-metal crown(s) (PFMs) [8]. As to the evidence, feldspathic/silica-based crowns had significantly lower survival, especially in posterior region [9], and comparison of different crowns showed the highest survival rates in PFMs (94.4%), densely sintered zirconia PFMs (90.4%), reinforced glass ceramic PFMs (89.1%), and glass infiltrated alumina PFMs (86.2%), respectively [10]. The main aspects of restorations, addressed by researchers, include the aesthetic aspects, survival, and their stability. Although these aspects are also very important, the chance of candidiasis infection is another important factor in superiority of different crowns. As an important factor in colonization of C. albicans, studies have investigated the adherence of this fungus to different restorative materials, including resin-composite [11], acrylic resin [12,13], and glass ionomer [14].
The fungal and bacterial community of dental plaque is subjected to physiological and compositional shifts as a result of environmental stresses generated by the placement of dental restoration and this is could lead to serious complications that result in the failure of the restoration [15,16]. The results of this study could provide the initial in-vitro results on colonization of C. albicans to different restorative materials. Because of none have compared the adherence between different crowns, especially two conventional restorations, including all-metal and PFMs. Health of support structures is as important as failure of a restoration, even more important. Therefore, this study aimed to investigate the adhesion and colonization of C. albicans to two conventional crowns.
Materials and Methods
Study design
This present in-vitro study is done in the immunology of infectious diseases research center, Rafsanjan University of Medical sciences, Rafsanjan, Iran from January to May 2015. Two types of all-metal and PFMs were prepared in circular-shaped (Dibateb Co., Tehran, Iran) discs with 5 mm diameter, 1 mm thickness (Figure 1) [17] and used to experiment the formation of biofilms. C. albicans was cultured on Sabouraud-dextrose agar medium (HiMedia, Netherlands) and incubated at 30°C for two days for preparation of new fungal colonies; then one colony of the mentioned fungus was placed in 100 cc of sterile Sabouraud-dextrose broth medium (liquid culture medium, HiMedia, Netherlands) and incubated on a shaker (Behdad, Tehran, Iran). Then a suspension of 1 × 105 cfu/ml (colony forming unit per ml) colony of C. albicans was counted. A total of 36 discs of two crown types (18 of each) were prepared as test specimens (Dibateb Co., Tehran, Iran), and three groups of six discs were placed in a plate containing Candida suspension for 40, 80, and 120 minutes separately; they were dipped in this suspension at the mentioned times and placed at 37°C.

Figure 1: The circular-shape of two different disks (ceramic and metal crown)
After the necessary time (40, 80 and 120 minutes), the specimens were removed and after a simple three-second washed with sterile physiological serum, they were placed in 1 ml sterile physiological serum (Razi Vaccine and Serum Research Institute, Karaj, Iran) and uniformly shaken for one minute with low circulation of vertex device (Behdad, Tehran, Iran) to allow the probable yeast cells adhering to the specimens be detached and suspended in the physiologic serum (Razi Vaccine and Serum Research Institute, Karaj, Iran). Subsequently, a certain amount (0.01 ml) of this suspension was cultured on the culture medium of Sabouraud-dextrose agar (HiMedia, Netherlands) with pour plate method [13]. The plates were placed in a 37°C-incubator for 48 hours. After the incubation time, the number of colonies was counted which was representative of the adhesion of C. albicans to the test specimens (Figure 2).
Figure 2: Colony count of C. albicans base of pure pale method
By multiplying the number of colonies counted in dilution amount, the final number of candida colonies per each disk, within a 1 ml sterile physiologic serum (Razi Vaccine and Serum Research Institute, Karaj, Iran), was calculated at 40, 80 and 120 minutes as cfu/ml. It should be noted that each colony is equivalent to one living cell. In the end, the results were recorded for each group at three times in the relevant checklist and given to the analyst, who was unaware of the groups.
