Research - (2021) Volume 9, Issue 6
Role of Immunohistochemistry Versus Hematoxylin and Eosin and Special Stains in Helicobacter Pylori Detection in Gastric Lesions
Ayushi Narain and Ponnuswamy Karkuzhali*
*Correspondence: Ponnuswamy Karkuzhali, Department of Pathology, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, India, Email:
Abstract
Helicobacter pylori can be demonstrated in tissue by Gram stain, Giemsa stain, haematoxylin & eosin stain, Warthin-Starry silver stain, acridine orange stain, and phase-contrast microscopy. Hence the present study was premeditated to analyse the role of Immunohistochemistry versus Hematoxylin and Eosin and special stains in detection of Helicobacter pylori in gastric lesions. A total of 455 gastric biopsy specimens were received over a period of 2 years from October 2015 to September 2017. Of these 100 cases were selected which had adequate data. 60 cases were randomly selected and analysed for Helicobacter pylori infection using H&E, Giemsa and IHC. The most associated endoscopic finding with gastritis in the present study was erosions followed by erythema (linear bands/erythematous mucosa). Sensitivity and specificity of Giemsa was 95.2 % and 100% respectively. Sensitivity and specificity of H&E was found to be 90.5 % and 100% respectively. Best results were obtained by Immunohistochemistry, especially when the density of organ ism was low. The cost, applicability and authenticity of the Giemsa stain make it an ideal stain in detecting Helicobacter pylori infection in gastric biopsies.
Keywords
IHC, Giemsa, H&E, Helicobacter pylori, Ulcer, Biopsy
Introduction
Helicobacter pylori, previously called as Campylobacter Pylori, a gram negative, micro aerophilic bacterium is found usually in the stomach. At least half the world's population 1s infected by the bacterium, making it the most widespread infection in the world [1]. Helicobacter pylori is a spiral campylobacter like bacteria, identified in 1982 by Australian scientists Barry Marshall and Robin Warren. Helicobacter pylori's helix shape is thought to have evolved to penetrate the mucoid lining of the stomach [2]. People infected at an early age are likely to develop more intense inflammation that may be followed by atrophic gastritis with a higher subsequent risk of gastric ulcer, gastric cancer, or both. It is accepted to be a major cause of chronic antral gastritis and there is evidence of involvement of this microbe in the etiology of duodenal ulcer and gastric carcinoma. Chronic gastritis is common problem worldwide. Previously many etiologic agents have been implicated to cause chronic gastritis such as smoking, NSAIDs, spicy foods and socioeconomic status [3]. But now it has been proved that in addition to the above Helicobacter pylori is the most common cause [4].
Hence, it is important to determine the presence of the organism in surgical pathology specimens and management of these two common diseases of the upper gastrointestinal tract. Antral biopsy specimens processed for histology provide an easier and cost-effective alternative means of diagnosing Helicobacter pylori infection. A topographic study of H. pylori density and distribution and the comparison of biopsy sites for the histopathological diagnosis of H. pylori conclude that two antral biopsy specimens, one from lesser and other from greater curvature have close to 100% sensitivity for detecting H. pylori infection.
Different special stains have also been used to detect Helicobacter pylori in these histological sections, but their specificity and sensitivity vary greatly. The haematoxylin and eosin stain are the most frequently used stain in histology. Modified Giemsa stain has been preferred because of its ease to perform and availability in most laboratories. All these stains depend on the morphology of the bacterium for identification.
Immunohistochemical techniques have been developed and the use of anti-Helicobacter pylori antibody which reacts with somatic antigens of the bacteria have been found to correlate with the presence of the bacteria. The aim of this study was to determine the reliability of modified Giemsa stain in comparison with Immunohistochemical technique in diagnosing Helicobacter pylori.
Materials and Methods
Source of data
This prospective study was carried out m the Department of Pathology, Sree Balaji Medical College and Hospital, with the help of Department of Medical Gastroenterology, Sree Balaji Medical College and Hospital, during October 2015 to September 2017. A total of 455antral gastric biopsies were received using Olympus CV- 170 endoscope and out of those 100 biopsy samples with adequate data were selected. Of these, random samples of 60 antral biopsies were taken for this study.
Inclusion criteria
All cases of gastritis detected by histopathology irrespective of age were included for study.
Exclusion criteria
Those with poor clinical data were excluded from the study.
