Research Article - (2019) Volume 7, Issue 1
The Comparison Effect of Ciprofloxacin and Cotrimoxazole in Infection Prophylaxis during Chemotherapy Treatment of Multiple Myeloma Patients
Reza Aghabozorgi, Mandana Majidian and Ali Arash Anoushirvani*
*Correspondence: Ali Arash Anoushirvani, Department of Hematology and Medical Oncology, Arak University of Medical Sciences, Arak, Iran, Email:
Abstract
Introduction: Infections are the main cause of mortality in half of the patients suffering multiple myeloma and disrupt the treatment process. Administration of the best prophylactic antibiotic with a minimum of complications is of prime importance. Aim of this study was to compare two antibiotics of Co-trimoxazole and Ciprofloxacin with different functional mechanisms.
Material and Method: 72 new patients suffering from multiple myeloma were admitted in this clinical trial study. The patients were divided into two equal groups by simple randomization. The first group received 500 mg ciprofloxacin capsules once daily, and the second group received co-trimoxazole tablets at a dose of 160/400 daily for 4 weeks. Occurrence of various types of infections were followed and compared by clinical and laboratory methods for a period of 6 months.
Results: Sex ratio, mean age, and the type of regimen prescribed to the patients were not different between two groups (p ≥ 0.05). The occurrence of infection in the ciprofloxacin group was 16.7% (6 cases) and 38.9% (14 cases) in the cotrimoxazole group during the 6 months (p ≤ 0.05). The most common type of infection in both groups was pneumonia. There was no significant difference in the occurrence of infection according to the type of regimen prescribed to the patients (p ≥ 0.05). No serious complication resulting in discontinuation of treatment was reported in the two groups. The bacterial resistance to ciprofloxacin was less than co-trimoxazole.
Conclusion: It was concluded that ciprofloxacin with less resistance can be routinely used in the prevention of various infections during the chemotherapy. In multiple myeloma, infection control as well as recognition of risk factors and phase of the disease has the main role in treatment.
<Abbrevations
The development of biology, genetics, medicine, pharmacy at the end of XX century, bring along cloning of mammals, made the map of the human genome, genetic investigation and gene therapy reach new successes, assisted reproduction to become available procedure
Introduction
Multiple myeloma is the neoplasm of cells of Plasma cell that accounts for about 10% of hematologic malignant disorders [1,2] estimates the annual prevalence of it to be 4.3 per 100,000 of population, according to which more than 20,000 new cases will be recorded each year in the United States. 13% of all hematologic cancers are found in whites and 33 percent in black people. In total, 10% of all hematologic cancers and one percent of cancers are included [3]. In the black population, multiple myeloma happens twice more than whites. This difference is generally related to the higher incidence of monoclonal gammopathy of undetermined significance in black people [4,5].
Risk factors are those that increase the probability of development and occurrence of diseases. There are the following risk factors for multiple myeloma:
The most common symptom of multiple myeloma is excessive fatigue associated with bone pain [6]. Anaemia exists in almost 75% of these patients, and osteolytic lesions of the skeletal system have been reported in 80% of patients. And accompanied with that, hypercalcium (15%) and increased serum creatinine (up to 2 mg/dL) exists in 20% of cases [6]. About 1% to 2% of patients suffering from multiple myeloma also have extramedullary disease at the beginning of treatment. And 8% also suffer from extramedullary lesions during their disease.
Due to the high incidence of infections in these patients, the matter of antibiotic prophylaxis, especially with new therapies, has recently been focus of attention. Recently, many studies have been conducted on the prevention of infections in these patients, including the function of the influenza vaccine and Pneumococcal, or the prevention of bacterial, fungal and viral infections in these patients. It is recommended that patients with multiple myeloma receive the influenza and Pneumococcal vaccines every year [7].