Statistical analyses
The collected data was input into the statistical software IBM SPSS Statistics for Windows version 18.0 (IBM Corp. 2009. Armonk, NY: IBM Corp) used for analysis. Results of numeric variable were presented as mean ± standard deviation (SD) and that of nominal variables by frequency (percentage) for categorical variables. Onesample Kolmogorov-Smirnov test showed that the data ere normally distributed in the two groups (p>0.05). Equality of variances was also confirmed by the Levene’s test (p=0.180). Comparison of colony count in each group among the three time intervals was tested by one-way repeated measures ANOVA. For all statistical tests, pvalues of 0.05 or less were considered statistically significant.
Results
The mean colony counts of C. albicans on all-metal and PFMs crowns at three intervals are shown in Table 1. As the results of two-way ANOVA test showed, the effect of type of crown was statistically significant on the colony counts and regardless of the time, mean colony count of C. albicans had significant differences in the vicinity of two crown types (df=1, F=685.120, p˂0.001). Also, the effect of time was significant and regardless of the crown type, mean colony count of C. albicans had significant differences in the measured intervals and increased with time (df=2, F=3808.908, p˂0.001) (Figure 3).
| Group | 40 minutes | 80 minutes | 120 minutes | p-value* | |
|---|---|---|---|---|---|
| All-metal | Min-max | 41-47 | 54-61 | 70-76 | <0.001 |
| Mean ± SD | 43.72 ± 1.99 | 57.28 ± 2.22 | 72.94 ± 1.89 | ||
| Metal-ceramic | Min-max | 24-32 | 72-81 | 104-117 | <0.001 |
| Mean ± SD | 28.11 ± 2.37 | 76.44 ± 3.07 | 110.44 ± 4.09 | ||
| p-value** | <0.001 | <0.001 | <0.001 | - | |
*Comparison between the three time intervals by one-way ANOVA; **Comparison between the two groups by independent two-sample t test
Table 1: Comparison of mean colony count of C. albicans in three time intervals between two groups of all-metal and metal-ceramic crown
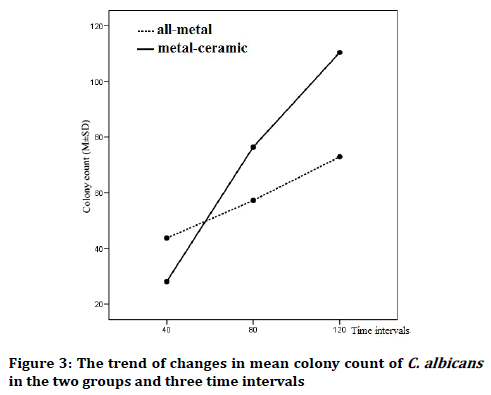
Figure 3: The trend of changes in mean colony count of C. albicans in the two groups and three time intervals
Time and crown type had a mutual effect and the slope of changes in mean colony count of C. albicans were significantly different in the measured intervals in two crown types (df=2, F=887.392, p˂0.001). Although colony count of C. albicans after 40 minutes was higher in all-metal crowns compared to PFMs, it significantly increased in PFMs in the next two intervals (80 and 120 minutes) (Table 1).
Discussion
The results of the present in-vitro study on PFM crown and all-metal crown showed statistically significant differences in mean colony count of C. albicans in different time intervals. The maximum time was considered 2 hours in this study (120 minutes) and the results of mean colony count of C. albicans were compared between the groups of crown types and among the three time intervals (40, 80, and 120 minutes). The lower growth of C. albicans over time in all-metal crowns than of PFMs could be due to the better ability of polishing in metal, compared to ceramic; apparently, the metal bubbles are easily observed and can be treated by the clinician [18]. The higher colonization of C. albicans in the first 40 minutes in all-metal crowns could also be due to the ion release in the first phase from metal [19]. Nevertheless, as far as the authors are concerned, no study has compared the fungal growth between these two crowns, for their results to be comparable to ours.
As the results indicated, the crown type and time had both significant increasing effects on mean colony count of C. albicans and also mutual effects with each other. The mean colony count of C. albicans was higher in all-metal crowns after 40 minutes while the mean colony count of C. albicans significantly increased in PFMs after 80 and 120 minutes. These results indicated that PFMs had a higher fungal growth compared to all-metal crowns after two hours.