Method of data collection
Out of the 455 cases, 100 cases had adequate clinical data. Of these 100 cases, 60 cases were selected at random. Those biopsy materials were processed, and sections were cut at 5 microns. Hematoxylin and eosin staining of sections was done. Histopathological examination of these sections were done. Section from gastric biopsy had been categorized using Sydney grading system based on activity, chronic inflammation, intestinal metaplasia, atrophy, Helicobacter pylori colonisation and the results were tabulated. Special stain (Giemsa stain) and Immunohistochemical study using In situ Rabbit polyclonal antibody for Helicobacter pylori was done on 60 cases and degree of antibody expression was scored in each case. Positive control was taken as, a stomach infected with H. pylori.
Staining procedures-Hematoxylin & eosin section- Paraffin procedure
• Wax removal-10-15 minutes.
• Xylene 1 and Xylene 2 for 10 minutes each.
• Put in acetone-10 dips each.
• Washed in water for 2 minutes.
• Harris hematoxylin is put and left for 8-10minutes.
• Wash in tap water for 2 minutes.
• Dipped in 1% acid alcohol-1 dip.
• Then wash under running water for 5-8 minutes.
• Dipped in Eosin-2 dips.
• Wash with tap water.
• Let it dry for 2-3 minutes.
• Mount with DPX (Di styrene, plasticizer, and xylene).
Result: Nucleus-stained blue colour. Connective tissue and cytoplasm-stained pink colour.
Giemsa staining technique
• Dewax in Xylol 1 and Xylol 2-10 minutes each.
• Two dips in acetone
• Wash under running tap water.
• 1 drop of Giemsa, 9 parts water (1:10 dilution) is placed on the section.
• Wait for 20 minutes.
• Wash with tap water
• Dehydrate, clear in xylene and mount in DPX.
Result: Microorganism-Dark blue; Background-pink pale blue.
Immunohistochemistry
Methods of tissue preparation for IHC
Materials
• Microtome.
• Xylene.
• Ethanol.
• Hydrogen peroxide.
• Distilled water.
• Microwave or pressure cooker.
• Counterstain: Hematoxylin.
• Tri sodium citrate.
• Tris’s buffer.
• Sodium chloride.
• Primary antibody.
• Secondary antibody.
• DAB.
Buffers
Tri-sodium citrate-1.21gms, Distilled water-1000ml; pH-6.0
Tris buffer-0.0605gms, Sodium chloride- 8gms, Distilled water- 1000ml; pH-7.0.
Procedure
Preparation of the paraffin slides
Identify the appropriate tissue block by confirming with the corresponding H & E.
stained slide.
Paraffin sections were cut at 5 micrometre and mounted on polylysine coated slides.
Slides were dipped into xylene (three times) for 5 minutes each to remove the paraffin.
Tissues were re-hydrated by dipping the slides into absolute ethanol two times each for 5 minutes.
Antigen retrieval pressure cooker
• Prepare trisodium citrate buffer-1000ml, pH-6.0.
• Trisodium citrate buffer was poured into the pressure cooker with slide carrier.
• Sections were left in the Tris buffer for about 7- 8 minutes or 2 whistles.
• Slides were then washed m tap water for 3- 5minutes followed by distilled water.
• Endogenous peroxidase was quenched by dipping slides into a fresh aqueous solution of 3% peroxide for 15 minutes.
• Slides were then washed m tap water for 3-5 minutes followed by distilled water.
• Then slides were rinsed with Tris buffer for each 5 minutes, three changes.
Detection of antigens in paraffin sections
• The primary antibody was added at a dilution selected usually for half an hour to 1 hour.
• Sections were soaked in Tris buffer for 10 minutes (2 x 5 minute washes).
• Secondary antibody was then added and left for half an hour.
• Sections were again soaked in Tris buffer for 10 minutes (2 x 5 minute washes).
• A solution of chromogen 3,3'- diamino benzidine (DAB) at 1mg /ml was made in Tris and added.
• Washed in tap water.
Counterstaining hematoxylin
• Slides were dipped in a solution of hematoxylin which was diluted at 1:1 in distilled water and stained for one minute to produce an exceptionally light nuclear counterstain.
• Then it was washed for 1 minute in distilled water
• Dehydrated by dipping in 95% ethanol for 1 minute, 100% ethanol for 1 minute.
• Then washed in xylene 3 times.
• Slides were then mounted and viewed under microscope.
Statistical analysis
The data was statistically analysed using Microsoft Excel 2016 and IBM SPSS ver.23. The significance of the results was assessed by determining the probability factor 'pvalue' using Pearson Chi Square test.
P</=0.05: Significant.
P<0.01: Highly significant.
P>0.05: Not significant.
