Research - (2020) Volume 8, Issue 1
Efficacy of Intra Periodontal Pocket Application with Combined Chlorhexidine and Hydrogen Peroxide on the Bleeding on Probing and Anaerobic Bacterial Viable Count
Ahmed Kareem1*, Basima Ghafory1 and Abbas S Al-Mizraqchi2
*Correspondence: Ahmed Kareem, Department of Periodontics, College of Dentistry, University of Baghdad, Iraq, Email:
Abstract
Introduction: Periodontal pocket is a pathologically deepening of gingival sulcus along with subgingival contamination of hard and soft tissues. Scaling and root planning is an effective treatment in addition to supra gingival plaque managing. Many studies show that the adjunctive use of sub gingival antimicrobial agents with scaling and root planning resulted in improvement clinically and microbiologically.
Aim: To determine if scaling and root planning alone or in combination with 0.2% chlorhexidine and 3% hydrogen peroxide gel (Glucosite gel) have a significant effect on total viable count of anaerobic bacteria and bleeding on probing. Materials and methods: A total 136 periodontal pockets of 20 patients of both sexes aged from 35 to 50 years which have at least two periodontal pockets ranged from 5-6 mm on each side participated in this study, treated with split mouth technique. One side treated via scaling and root planning only, while the other side combined with glucosite gel. Bleeding on probing and total viable anaerobic count was recorded at day zero and after 4 weeks. With exclusion patients who were pregnant, smokers, had systemic diseases or who on orthodontic or periodontal treatment. SPSS for windows was used for data analysis.
Results: Glucosite gel adjunctive to scaling and root planning reduced significantly bleeding on probing and subgingival viable anaerobic bacteria in comparison to scaling and root planning alone.
Conclusion: Applications of glucosite gel subgingivally in combination with scaling and root planning have additional effects on clinical and microbiological periodontal outcomes.
Keywords
Impacted mandibular third molars, Surgical difficulty, Postoperative inflammatory response
Introduction
The primary treatment of chronic periodontitis involving supra and sub gingival plaques removing along with scaling and root planning (SRP) to reducing a load of periodontal pathogens [1], this local treatment has a considerable limitations, as that impossible to eradicate whole subgingival bacteria from an infected sites mechanically [2] and modification of subgingival microorganisms occur after scaling and root planning [3] and viable microorganisms which survive after SRP re-colonize, resulting in new formation of biofilm [4]. So that, an antimicrobial agents have been developed as an adjunct to SRP to improve mechanical instrumentation effectiveness and prevent plaque regeneration which lead to minimizing clinical features of periodontal disease [5,6].
Chlorhexidine (CHX) is a potent anti-bacterial, anti-fungal and anti-viral agent which used broadly [5]. According to its concentration, it can be bacteriostatic or bactericidal. The CHX substantively is the main important special feature [7]. Hydrogen peroxide (H2O2) is an oxygenating material has a wide range of antimicrobial activity [8] along with action of disturbing biofilm via intensifying the oxygen saturation subgingivally [9], accordingly, effect on pro inflammatory cytokines fabrication within periodontal pockets. [10]. Oosterwaal, et al. concluded that the CHX gel can be diminished microbial load inside periodontal pockets by 99% during thirty minutes following application [11]. Paolantonio, et al. found that the SRP accompanied with CHX gel ending in a significant improvement in outcomes clinically [12]. On the other hand, some studies [13,14] showed that using localized anti-microbial releasing systems look to be advantageous more than SRP alone in managing periodontal pockets. In many studies [15-21], hydrogen peroxide and chlorhexidine were used separately and conflicting outcomes were found, therefore we have carried out this study to assess the effectiveness of glucosite gel which contains a combination of chlorhexidine 0.2% and hydrogen peroxide 3% as an adjunct to SRP, and to compare these treatment modalities with the results obtained by SRP alone.
AIM
To provide additional data on the clinical and microbiological effect of combined (CHX and H2O2) gel when used as adjunct to SRP.