Since infections are responsible for an increase in mortality and morbidity rates in these patients, reducing these infections with antibiotics is important if prophylaxis works. Administration of effective antibiotic with minimal complications and interactions with the treatment is of prime importance. Therefore, in this study, two different groups of antibiotics with different functional mechanisms and different bacterial spectrum were compared in a clinical trial study.
Materials and Methods
In this clinical trial study, 72 patients suffering from multiple myeloma who referred to the clinic and the hematologic admission ward of Khansari and Amir-Al- Momenin Hospitals of Arak city, Iran, participated in the study based on inclusion and exclusion criteria.
The main inclusion criteria were plasma cell more than 10% in bone marrow biopsy. The other inclusion criteria were as: patients with multiple myeloma with proven disease based on bone marrow specimens and the other criteria, patients aged between 30 and 70 years, proper liver and kidney condition and in the first chemotherapy course.
Exclusion criteria were as: Previous history of chemotherapy, AIDS, alcohol abuse, thyroid dysfunction, history of immunodeficiency for any reason, smoking and drug abuse.
During the study, 3 patients stopped taking part in the study or were excluded from the study. Finally from 75 patients, 72 patients were enrolled in the study.
Patients were aged between 30-70 years, and had adequate liver and kidney status that were under primary chemotherapy. Patients undergoing primary chemotherapy were randomly included in one of two treatment groups. Therefore, it was entirely coincidental that the patient may be in one of the four therapeutic groups of thalidomide, dexamethasone or thalidomide group, dexamethasone, volcid or volcid group, Cyclophosphamide, dexamethasone, or melphalan group, prednisolone, thalidomide. Patients with prior history of chemotherapy, existence of HIV neuropathy, alcohol abuse and thyroid dysfunction for at least three months before entering the study, and smoking, using drugs, having pain in lower part of back and consuming other chemotherapy drugs were excluded from the study.
The patients were divided into two equal groups using the simple random method of coin tossing. Group A, received 500 mg ciprofloxacin capsule once a day and group B also received co-trimoxazole tablets at a dose of 160 mg-400 mg daily. Patients of both groups received medications.
After this treatment period, patients were evaluated and followed up for six months and both groups were compared in regard of the incidence rate of infections. In order to investigate the incidence of infection, chest X-ray was taken from patients with fever respiratory symptoms, and in the case of presence of clinical signs and graphic findings in favour of pneumonia, they were recorded in the special sheet of the patient. During the chemotherapy period, patients were examined to check out for sepsis or any other infection and take required measures. To check the incidence of urinary infections, every two weeks urine culture was made from patients (the analysis was also made if there were signs of burning and frequent urination). If patients were hospitalized with suspected symptoms of infection, a complete examination including chest X-ray, urine culture, blood culture and ESR, CRP was performed for patients to inspect results in case of any specific infection.
If patients were hospitalized due to sepsis, according to the related criteria the patient was placed in the list of patients suffering from sepsis. In the event of existence of abscess, gram stain, culture and PCR were performed. In case of skin lesions, PCR and gram stain and culture were investigated. In patients with resistant fever, in order to examine endocarditis, thoracic echocardiography and blood culture and PCR from blood sample was done and the patient was examined for criterion of DUKE. If there were signs of infection and the presence of pleural effusion or ascites, the culture and smear of these fluids was sent. In case of suspected pneumonia, gram stain and sputum culture were also performed, and PCR was examined for some patients in the case of doing BAL sample. Mild infections that most of them include upper respiratory tract infections and fungal and viral diseases such as herpes were not studied in this research.
The person who recorded information had no knowledge of the group to which the patient belonged, and also taking into consideration the medical ethics, patients were unknowing about their placement in the related groups.
Data analysis method
After completion of the checklist and entering information in SPSS-20 software, based on the goals and assumptions, descriptive statistics including tables, diagram and dispersion and central indexes were obtained, and the analytical analysis was performed with appropriate methods such as independent and paired ttest, or non-parametric methods to test the mean difference in different groups.