Results
Out of 100 patients who presented with gastritis, 47% were in the age group of 20-40 years and gastritis was found to be more common after 20 years. M:F ratio was 1.77: 1. The p value was 0.405 which was statistically not significant (Table 1).
| Age | Male | Female | Total |
|---|---|---|---|
| 0-20 years | 01 (1.5%) | 00 (0.00%) | 01 (1%) |
| 21-40 Years | 30 (46.9%) | 17 (47.2%) | 47 (47%) |
| 41-60 Years | 24 (37.5%) | 17 (47.2%) | 41 (41%) |
| >60 | 09 (14%) | 02 (5.5%) | 11 (11%) |
| Total | 64 (64%) | 36 (36%) | 100 |
Table 1: Age and sex distribution of 100 patients with gastritis.
Out of 100 cases 93% were found to be of lower socioeconomic status. The socioeconomic status was assessed using Modified Kuppuswamy scale (proposed updating for Jan' 17). The scores were given on the basis of factors such as education, occupation, monthly income (Table 2).
| Socioeconomic status | Number of patients |
|---|---|
| Lower (Score <5, Class V) | 15 |
| Upper lower (Score=5-10, Class IV) | 78 |
| Lower middle (Score=11-15, class III) | 4 |
| Upper middle (Score=16-2, clss II) | 3 |
| Upper (Score=26-29, Class I) | 0 |
| Total | 100 |
Table 2: Socioeconomic status of 100 patients with gastritis.
The most common symptom that the patients with gastritis exhibited in the present study was found out to be abdominal pain with majority (38%), presenting it (Table 3).
| S. No | Clinical symptoms | No. of patients |
|---|---|---|
| 1 | Abdominal pain | 38 |
| 2 | Loss of appetite | 20 |
| 3 | Vomiting | 16 |
| 4 | Abdominal bloating | 12 |
| 5 | Loss of weight | 10 |
| 6 | Severe anaemia | 4 |
| Total | 100 | |
Table 3: Clinical symptoms of 100 patients associated with gastritis.
Out of 100 cases the most common endoscopic finding (41%) related to gastritis were erosions. Out of the randomly selected 60 patients, 38 cases which were positive for H.pylori by H&E showed antral erosions as the most common endoscopic finding (Table 4).
| S. No | Endoscopic findings | No. of cases |
|---|---|---|
| 1 | Erosions (Antral/Corpal/Fundus) | 41 |
| Flat | 21 | |
| Raised/Nodular | 14 | |
| Hemorrhagic | 6 | |
| 2 | Erythem/Redness (Diffuse or Patchy) | 13 |
| 3 | Mucosal edema | 12 |
| 4 | Gastric ulcer | 10 |
| 5 | Mucus on gatric mucosa | 10 |
| 6 | Prominenr gastric folds | 10 |
| 7 | Prepy;oriculcaer/Duodenal ulcer | 4 |
| Total | 100 | |
Table 4: Endoscopic findings amongst 100 patients with gastritis.
Out of 100 cases of gastritis, 34 cases were smokers, 39 were alcoholics and 42 were tobacco chewers. The P value obtained by Chi Square test for smoking, alcohol, and tobacco was 0.009, 0.006, and 0.025, respectively. It was also observed that prolonged duration of smoking and alcohol increases the risk of H. pylori associated gastritis (Table 5). Table 6 illustrates Sydney scoring ingastric biopsy of 100 cases presented with symptoms. Out of 60 cases studied with IHC for H. pylori, 42 cases were positive and 18 were negative (Table 7).
| Duration | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1-5 years | 6-10 Years | 11-15 years | >15 Years | Present | Absent | |||||||
| Risk factor | M | F | M | F | M | F | M | F | M | F | M | F |
| Smoking | 10 | 1 | 8 | 0 | 10 | 0 | 5 | 0 | 33 | 1 | 31 | 35 |
| Alcohol | 10 | 2 | 12 | 1 | 8 | 0 | 6 | 0 | 36 | 3 | 28 | 33 |
| Tobacco | 6 | 8 | 8 | 4 | 8 | 2 | 6 | 0 | 28 | 14 | 36 | 22 |
Table 5: Risk factors such as smoking, intake of alcohol, tobacco in 100 patients with gastritis.
| Sydney score | Activity | Chronic inflammation | Intestinal metaplasia | Atrophy | H. Pylori |
|---|---|---|---|---|---|
| 1 | 75 | 58 | 7 | 24 | 36 |
| 2 | 13 | 35 | 3 | 0 | 24 |
| 3 | 0 | 7 | 0 | 0 | 6 |
Table 6: Sydney scoring in gastric biopsy of 100 cases presented with symptoms.
| No. of Cases | Helicobacter pylori +ve | Helicobacter pylori-ve |
|---|---|---|
| 60 | 42 (70%) | 18 (30%) |
Table 7: Immunohistochemistry results of 60 cases of antral biopsy for Helicobacter pylori.