Materials and Methods
Glucosite gel from Cerkamed medical company containing chlorhexidine 0.2% and hydrogen peroxide 3%. The activity of the glucosite gel was assessed microbiologically by agar well diffusion method in comparison with chlorhexidine and hydrogen peroxide separately and larger inhibition zones for glucosite were recorded (Figures 1 and 2) [22].
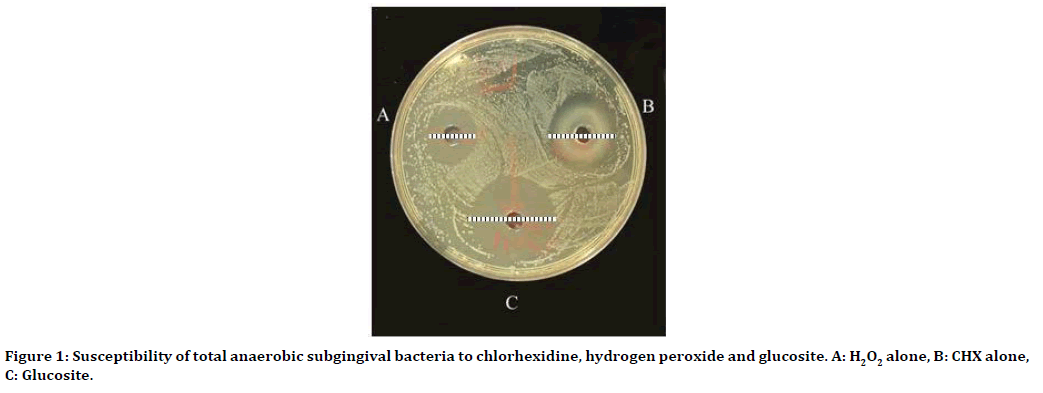
Figure 1. Susceptibility of total anaerobic subgingival bacteria to chlorhexidine, hydrogen peroxide and glucosite. A: H2O2 alone, B: CHX alone, C: Glucosite.

Figure 2. Glucosite gel.
Subject selection
Twenty male and female subjects of 35-50 years old, from the department of Periodontology, in the specialized dental center in Missan, enrolled voluntarily and well explained about the aim and purposes of the study and given informed consent to participate in it, each patient treated with split mouth technique, the left side which included 68 periodontal pockets treated with SRP combined with glucosite gel application. While the right side which also included 68 periodontal pockets treated with SRP only. The method of split mouth had a benefit of variables removing among subjects. They were subjected to a questionnaire, including medical, dental and social history, followed by full examinations of clinical periodontal parameters of inflammation:
Plaque index (PLI) and bleeding on probing (BOP).
Exclusion criteria
→ Patients that undertaken periodontal treating in three months just preceding the study, as well as patients has appliances of orthodontic.
→ Patients suffering from systemic diseases, which may have an impact on the periodontium.
→ Uncooperative patients who failed to preserve proper oral hygiene.
→ Current pregnancy or lactation.
→ Patients on antibiotics and/or antiinflammatory drugs during the last 3 months.
→ Patients allergic to CHX and/or H2O2.
→ Smokers as well as alcohol drinkers.
→ Teeth with fixed crowns or bridges and sites with furcation involvement.
A. Sequence of the study subject/ ite selection
Before baseline examination, full mouth supragingival scaling with ultrasonic scalar was done, motivation and oral hygiene instructions were given, concerning instruction in brushing and the use of appropriate interdental cleaning aids until the oral hygiene status reached to mean PLI less than (0.8). All selected sites with periodontal pocket depth 5 to 6 mm.
B. Clinical periodontal parameters recordings
1. Assessment of (PLI): Löe 1964 [23].
2. Assessment of (BOP) [24].
C. Subgingival biofilm sampling
Isolation of treated sites with sterile cotton rolls and removing supragingival plaque using sterile curette, and dried with air. Sampling undertaken by sterile paper point (size F1 dents ply millefer) which pushed into deepest part of pocket for 15 seconds carefully to avoid mechanical injury (Figure 3) [12].