Several studies have reported the increased attachment of C. albicans to artificial materials, such as dental prostheses, catheters, and restorative materials, suggested to be due to the biofilm formation of candida and better surface adherence [20-22]. Burgers et al. [23] studied the adherence of C. albicans in composite resin materials (compomer, ormocer, novel silorane, and conventional hybrid composites) and showed less adhesion of fungi in compomers and ormocers. Lawaf et al. [14] showed high adherence of C. albicans strains to glass ionomer fragments, especially in the absence of human whole saliva. Although, the results of these studies confirm the general results of the present study on higher adherence of C. albicans to restorative materials [11], the results are not comparable with regard to types of restorative materials.
According to the general opinion, the adhesion of C. albicans to Herkulin (resin restorative materials) is reduced by whole saliva and secretory immunoglobulin A [11,14,24,25]. Also, other materials in saliva have been suggested to detach C. albicans from the cell wall in plastic plates [26]. These results emphasize on the management of C. albicans in patients with conditions reducing the saliva flow or pH [27], such as acquired immunodeficiency syndrome, and salivary disorder like Sjogren’s syndromes [6]. As to the evidence, diabetic patients may have altered saliva flow, therefore, paying attention to C. albicans in these patients and including the clinical assessment and educating patients on appropriate care are valuable [28]. The role of saliva in reducing colonization of C. albicans can have treatment targets, as Candida strains have high resistance to different antibiotics [29,30].
Generally, there is little information about the colonization and adherence of C. albicans to different restorative materials. In the present study, the colonization of C. albicans to two conventionally used crowns were compared to provide valuable information on the choice of the most appropriate crown material; nevertheless, this study may have also some limitations. The most important limitation of this study could be the fact that the presence of saliva has not been evaluated. As to the evidence, the presence of saliva significantly reduced the colonization and adhesion of C. albicans to restorative materials [11,14,24]. Meanwhile, the crown materials in the present study has not been used and investigated in other studies [11,14,24] and further research is required to illuminate different aspects of C. albicans colonization in all-metal and PFMs.
Conclusion
Base of our survey, the adhesion of C. albicans to all-metal crowns was significantly less than metal-ceramic crowns after 80 minutes and 120 minutes. Further studies are required to evaluate the colonization of C. albicans to allmetal and PFMs in the presence of whole human saliva.
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this article.
References
- Akpan A, Morgan R. Oral candidiasis. Postgrad Med J 2002; 78:455-9.
- Farah CS, Ashman RB, Challacombe SJ. Oral candidosis. Clin Dermatol 2000; 18:553-62.
- Qi Q, Hu T, Zhou X. Frequency, species and molecular characterization of oral Candida in hosts of different age in China. Oral Pathol Med 2005; 34:352-6.
- Farah CS, Lynch N, McCullough MJ. Oral fungal infections: An update for the general practitioner. Aust Dnt J 2010; 55:48-54.
- Mayer FL, Wilson D, Hube B. Candida albicans pathogenicity mechanisms. Virulence 2013; 4:119-28.
- Khan M, Iqubal MA, Shukla AK, et al. Oral candidiasis-A review. Int J Health Sci Res 2014; 4:240-5.
- Akdeniz BG, Koparal E, Sen BH, et al. Prevalence of Candida albicans in oral cavities and root canals of children. ASDC J Dent Chil 2002; 69:289-92.
- Creugers NH, Mentink AG, Fokkinga WA, et al. 5-year follow-up of a prospective clinical study on various types of core restorations. Int J Prosthodont 2005; 18:34-9.
- Sailer I, Makarov NA, Thoma DS, et al. All-ceramic or PFMs tooth-supported fixed dental prostheses (FDPs)? A systematic review of the survival and complication rates. Part I: Single crowns (SCs). Dent Mater 2015; 31:603-23.
- Pjetursson BE, Sailer I, Makarov NA, et al. All-ceramic or metal-ceramic tooth-supported fixed dental prostheses (FDPs)? A systematic review of the survival and complication rates. Part II: Multiple-unit FDPs. Dent Mater 2015; 31:624-39.