Out of the 60 cases, Helicobacter pylori was positive in 42 cases (70%) of which 9 were female 33 were male. Helicobacter pylori was negative in 18 cases (Table 8). Out of 42 males, 33(78.5%) were Helicobacter pylori positive and out of 18 females, 09(50%) were Helicobacter pylori positive. Male: female ratio was 2.3:1 (Table 9). The most common age group affected was in between 31- 40 years out of the 60 cases in whom Helicobacter pylori was detected using Immunohistochemical technique (Table 10).
| Method | H & E | Giesma | IHC | |||
|---|---|---|---|---|---|---|
| Positive | Negative | Positive | Negative | Positive | Negative | |
| Male | 30 | 12 | 32 | 10 | 33 | 9 |
| Female | 8 | 10 | 8 | 10 | 9 | 9 |
| Total number of cases (%) | 38 (63.3) | 22 (36.7) | 40 (66.7) | 20 (33.3) | 42 (70) | 18 (30) |
Table 8: Male and female distribution of positive & negative cases of Helicobacter pylori in H&E, Giemsa, IHC.
| Sex | No. of patients | H. Pylori (-ve) | H. Pylori (+ve) |
|---|---|---|---|
| Male | 42 (70%) | 9 (21%) | 33 (78.5%) |
| Female | 18 (30%) | 9 (50%) | 9 (50%) |
| Total number of cases | 60 (100%) | 18 (30%) | 42 (70%) |
Table 9: Male: Female ratio of Helicobacter pylori infection.
| Age | Total number of cases | Negative | Positive |
|---|---|---|---|
| 10-20 Years | 1 (1.7%) | 0 (0%) | 1 (1.7%) |
| 21-30 Years | 12 (20%) | 2 (3.3%) | 10 (16.7%) |
| 31-40 Years | 17 928.3%) | 4 (6.7%) | 13 (21.7%) |
| 41-50 Years | 12 (20% | 4 (6.7%) | 8 (13.3%) |
| 51-60 Years | 11 (18.3%) | 5 (83%) | 6 (10%) |
| 61-70 Years | 3 (5%) | 1 (1.7%) | 2 (3.3%) |
| >70 Years | 4 (67%) | 2 (3.3%) | 2 (3.3%) |
| Total | 60 (100%) | 18 (30%) | 42 (70%) |
Table 10: Age distribution of Helicobacter pylori infection in 60 cases.
Table 11 shows the Grading of Helicobacter pylori infection in gastric biopsy using Sydney scoring system in various staining methods like H&E, Giemsa, IHC. From the table 12 it was concluded that Helicobacter pylori infection was most common in the age group of 3 l-40yrs. Out of 60 cases, IHC detected Helicobacter pylori in 42 cases with a positivity of 70%. 2 cases which were negative by Giemsa stain were found to be positive on using IHC. 18 cases were negative for both Giemsa and IHC. The p value performing the chi square test was 0.000 which was statistically significant (Table 13). Sensitivity- 95 .2%, Specificity- 100%, Positive predictive value- 100% Negative predictive value- 90%, % of false negative- 4.8, % of false positive-0. Out of 60 cases, IHC detected Helicobacter pylori in 42 cases, whereas H&E detected Helicobacter pylori in 38 cases only, in 18 cases both H&E and IHC were negative (Table 14). The p valueobtained by performing the chi square test was 0.00, which was statistically significant. Sensitivity-90.5%, Specificity-100%, Positive predictive value- 100%, Negative predictive value- 81.8%, % of false negative-9.5%, % of false positive–0. Results are further detailed in figures (Figure 1 to Figure 16).
| Staining method | Total number of cases | H. Pylori positive | Grade I | Grade II | Grade III | H. Pylori Negative |
|---|---|---|---|---|---|---|
| H & E | 60 | 38 (63%) | 22 (36.7%) | 14 (23%) | 2 (3%) | 22 (36.7%) |
| Giemsa | 60 | 40 (66.7%) | 23 (38%) | 1 (25%) | 2 (3%) | 20 (33%) |
| IHC | 60 | 42 (70%) | 2 (41.7%) | 1 (2%) | 2 (3%) | 18 (30%) |
Table 11: Grading of Helicobacter pylori infection in gastric biopsy using Sydney scoring system in various staining methods like H&E, Giemsa, IHC.