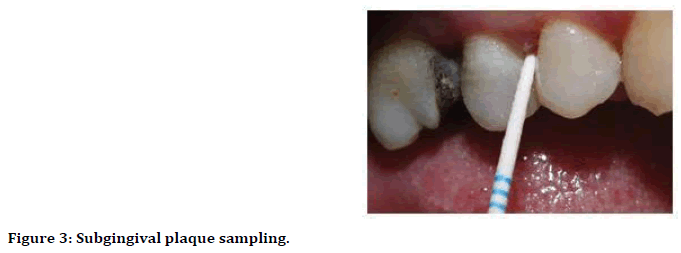
Figure 3. Subgingival plaque sampling.
D. Microbiological procedure
The plaque sample (paper point) immersed in glass universal tubes containing 3 mm glass beads and 10 ml of phosphate buffer saline and vortexed by vortex mixer for 2-3 minutes. Afterward, serial dilution steps were prepared using sterile phosphate buffer saline; 0.1 ml was withdrawn from each dilution (10-3, 10-5) and spread into the petri dishes that contained brain heart infusion agar using sterile microbiological spreader on the plates. Followed by incubation anaerobically with gas pack supplied in an anaerobic jar for 48 hrs at 37°C (Figure 4)[22].

Figure 4. Incubator for incubation anaerobic bacteria with gas pack in an anaerobic jar.
E. Intrapplication of glucosite
After SRP performed under local anesthesia for the selected sites, glucosite gel applied gently subgingivally in left periodontal pockets until flowed out from the gingival margin, three times during ten minutes [25].
Statistical Analysis
Data were analyzed using SPSS software version 23 release in 2019. Shapiro-Wilk test was used to check the normality of the data. The statistics used were descriptive along with inferential (p<0.05) considered significant in the statistical evaluation.
Results
Table 1 illustrates the mean percentage of bleeding on probing at zero time and after one month and statistical differences between them for each side. The mean percentage at baseline was (77.985), (73.48) while after one month became (47.975), (22.995) for right and left sides respectively and the results were found to be of highly significant difference for both sides (P<0.01).
| Variable | Side | No. of sites | Visit | Percentage (BOP) | z test | p- value |
|---|---|---|---|---|---|---|
| BOP % | Right (SRP) | 68 | Baseline | 77.985 | 1.739 | 0.005 (HS) |
| After one month | 47.975 | |||||
| Left (SRP + glucosite) | 68 | Baseline | 73.48 | 2.214 | 0.000 (HS) | |
| After one month | 22.995 | |||||
| HS=highly significant at P<0.01 | ||||||
Table 1: Statistical difference of bleeding on probing among visits in both sides.
Table 2 show the statistical difference of bleeding on probing among right side (SRP) and left side (SRP with glucosite) after one month of treatment. The means percentage were (47.975), (22.995) for right and left sides respectively and the results were found to be of significant difference (p<0.05).
| Variable | no. of sites | Sides | percentage | z test | p value |
|---|---|---|---|---|---|
| BOP % | 136 | Right | 47.975 | 1.423 | 0.035 (S) |
| Left | 22.995 |
Table 2: The statistical difference of bleeding on probing among sides after one month treatment.
Table 3 demonstrate mean, median, SD, minimum and maximum of anaerobic bacterial viable count (CFU\ml) X 103 for right side (SRP) and statistical difference between visits. The mean at baseline was (25.16) while it became (11.65) after one month of treatment and the results were found to be of highly significant difference (p<0.01).
| Median | Mean | ± S.D. | Min. | Max. | t-test | p-value | |
|---|---|---|---|---|---|---|---|
| Baseline | 22.838 | 25.16 | 9.45 | 14.92 | 49.36 | 5.644 | 0.000 (HS) |
| After one month | 10.838 | 11.65 | 5 | 4.27 | 26.29 | ||
| HS=highly significant at P<0.01 | |||||||
Table 3: Descriptive analysis and intra group comparison of anaerobic bacterial viable count (CFU\ml) × 103 for right side (SRP).