- Maza JL, Elguezabal N, Prado C, et al. Candida albicans adherence to resin-composite restorative dental material: Influence of whole human saliva. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2002; 94:589-92.
- Moura JS, da Silva WJ, Pereira T, et al. Influence of acrylic resin polymerization methods and saliva on the adherence of four Candida species. J Prosth Dent 2006; 96:205-11.
- Sadeghpour SM, Falahati M, Ashrafi KM, et al. The comparison of the bayer acryl and acropars acryl effect on the adhesion of Candida albicans. Razi J Med Sci 2011; 18:20-6.
- Lawaf S, Azizi A. Candida albicans adherence to glass ionomer restorative dental material. J Dental Res Den Clin Dent Prospects 2009; 3:52-5.
- Takahashi N, Nyvad B. The role of bacteria in the caries process: Ecological perspectives. J Dent Res 2011; 90:294-303.
- Kilian M, Chapple IL, Hannig M, et al. The oral microbiome-An update for oral healthcare professionals. Br Dent J 2016; 21:657-66.
- Mikulewicz M, Chojnacka KW. Trace metal release from orthodontic appliances by In vivo studies: A systematic literature review. Biol Trace Elem Res 2010; 137:127-38.
- Kokubo Y, Ohkubo C, Tsumita M, et al. Clinical marginal and internal gaps of procera allceram crowns. J Oral Rehabil 2005; 32:526-30.
- Ronsani MM, Mores RAU, Meira TM, et al. Virulence modulation of Candida albicans biofilms by metal ions commonly released from orthodontic devices. Microb Pathog 2011; 51:421-5.
- Busscher HJ, Rinastiti M, Siswomihardjo W, et al. Biofilm formation on dental restorative and implant materials. J Dent Res 2010; 89:657-65.
- Qilo M, Bakken V. Biofilm and dental biomaterials. Materials (Basel) 2015; 8:2887-900.
- Seneviratne CJ, Jin L, Samaranayake LP. Biofilm lifestyle of candida: A mini review. Oral Dis 2008; 14:582-90.
- Burgers R, Schneider-Brachert W, Rosentritt M, et al. Candida albicans adhesion to composite resin materials. Clin Oral Invest 2009; 13:293-9.
- Lyon JP, de Resende MA. Correlation between adhesion, enzyme production, and susceptibility to fluconazole in Candida albicans obtained from denture wearers. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2006; 102:632-8.
- Elguezabal N, Maza JL, Ponton J. Inhibition of adherence of Candida albicans and Candida dubliniensis to a resin composite restorative dental material by salivary secretory IgA and monoclonal antibodies. Oral Dis 2004; 10:81-6.
- Kamagata-Kiyoura Y, Abe S, Yamaguchi H, et al. Detachment activity of human saliva in vitro for Candida albicans cells attached to a plastic plate. J Infect Chemother 2003; 9:215-20.
- Torres SR, Peixoto CB, Caldas DM, et al. Relationship between salivary flow rates and Candida counts in subjects with xerostomia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2002; 93:149-54.
- Mohammadi F, Javaheri MR, Nekoeian S, et al. Identification of Candida species in the oral cavity of diabetic patients. Curr Med Mycol 2016; 2:1-7.
- Cleveland AA, Harrison LH, Farley MM, et al. Declining incidence of candidemia and the shifting epidemiology of Candida resistance in two US metropolitan areas, 2008-2013: Results from population-based surveillance. PloS One 2015; 10:e0120452.
- Sanglard D, Odds FC. Resistance of Candida species to antifungal agents: Molecular mechanisms and clinical consequences. Lancet Infect Dis 2002; 2:73-85.
Author Info
Farimah Sardari1* and Fazeh Khosravi2
1Oral Medicine Department, Dental School, Rafsanjan University of Medical Sciences, Rafsanjan, Iran2Dentist, Private Dental Clinic, Rafsanjan, Iran
Citation: Farimah Sardari, Fazeh Khosravi, Comparison of Candida albicans adhesion and colonization on all-metal and metalceramic crowns , J Res Med Dent Sci, 2019, 7(2): 93-97
Received: 26-Nov-2018 Accepted: 20-Mar-2019