| Grade | <20 Years | 21-30 Years | 31-40 Years | 41-0 Years | 51-60Years | 61-70 Years | >70 Years | Total number of cases | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M | F | M | F | M | F | M | F | M | F | M | F | M | F | ||
| Grade 1 | 0 | 0 | 5 | 2 | 8 | 2 | 3 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 25 |
| Grade 2 | 0 | 0 | 2 | 1 | 2 | 1 | 3 | 0 | 4 | 0 | 1 | 0 | 1 | 0 | 15 |
| Grade 3 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Total | 1 | 0 | 7 | 3 | 10 | 3 | 7 | 1 | 5 | 1 | 1 | 1 | 2 | 0 | 42 |
Table 12: Age and sex distribution among Helicobacter pylori positive cases by immunohistochemistry.
| Total number of cases: 60 | ||
|---|---|---|
| Positive | Negative | |
| Giemsa | 40 | 20 |
| IHC | 42 | 18 |
Table 13: Comparison between IHC and Giemsa.
| Total number of cases: 60 | ||
|---|---|---|
| Positive | Negative | |
| H & E | 38 | 22 |
| IHC | 42 | 18 |
Table 14: Comparison of H&E and IHC.
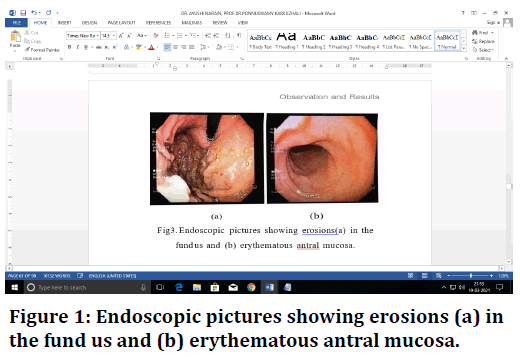
Figure 1. Endoscopic pictures showing erosions (a) in the fund us and (b) erythematous antral mucosa.

Figure 2. H &E (40x), showing inflammatory infiltrate in the lamina propria.
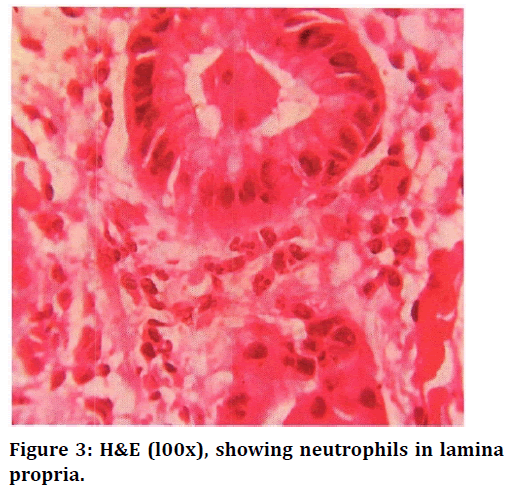
Figure 3. H&E (l00x), showing neutrophils in lamina propria.

Figure 4. H & E (40x), showing inflammatory cells forming lymphoid aggregate.
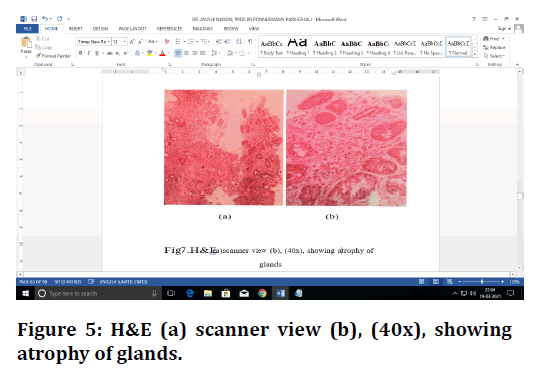
Figure 5. H&E (a) scanner view (b), (40x), showing atrophy of glands.
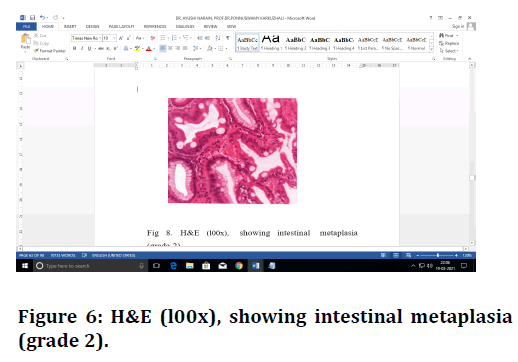
Figure 6. H&E (l00x), showing intestinal metaplasia (grade 2).

Figure 7. H & E (l00x), showing H. pylori in luminal surface.
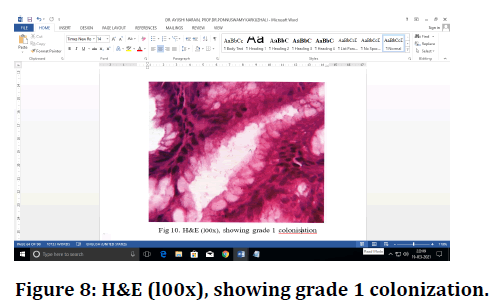
Figure 8. H&E (l00x), showing grade 1 colonization.