Table 4 demonstrate mean, median, SD, minimum and maximum of anaerobic bacterial viable count (CFU\ml)×103 for left side (SRP with glucosite) and statistical difference between visits. The mean at baseline was (23.715) while it became (1.495) after one month of treatment and the results were found to be of highly significant difference (p<0.01).
| Median | Mean | ± S.D. | Min. | Max. | t-test | p-value | |
|---|---|---|---|---|---|---|---|
| Baseline | 21.19 | 23.715 | 8.445 | 13.45 | 47.27 | 11.222 | 0.000 (HS) |
| After one month | 0.1425 | 1.495 | 2.661 | 0.095 | 8.605 | ||
| HS=highly significant at P<0.01 | |||||||
Table 4: Descriptive analysis and intra group comparison of anaerobic bacterial viable count (CFU\ml) ×103 for left side (SRP combined with glucosite).
Table 5 illustrates intergroup comparison of anaerobic bacterial viable count (CFU\ml) X 103 between right and left sides after one month of treatment. The means were (11.65), (1.495) for right and left sides respectively and the results were found to be of highly significant difference (p<0.01).
| Variable | t-test | Df | p-value |
|---|---|---|---|
| CFU anaerobic | 4.804 | 38 | 0.000 (HS) |
| HS=highly significant at P<0.01 | |||
Table 5: Intergroup comparison of anaerobic bacterial viable count (CFU\ml) × 103 between right and left sides after one month of treatment.
Discussion
Local delivery agents offer the advantages of high concentrations at the target site with reduced dosage, fewer applications, and high patient acceptability. Thus, adjunctive use of local drug delivery may provide a beneficial response, especially in specific areas where conventional forms of therapy might fail [26].
The result of the present study showed that the clinical parameter (BOP) in addition to microbial results were improved significantly on day 30 as compared to zero day and a positive therapeutic role was achieved for both sides in the split mouth study. This decreasing in BOP scores in sites treated with combined SRP and glucosite, also its obvious in intergroup and intragroup levels and this comes in agreement with many studies where they study the effect of either CHX alone or H2O2 alone. These studies including Heasman et al., Cosyn et al., Sahebjam et al., Gottumukkala et al., Deas et al., Lecic 2016. [21,27-31].
The different in protocol and formulation of CHX might explain the variation with Paolantonio, et al. who reported that no significant effect of adjunctive use of CHX treatment with SRP [20]. The inter comparison between both sides in the split mouth study revealed that there were significant differences in microbiological outcomes after one month of treatment. This finding matched well with Rosling, et al. Nandini, et al. [32,33]. While disagree with Wennstrom, et al. who reported that irrigation with chlorhexidine or hydrogen peroxide did not induce any changes in total viable counts in periodontal pockets. This difference in results could be caused by protocol of study, intervals and measurements [17].
Since the high viscosity of a gel, one would expect a lower clearance of the active ingredient from periodontal pockets, thereby further promoting pharmacotherapeutic effects.
The antimicrobial effects of hydrogen peroxide (H2O2) have been studied both in vitro and in vivo and resulted that it released oxygen which has some bactericidal [16,34] at low concentrations, which has long been used as an oral debriding agent and wound cleanser, also has an extensive history of topical application in mouth rinses and antiseptic gels, with long-term safety record[35] besides altering the biofilm that result in reduced host inflammatory response [10]. Chlorhexidine (CHX) it is a broad-spectrum bactericidal agent. Its propensity to bind to the surface of tissues offers a long-lasting antimicrobial effect causing reduction in pellicle formation, alteration of bacterial adherence to teeth [36]. Shahriari, et al. confirmed the synergistic effect between chlorhexidine and hydrogen peroxide [37].
Conclusion
The results of the study indicated that SRP with glucosite or SRP alone improved clinical and microbiological outcomes. Glucosite gel as an adjunct to scaling and root planning significantly reduced bleeding on probing and total viable anaerobic bacteria when compared with scaling and root planning alone.
References
- Cugini M, Haffajee AD, Smith C, et al. The effect of scaling and root planning on the clinical and microbiological parameters of periodontal diseases: 12‐month results. J Clin Periodontol 2000; 27:30-36.