Data collection
All data of the present study were obtained through questionnaire and information collecting form.
Ethical considerations
The present study was conducted after obtaining permission from the Ethics Committee. Code is the ethics of study. Names of patients were mentioned as codes in the data collection questionnaire. Oral and written consent was received from all patients participating in this study. And we made a commitment not to publish the raw results. Also, in the case of any resistant infections, or the occurrence of antibiotic-related complications or drug resistance, the patient was excluded from the study and all appropriate diagnostic and therapeutic measures were taken. All the principles and rules of the Helsinki instruction were observed.
Results
A total of 72 patients suffering from multiple myeloma who were planned to undergo primary chemotherapy were included in this study. The collected data showed that there were 26 males (72.2%) and 10 females (27.8%) in the group co-trimoxazole, and also 24 males (66.7%) and 12 females (33.3%) in ciprofloxacin group. Data analysis with Chi-square test showed that there was no significant difference between the gender of two groups (p=0/798).
The mean age of patients in the co-trimoxazole group was 64.1 ± 6.9 years old, and in the ciprofloxacin group was 68.7 ± 7.2 years. Data analysis with independent ttest showed that there was no significant age difference between the two groups (p=0.945).
Frequency distribution of the type of regimen prescribed to the two groups of ciprofloxacin and co-trimoxazole was shown in Figure 1. According to the Fishers exact test, there was no significant difference in the two groups (p=0.082).
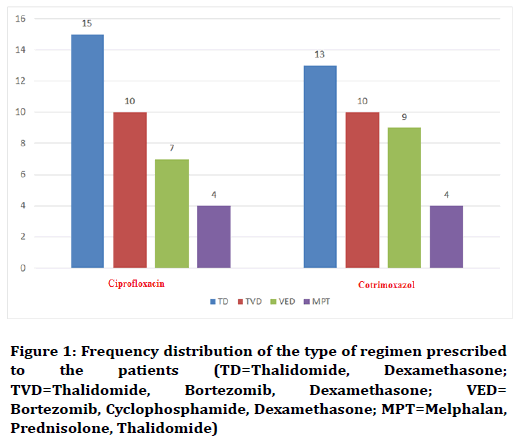
Figure 1. Frequency distribution of the type of regimen prescribed to the patients (TD=Thalidomide, Dexamethasone; TVD=Thalidomide, Bortezomib, Dexamethasone; VED=Bortezomib, Cyclophosphamide, Dexamethasone; MPT=Melphalan, Prednisolone, Thalidomide)
In this study, for some patients, Kyprolis medicine was also used. Accordingly, 2 patients (5.5%) in the cyprofloxacin group and one patient (2.8%) in the cotrimoxazole group were treated with kyprolis. Data analysis using chi-square test showed no significant difference in this field (p=0.555).
The final outcome which was considered in the present study was the rate of infection of the patients. The results of this study indicated that in the group of co-trimoxazole 14 patients (38.9%), and in the ciprofloxacin group, 6 patients (16.7%) suffered from various types of infection. Data analysis with chi-square test showed that the incidence of infection in the co-trimoxazole group was significantly higher than the other group (p=0.035).
The rate of incidence of infection according to the type of regimen prescribed to the patients showed no significant difference in the two groups based on the statistical test of Mann-Whithney (Table 1).
| Group | TD | TVD | VED | MPT | P |
|---|---|---|---|---|---|
| Ciprofloxacin | 2 | 2 | 1 | 1 | 0.061 |
| Co-trimoxazole | 5 | 3 | 4 | 2 | |
| Total | 7 | 5 | 5 | 3 | 20 |
Table 1: Frequency distribution of infection incidence according to the type of regimen prescribed to both groups
In regard of type of infection, in co-trimoxazole group, 7 participants (19.4%) experienced pneumonia, 3 participants (8.3%) had urinary infections, 2 had sepsis (5.6%) and 2 (5.6%) got cellulite. In the ciprofloxacin group, 4 participants (multiple myeloma .1%) suffered from pneumonia, 1 (2.8%) experienced urinary infections and 1 (2.8%) had sepsis. Chi-square test showed that there was no significant difference in regard to the type of infection between the two groups (p=0.250).