Figure 9. H&E (l00x), showing grade 2 colonization H. Pylori.

Figure 10. H & E (l00x), showing grade 3 colonization.
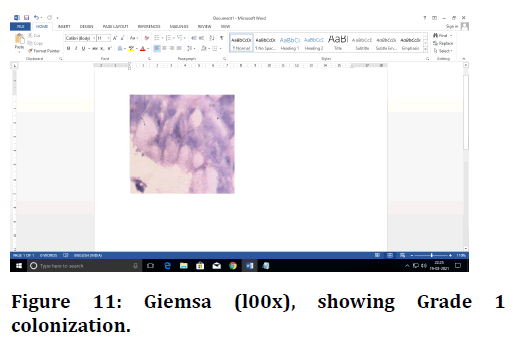
Figure 11. Giemsa (l00x), showing Grade 1 colonization.
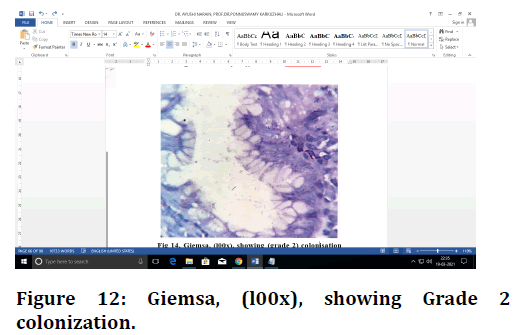
Figure 12. Giemsa, (l00x), showing Grade 2 colonization.
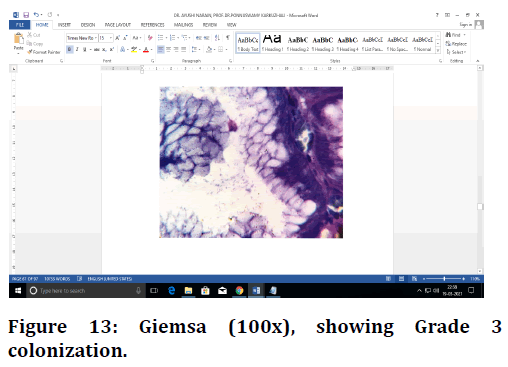
Figure 13. Giemsa (100x), showing Grade 3 colonization.
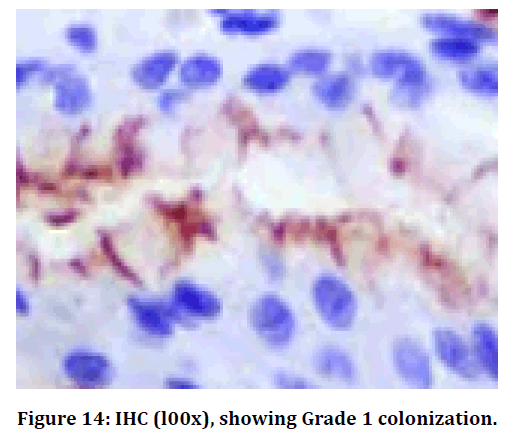
Figure 14. IHC (l00x), showing Grade 1 colonization.
Figure 15. IHC (l00x), showing Grade 2 colonization.

Figure 16. IHC (l00x), showing Grade 3 colonization.
Discussion
Helicobacter pylori infects the stomach and causes chronic active gastritis, which can lead to peptic ulcer disease, gastric adenocarcinoma, and mucosa-associated lymphoid tissue lymphoma. In this present study a total of 100 samples of antral biopsies were studied. For all these cases, clinical data and risk factors related to gastritis were collected. Out of 100 cases 60 cases were randomly selected for Giemsa and IHC. In a study conducted by Rajesh et al [5] 92 out of 265 cases were found to be positive for Helicobacter pylori with over all prevalence of 34.71%. Out of 603 cases, Helicobacter pylori was positive in 345 cases with prevalence of about 57.2% in the study conducted by Adisa et al. [6]. In the present study, out of 100 cases, 42 cases (42%) were positive for Helicobacter pylori. Infection with Helicobacter pylori occurs worldwide, but the prevalence differs among countries and various population groups in the same country. It is more common in developing countries, low socioeconomic strata, and overcrowded settings. The prevalence of Helicobacter pylori infection varies widely by geographic area, age, race, ethnicity, and socio-economic status. Javed et al. [7] in his study concluded that 80% of patients belonged to lower and middle class.