- Brayer WK, Mellonig JT, Dunlap RM, et al. Scaling and root planning effectiveness: The effect of root surface access and operator experience. J Periodontol 1989; 60:67-72.
- Mousquès T, Listgarten MA, Phillips RW. Effect of scaling and root planning on the composition of the human subgingival microbial flora. J Periodontal Res 1980; 15:144-151.
- Resposo S. Differences between biofilm growth before and after periodontal therapy. J Dent Res 2008; 87:473-480.
- Rusu D, Stratul SI, Sarbu C, et al. Evaluation of a hydrophobic gel adhering to the gingiva in comparison with a standard water‐soluble 1% chlorhexidine gel after scaling and root planning in patients with moderate chronic periodontitis. A randomized clinical trial. Int J Dent Hyg 2017; 15:53-64.
- Aziz MA, Shukri M, Ghafory B. Clinical effectiveness of subgingival application of 25% metronidazole gel as a monotherapy and as adjunctive treatment of chronic periodontitis. MDJ 2012; 9:245-251.
- Balagopal S, Arjunkumar R. Chlorhexidine: The gold standard antiplaque agent. J Pharm Sci Res 2013; 5:270-274.
- Rashed HT. Evaluation of the effect of hydrogen peroxide as a mouthwash in comparison with chlorhexidine in chronic periodontitis patients: A clinical study. J Int Soc Prev Community Dent 2016; 6:206-212.
- Lu M, Hansen EN. Hydrogen peroxide wound irrigation in orthopaedic surgery. J Bone Jt Infect 2017; 2:3-9.
- Keller D, Buechel M. Periodontal treatment with direct medication delivery of hydrogen peroxide and oxygen. Oral Health Case Rep 2017; 3:1-8.
- Oosterwaal P, Mikx FH, vant Hof MA, et al. Short‐term bactericidal activity of chlorhexidine gel, stannous fluoride gel and amine fluoride gel tested in periodontal pockets. J Clin Periodontol 1991; 18:97-100.
- Paolantonio M, D'Ercole S, Pilloni A, et al. Clinical, microbiologic, and biochemical effects of subgingival administration of a Xanthan‐based chlorhexidine gel in the treatment of periodontitis: A randomized multicenter trial. J Periodontol 2009; 80:1479-1492.
- Radvar M, Pourtaghi N, Kinane D. Comparison of 3 periodontal local antibiotic therapies in persistent periodontal pockets. J Periodontol 1996; 67:860-865.
- Kinane D, Radvar M. A six‐month comparison of three periodontal local antimicrobial therapies in persistent periodontal pockets. J Periodontol 1999; 70:1-7.
- Wennström J, Lindhe J. Effect of hydrogen peroxide on developing plaque and gingivitis in man. J Clin Periodontol 1979; 6:115-130.
- Amigoni NA, Johnson GK, Kalkwarf KL. The use of sodium bicarbonate and hydrogen peroxide in periodontal therapy: A review. J Am Dent Assoc 1987; 114:217-221.
- Wennstrom JL, Dahlén G, Gröndahl K, et al. Periodic subgingival antimicrobial irrigation of periodontal pockets II. Microbiological and radiographical observations. J Clin Periodontol 1987; 14:573-580.
- Al-Saeed MY, Babay N. The use of povidone–iodine and hydrogen peroxide mixture as an adjunct to non-surgical treatment of slight to moderate chronic periodontitis. Saudi Dent J 2009; 21:127-133.
- Mohammed M, Abdel–Nasser M, Usama M. Evaluation of topical application of CHLO-SITE (chlorhexidine gel) in management of chronic periodontitis. Suez Canbal Univ J 2008; 11:35-40.
- Paolantonio M, D'Angelo M, Grassi RF, et al. Clinical and microbiologic effects of subgingival controlled‐release delivery of chlorhexidine chip in the treatment of periodontitis: A multicenter study. J Periodontol 2008; 79:271-282.