Discussion
In this study, prophylaxis antibiotics were used to prevent primary infections in patients with multiple myeloma who were undergoing chemotherapy. In total, 16.7% of patients under prophylaxis with ciprofloxacin, and 38.9% of patients with co-trimoxazole prophylaxis, got infection which indicated a significant difference between the two groups. Our study showed that using ciprofloxacin can more effectively prevent the onset of infection in patients suffering from multiple myeloma.
Infection is one of the most important causes of mortality in patients with multiple myeloma. Increasing the susceptibility of patients suffering from multiple myeloma to infection is due to the mutual interaction of anti-neoplastic therapies and complications related to age and disease [4-6].
The inherent immune deficiency resulting from multiple myeloma entangles different parts of immune system including functional impairment of B cells [3], T cells [5] and Natural killer cells [8]. However, what mechanism is more relevant to the incidence of infection than others is still unclear, but polyclonal hypogammaglobulinemia is classically associated with occurrence of infection by capsular bacteria such as Streptococcus pneumoniae [6] and Hemophilus influenzae [9].
Studies from the 1980s indicated a decrease in the frequency of episodes of fever and the prevalence of infection among patients who received preventive antibiotics [10,11]. The strongest evidences are related to the prophylaxis with fluoroquinolone, which is indicative of their association with decrease of fever, fixed infection and blood infections by gram-positive and gram-negative bacteria [12-14]. Therefore, in high-risk patients suffering cancer and undergoing chemotherapy, prophylaxis with quinolones is recommended by the American Association of Infectious Diseases [15].
Routine use of prophylactic antibiotics in patients with multiple myeloma has not been recommended, because the research conducted in this field is still insufficient. In older studies, the prophylaxis with co-trimoxazole showed effective impacts in prevention of primary infection in patients with multiple myeloma, but recently because of the vast resistance of most bacterial species to co-trimoxazole, the use of this drug is recommended to prevent mild infection [16,17]. The present study also confirms this issue. Gay et al. reported that despite the daily use of clarithromycin, the incidence of severe infections in patients with multiple myeloma was not changed significantly [18].
In a study Vesole et al. evaluated in the first two months, the incidence of infection in patients suffering from multiple myeloma who were recently undergoing chemotherapy. In their study patients were divided into three groups of ciprofloxin, co-trimoxazole and control. The overall incidence of infection in this clinical trial was 21.8% on average, and the three groups did not show significant difference [19]. In the present study, in contrast to the above mentioned study, ciprofloxacin was more effective than co-trimoxazole.
In a study conducted by Jung et al., patients suffering from multiple myeloma who were undergoing chemotherapy with bortezomib received 500 mg of levofloxacin per day as a preventive action [20]. Levofloxacin was very effective in reducing the infection in these patients.
In the study of Palumbo et al. done in 2006, the therapeutic effects, survival, and complications of the two therapies of Mulfalan, Prednisolone [MP] and Mulfalan, Prednisolone, Thalidomide [MPT] were compared, and it was determined that MPT was a selective type of regimen prescribed to the patients, especially in the elderly with multiple myeloma and had the highest effectiveness with the minimum of complications. The rate of response in this method was significantly higher than the MP method, but the incidence of venous thrombosis was reported higher in this method. Similar to the results of our study, the chance of infection in these two therapeutic methods was the same [21].
In the study of Richardson et al. performed in 2007, similar to the results of the present study, dexamethasone versus dexamethasone and thalidomide, did not lead to an increased risk of infection [22].
Also, in the study of Facon et al. in 2007, there was no difference in the incidence of infection in the type of regimen prescribed to the patients of Melphalan and Prednisolone [MP] compared to Melphalan, Prednisolone and Thalidomide [MPT] [23].