In the present study, Helicobacter pylori infection was found to be more prevalent in low socio-economic group (93%). According to Javed et al. [7] prevalence of Helicobacter pylori infection increases with age. Maximum number of Helicobacter pylori infection was seen in the age group of 46-55yrs. Adisa et al. [6] in his retrospective study observed that the prevalence of Helicobacter pylori associated gastritis was maximum between the age group of 41 to 50 years. A prospective study done by Rajesh et al [5] showed maximum prevalence of Helicobacter pylori infection in the age group of 36- 45 years. Few studies such as Shokrzadeh et al. [8] and Kaore et al. [9] reported increased H. pylori infection in age groups of 20-40 years than the older age group. Study conducted by Adlekha et al [10] H. pylori prevalence was highest in the age group of 81-90years with 75% patients showing H. pylori positivity. In the current investigation, the highest number of positive cases fall between the age group of 21-40 years. Rajesh et al [5] in his study showed that among the Helicobacter pylori positive patient 64.13% were males and 35.87% were females. The prevalence of Helicobacter pylori gastritis among males was 46.8% and females was 53.2% in a study conducted by Adisa et al. [6]. A study by Kaore et al [9] showed higher prevalence in male gender. Yangchun Zhu et al. [11] states that females had a higher infection rate than male. In the present study out of 42 Helicobacter pylori positive cases by IHC, 33 (79%) were males and 9 (21.4%) were females.
Sensitivity of H&E stain is low due to lack of contrast between the bacteria and the surrounding tissue. The specificity of H&E is also low due to its nonspecific staining of non-Helicobacter pylori bacteria in the stomach. Modified Giemsa is a cheap, easily applicable stain that can be performed in 15 minutes. The results are reliable. The sensitivity and specificity value are acceptable. Lack of contrast is the disadvantage of the Giemsa technique, but careful observation allows identification of the organisms correctly. Helicobacter pylori immunohistochemistry is an expensive and timeconsuming technique with duration of procedure, ranging from 1 hour to 24 hours. Sensitivity and specificity are high for the detect ion of Helicobacter pylori using IHC. In the current study, the histochemical methods such as H&E, Giemsa and IHC were analysed and compared for the sensitivity and specificity. In detecting the Helicobacter pylori. Wabinga et al. [12] evaluated the staining ability of Giemsa and IHC in gastric biopsies and inferred that the sensitivity of Giemsa stain was 85%, specificity was 89%, positive predictive value was 93% and negative predictive value was 74%. Hartman and Owens compared the routine stains and IHC [13]. The authors have noted that the sensitivity of special stains in the IR study was 62% and that of IHC was 97-100%. Kacar et a1 [14] observed that the sensitivity and specificity for H&E, Giemsa and IHC in detection of Helicobacter pylori was 97%, 80%, 97%, 90%, 100%, respectively. Babic et a1 [15] compared the sensitivity of detection of Helicobacter pylori in gastric biopsies and resected specimens using modified Giemsa and IHC which showed the sensitivity of Giemsa to be 73.3% and 90% for IHC. In the current study, sensitivity and specificity of Giemsa was 95.2% and 100%, respectively. Sensitivity and specificity of H&E was 90.5% and 100% respectively.
Consumption of alcohol, cigarette smoking and tobacco chewing are some etiologic factors that have a close relation with acid peptic diseases. Chronic active gastritis is reportedly associated with chronic alcohol ingestion. However, the inflammatory changes are likely to be related to concurrent Helicobacter pylori infection that is common among alcoholics. Furthermore, chronic alcoholism is also correlated with the presence of gastric metaplasia [16].
In a study conducted by Pandey et al [17] Helicobacter pylori infection showed an association with tobacco chewing.
Javed et a1. [7] in his study of 50 cases (40 males, 10 females) of patients with gastritis observed 28% of patients were smokers, 12% of them were tobacco chewers and 2% were alcoholics. Ananth et al [18] in his study of 146 chronic atrophic gastritis patients found out that the prevalence of H. pylori among smokers was 54.16% and among alcoholics was 54.28%. In present study 34% cases were smokers, 39% were alcoholics and 42% were tobacco chewers.
Conclusion
In the present study the age group of patients were in the range of 18-75 years with peak incidence occurring between 21-40 years. It was observed that Helicobacter pylori infection was more common in males and in the low socio-economic status. The most common presenting symptom associated with gastritis was abdominal pain. Risk factors associated with gastritis in present study were smoking, alcohol intake, and tobacco chewing in most of the patients. The most associated endoscopic finding with gastritis in the present study was erosions followed by erythema (linear bands/ erythematous mucosa).