- Lecic J, Cakic S, Janjic Pavlovic O, et al. Different methods for subgingival application of chlorhexidine in the treatment of patients with chronic periodontitis. Acta Odontol Scand 2016; 74:502-507.
- Al-Mizrakchi A. Adherence of mutans Streptococci on teeth surfaces: Microbiological and biochemical studies. PhD Thesis, Al-Mustansiriya University, Baghdad 1998.
- Silness J, Löe H, Periodontal disease in pregnancy II. Correlation between oral hygiene and periodontal condition. Acta Odontol Scand 1964; 22:121-135.
- Newman MG, Takei HH, Carranza FA. Carranza's clinical periodontology. 12th Edn Philadelphia: WB Saunders Co. 2002.
- Cosyn J, Sabzevar MM. A systematic review on the effects of subgingival chlorhexidine gel administration in the treatment of chronic periodontitis. J Periodontol 2005; 76:1805-1813.
- Greenstein G, Tonetti M. The role of controlled drug delivery for periodontitis. The research, science and therapy committee of the American academy of periodontology. J Periodontol 2000; 71:125-140.
- Heasman P, Heasman L, Stacey F, et al. Local delivery of chlorhexidine gluconate (PerioChipTM) in periodontal maintenance patients. J Clin Periodontol 2001; 28:90-95.
- Cosyn J, Wyn I, De Rouck T, et al. Subgingival chlorhexidine varnish administration as an adjunct to same‐day full‐mouth root planning. I. Clinical observations. J Periodontol 2007; 78:430-437.
- Sahebjam Atabaki M. Clinical Effect of Periodontal Pocket Irrigation with H2O2. Avicenna J Dent Res 2011; 3:53-59.
- Gottumukkala SN, Koneru S, Mandalpu N, et al. Effectiveness of sub gingival irrigation of an indigenous 1% curcumin solution on clinical and microbiological parameters in chronic periodontitis patients: A pilot randomized clinical trial. Contemporary Clin Dent 2013; 4:186-193.
- Deas DE, Moritz AJ, Sagun RS, et al. Scaling and root planning vs. conservative surgery in the treatment of chronic periodontitis. Periodontol 2016; 71:128-39.
- Rosling BG, Slots J, Webber RL, et al. Microbiological and clinical effects of topical subgingival antimicrobial treatment on human periodontal disease. J Clin Periodontol 1983; 10:487-514.
- Nandini N, Vidya D, Komal A. comparative evaluation of 1% curcumin solution and 0.2% chlorhexidine irrigation as an adjunct to scaling and root planning in management of chronic periodontitis: A clinico-microbiological study. J Pharm Biomed Sci 2012; 14:1-10.
- De Renzis FA. Endotoxin-inactivating potency of hydrogen peroxide: Effect on cell growth. J Dent Res 1981; 60:933-935.
- Marshall MV, Cancro LP, Fischman SL. Hydrogen peroxide: A review of its use in dentistry. J Periodontol 1995; 66:786-796.
- Badiea RA. Efficacy of intra-pocket application of two antimicrobial agents as an adjunct to mechanotherapy of chronic periodontitis: A comparative study. Tikrit J Dent Sci 2014; 3:83-93.
- Shahriari S, Mohammadi Z, Mokhtari MM, et al. Effect of hydrogen peroxide on the antibacterial substantivity of chlorhexidine. Int J Dent 2010; 2010:1-4.
Author Info
Ahmed Kareem1*, Basima Ghafory1 and Abbas S Al-Mizraqchi2
1Department of Periodontics, College of Dentistry, University of Baghdad, Iraq2Department of Microbiology, College of Dentistry, University of Baghdad, Iraq
Citation: Ahmed Kareem, Basima Ghafory, Abbas S Al-Mizraqchi, Efficacy of Intra Periodontal Pocket Application with Combined Chlorhexidine and Hydrogen Peroxide on the Bleeding on Probing and Anaerobic Bacterial Viable Count, J Res Med Dent Sci, 2020, 8(1): 125-131.
Received: 04-Jan-2020 Accepted: 28-Jan-2020