In the study conducted by Dimopoulos et al. in 2007, the MPT regimen compared with MP increased the risk of infection incidence [24].
The results of a study done by Nencioni et al. [25] and Wang et al. [26], also indicated that prescription of melphalan, with the exception of a slight increase in varicella zoster and herpes simplex, did not increase the risk of infection in patients with multiple myeloma.
Finally, the different results of some studies, regarding the risk of infection, can be related to the sample size under study, the method of infection screening, the phase of the disease, the type of complications and associated illnesses, and the duration of follow-up.
In the present study no cases of drug allergy and complications related to co-trimoxazole and ciprofloxacin were observed.
The use of more appropriate antibiotic types in patients suffering from malignant diseases is of great importance. Although previous studies have shown that prevention with ciprofloxacin is effective in preventing gramnegative infections, but there are concerns about the increased resistance of Gram-positive pathogens to that. One of the limitations of this study was the relatively low sample size, which can be justified due to the limited time of the study. On the other hand, another group under the title of control group would have been added to this study so its difference with the two intervention groups could be investigated.
Conclusion
Considering the existence of a significant difference between co-trimoxazole and ciprofloxacin, it seems that like some other studies, in Iran, the bacterial resistance to ciprofloxacin is lower than that of co-trimoxazole as well. Therefore, it appears as though that this antibiotic or its similar types classified in one group can be routinely used to prevent various infections during the chemotherapy course of patients with multiple myeloma. Moreover, the risk of infection has no relation to the type of regimen prescribed to the patients, and the key to the success in prevention and treatment of infection in patients suffering from multiple myeloma is the recognition of risk factors and phase of the disease.
Acknowledgments
The results were obtained from M.D. dissertation by No. 1182 and the authors are very grateful to the Vice-Chancellor of research and technology of Arak University of medical Sciences, Arak, Iran.
Conflict of Interest
The authors declared no potential conflict of interests with respect to the authorship and/or publication of this paper.
References
- Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International myeloma working group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014; 15:e538-48.
- Cecilie Blimark, Erik Holmberg, Ulf-Henrik Mellqvist, et al. Multiple myeloma and infections: A population-based study on 9253 multiple myeloma patients. Haematologica 2015; 100:107–13.
- Kyle RA, Therneau TM, Rajkumar SV, et al. Incidence of multiple myeloma in Olmsted County, Minnesota: Trend over 6 decades. Cancer Cytopathol 2004; 101:2667-74.
- Landgren O, Gridley G, Turesson I, et al. Risk of monoclonalgammopathy of undetermined significance [MGUS] and subsequent multiple myeloma among African American and white veterans in the United States. Blood 2006; 107:904-6.
- Landgren O, Graubard BI, Katzmann JA, et al. Racial disparities in the prevalence ofmonoclonal gammopathies: A population-based study of 12,482 persons from the National Health and Nutritional Examination Survey. Leukemia 2014; 28:1537-42.
- Kyle RA, Gertz MA, Witzig TE, et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc 2003; 78:21-33.
- Malnick SD, Atali M, Israeli E, et al. Trimethoprim/sulfamethoxazole-induced rash, fever, abnormal liver function tests, leukopenia, and thrombocytopenia. Ann Pharmacother 1993; 27:1139-40.
- Rajkumar SV, Blood E, Vesole D, et al. Phase III clinical trial of thalidomide plus dexamethasone compared with dexamethasone alone in newly diagnosed multiple myeloma: A clinical trial coordinated by the Eastern Cooperative Oncology Group. J Clin Oncol 2006; 24:431-6.
- Rajkumar SV, Rosiñol L, Hussein M, et al. Multicenter, randomized, double-blind, placebo-controlled study of thalidomide plus dexamethasone compared with dexamethasone as initial therapy for newly diagnosed multiple myeloma. J Clin Oncol 2008; 26:2171.