In majority of the patients’ dietary habits associated with gastritis were spicy food intake. Sensitivity and specificity of Giemsa was 95.2 % and 100% respectively. Sensitivity and specificity of H&E was found to be 90.5 % and 100% respectively. Best results were obtained by Immunohistochemistry, especially when the density of organ ism was low. Immunohistochemistry is recommended for detection of Helicobacter pylori where the other two stains have low detection rate.
The cost, applicability and authenticity of the Giemsa stain make it an ideal stain in detecting Helicobacter pylori infection in gastric biopsies.
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgments
The encouragement and support from Bharath University, Chennai is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- Salih BA. Helicobacter pylori infection in developing countries: The burden for how long?. Saudi J Gastroenterol 2009; 15:201-207.
- Brown LM. Helicobacter pylori: Epidemiology and routes of transmission. Epidemiol Rev 2000; 22:283–297.
- Anderson J, Anderson JV, Baker LRI, et al. Gastrointestinal disease. In: Kumar and Clark Clinical medicine: 5th Edn. Philadelphia: WB Saunders; 2002; 272.
- Minhas KZ, Goraya F, Javid S, et al. Endoscopic and Histopathological evaluation of 306 dyspepsic patients. Pak J Gastroentrol 2003; 17:4-7.
- Kumar R, Bano G, Kapoor B, et al. Clinical profile in H. pylori positive patients in Jammu. Prevalence 2006; 92:34-71.
- https://www.semanticscholar.org/paper/Helicobacter-Pylori-Associated-Gastritis-in-A-Study-Adisa-Musa/b997b108d75fa735248f7ddcdf1569f623495518
- Javed M, Amin K, Muhammad D, et al. Prevalence of H. pylori. Professional Med 2010; 17:431-439.
- Shokrzadeh L, Baghaei K, Yamaoka Y, et al. Prevalence of Helicobacter pylori infection in dyspeptic patients in Iran. Gastroenterol Insights 2012; 4:24-27.
- Kaore NM, Nagdeo NV, Thombare VR. Comparative evaluation of the diagnostic tests for Helicobacter pylori and dietary influence for its acquisition in dyspeptic patients: A rural hospital based study 1n central India. JCDR 2012; 6:63 6-41.
- S Adlekha, T Chadha, P Krishnan, et al. Prevalence of Helicobacter Pylori infection among patients undergoing upper gastrointestinal endoscopy in a medical college hospital in Kerala, India. Ann Med Health Sci Res 2013; 3:559-563.
- Yangchun Zhu, Xiaoying Zhou, Junbei Wu, et al. Risk factors and prevalence of helicobacter pylori infection in persistent high incidence area of gastric carcinoma in Yangzhong City. Gastroenterol Res Practice 2014; 2014:10.
- Wabinga HR. Comparison of immunohistochemical and modified Giemsa stains for demonstration of Helicobacter pylori infection in an African population. Afr Health Sci 2002; 2:52-55.
- Hartman DJ, Owens SR. Are routine ancillary stains required to diagnose Helicobacter infection in gastric biopsy specimens? An institutional quality assurance review. Am J Clin Pathol 2012; 137:255-260.
- Kacar F, Culhac N, Ytikselen V, et al. Histologic demonstration of helicobacter pylori in gastric biopsies: Which is the best staining method?. Internet J Pathol 2003; 3.
- Babic T, Basic H, Miljkovic B, et al. Detection of Helicobacter pylori in gastric biopsy and resection specimens. Vojnosanit Pregl 2005; 62:39-43.
- Ko JK, Cho CH. Alcohol drinking and cigarette smoking: a "partner" for gastric ulceration. Free China Edn 2000; 63:845-54.
- Arvind P, Satyendra CT, Sutapa M, et al. Carcinogenic Helicobacter pylori in gastric pre-cancer and cancer lesions: Association with tobacco chewing. World J Gastroenterol 2014; 20:6860- 6868.
- Ananth AE, Saleem M, Gopal R, et al. Prevalence of Helicobacter Pylori infection in patients with atrophic gastritis and role of dark field microscopy in diagnosis. J Evolution Med Dent Sci 2015; 4:15873-15875.
Author Info
Ayushi Narain and Ponnuswamy Karkuzhali*
Department of Pathology, Sree Balaji Medical College & Hospital Affiliated to Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Ayushi Narain, Ponnuswamy Karkuzhali, Role of Immunohistochemistry Versus Hematoxylin and Eosin and Special Stains in Helicobacter Pylori Detection in Gastric Lesions, J Res Med Dent Sci, 2021, 9(6): 123-132
Received: 08-May-2021 Accepted: 15-Jun-2021