- Regelink JC, Minnema MC, Terpos E, et al. Comparison of modern and conventional imaging techniques in establishing multiple myeloma‐related bone disease: A systematic review. Br J Haematol 2013; 162:50-61.
- Roodman GD. Pathogenesis of myeloma bone disease. Leukemia 2009; 23:435.
- Kumar S, Fonseca R, Ketterling RP, et al. Trisomies in multiple myeloma: Impact on survival in patients with high-risk cytogenetics. Blood 2012.
- Rajan AM, Rajkumar SV. Interpretation of cytogenetic results in multiple myeloma for clinical practice. Blood Cancer J 2015; 5:e365.
- Kuehl WM, Bergsagel PL. Multiple myeloma: Evolving genetic events and host interactions. Nat Rev Cancer 2002; 2:175.
- Bergsagel PL, Kuehl WM. Chromosome translocations in multiple myeloma. Oncogene 2001; 20:5611.
- Fonseca R, Bailey RJ, Ahmann GJ, et al. Genomic abnormalities in monoclonal gammopathy of undetermined significance. Blood 2002; 100:1417-24.
- Seidl S, Kaufmann H, Drach J. New insights into the pathophysiology of multiple myeloma. Lancet Oncol 2003; 4:557-64.
- Gay F, Rajkumar SV, Coleman M, et al. Clarithromycin (Biaxin)‐lenalidomide‐low‐dose dexamethasone (BiRd) versus lenalidomide‐low‐dose dexamethasone (Rd) for newly diagnosed myeloma. Am J Hematol 2010; 85:664-9.
- Vesole DH, Oken MM, Heckler C, et al. Oral antibiotic prophylaxis of early infection in multiple myeloma: A URCC/ECOG randomized phase III study. Leukemia 2012; 26:2517.
- Jung SH, Kang SJ, Jang HC, et al. Effect of levofloxacin prophylaxis for prevention of severe infections in multiple myeloma patients receiving bortezomib-containing regimens. Int J Hematol 2014; 100:473-7.
- Palumbo A, Bringhen S, Caravita T, et al. Oral melphalan and prednisone chemotherapy plus thalidomide compared with melphalan and prednisone alone in elderly patients with multiple myeloma: Randomised controlled trial. Lancet 2006; 367:825-31.
- Richardson P, Mitsiades C, Schlossman R, et al. The treatment of relapsed and refractory multiple myeloma. Hematology Am Soc Hematol Educ Program 2007; 2007:317-23.
- Facon T, Mary JY, Hulin C, et al. Melphalan and prednisone plus thalidomide versus melphalan and prednisone alone or reduced-intensity autologous stem cell transplantation in elderly patients with multiple myeloma (IFM 99–06): A randomised trial. Lancet 2007; 370:1209-18.
- Dimopoulos M, Spencer A, Attal M, et al. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med 2007; 357:2123-32.
- Nencioni A, Garuti A, Schwarzenberg K, et al. Proteasome inhibitor‐induced apoptosis in human monocyte‐derived dendritic cells. Eur J Immunol 2006; 36:681-9.
- Wang M, Dimopoulos MA, Chen C, et al. Lenalidomide plus dexamethasone is more effective than dexamethasone alone in patients with relapsed or refractory multiple myeloma regardless of prior thalidomide exposure. Blood 2008; 112:4445-51.
Author Info
Reza Aghabozorgi, Mandana Majidian and Ali Arash Anoushirvani*
Department of Hematology and Medical Oncology, Arak University of Medical Sciences, Arak, IranCitation: Reza Aghabozorgi, Mandana Majidian, Ali Arash Anoushirvani, The comparison effect of ciprofloxacin and cotrimoxazole in infection prophylaxis during chemotherapy treatment of multiple myeloma patients, J Res Med Dent Sci, 2019, 7(1): 1-6
Received: 10-Dec-2018 Accepted: 04-Jan-2019 Published: 28-Feb-2019
