Research - (2021) Volume 9, Issue 4
The Relation of Salivary Cathelicidin and Beta-Defensin with Dental Caries of Schoolchildren
Ghufran Mohammed SS Al-Ali1*, Zainab Juma Jafar1 and Batool Hasan AL-Ghurab2
*Correspondence: Ghufran Mohammed SS Al-Ali, Department of Pediatric and Preventive Dentistry, College of Dentistry, University of Baghdad, Iraq, Email:
Abstract
Background: Salivary antimicrobial peptides contribute to oral health by playing a role in innate response against oral diseases, owing to their antimicrobial, antiplaque and immunomodulatory activity. Aim of study: This study was performed to determine the levels of cathelicidin LL-37 and beta-defensins in saliva of children, and assess the relationship of both biomarkers to dental caries prevalence and severity. Materials and Methods: Eighty-eight schoolchildren of age 6-7 years participated in the study. The sample was divided into two groups; study group of 44 children with moderate/severe caries and control group of 44 children free from caries. Oral examination and measurement of clinical parameters of caries experience by dmf index, caries severity by ICDAS index. Saliva samples were collected from both groups, then submitted to enzyme-linked immunosorbent assay. Results: A non-significant difference was found in salivary level of cathelicidin LL-37 between the two groups. However, the results revealed that the salivary level of beta-defensin was significantly decreased in the study group than in the control group. Moreover, a significant negative correlation was found between both peptides with caries experience and caries severity. Conclusion: The results support the idea of a protective role of antimicrobial peptides as important immune molecules in maintaining the oral health against caries. Therefore, it could be a gate for more advanced future biological screening method for caries susceptibility and new strategies for prevention.
Keywords
Dental caries, Salivary, Schoolchildren, Oral examination, Cathelicidin
Introduction
A good oral health contributes to maintaining the overall health [1]. Dental caries is one of the most widespread oral diseases. Caries in primary dentition was described as the 12th most prevalent medical condition among children [2]. Despite being extensively researched, a persisting ambiguity and incertitude about its pathogenesis and predicting factors still exist [3]. Saliva has a protective role for being a source for many proteins and peptides that have antimicrobial activity such as lactoferrin, immunoglobulin, lysozyme and cationic peptides [4]. The two major antimicrobial peptide families (AMPs) found in oral cavity; are cathelicin LL37 and defensin (alpha & beta). These peptides found in saliva, gingival crevicular fluid (GCF) and oral mucosa. Beta-defensins are expressed and derived from oral epithelium, while neutrophils the main source for alpha defensin and cathelicidin. These salivary AMPs make the first line of innate immune defense in human oral cavity [5,6]. AMPs were observed to have a synergistic interaction in activity. The main mechanism of action is cell killing by membrane lysis, though cell killing via intracellular targets also exists [7,8]. Cathelicidin LL-37 has a broad antimicrobial activity against both cariogenic & periopathogenic bacteria [9]. AMPs help ensuring the normal flora in a steady condition in the mouth, intestine and skin niches [10]. Salivary cationic AMPs have a potential to serve as proteomic biomarkers for various oral diseases such as dental caries [11], however the applicability is still submitted to current research and debate.
Materials and Methods
The sample consisted of eighty-eight schoolchildren aged 6-7 years. And was divided into two groups each has equal number of boys 22 and girls 22; the study group comprises 44 children with caries in moderate and severe levels ICDAS (d3, d4, d5, and d6) according to WHO, 2013 criteria and control group of 44 children were free from caries. A formal permission was taken from authorities in general directorate of education in Mosul city as well as schools’ managers for children examination and a consent and information papers were sent to children’s parents about study to have their approval. In addition to the ethical committee approval to the protocol that was obtained earlier from dentistry college university of Baghdad before proceeding in the study. Information collection verified all children were healthy without systemic disease or known medication usage chronic or at time of the study. Oral examination and measurement was performed of clinical parameters of caries experience (dmf according to WHO), caries severity (ICDAS). Saliva samples were collected from both groups after finishing information collection and performing oral examination of children. Children were previously instructed to refrain from drinking and eating at least one hour before saliva collection, and the procedure for collection was clearly demonstrated to children. A sample of approximately 5ml. of unstimulated saliva was collected from each child. Spitting method was used for collecting saliva in clean plastic containers labeled with a specific code that was also registered in each child information case sheet. The procedure needed about 3-5 minutes. Finally, the samples were kept in cooling box before being transported to laboratory for the next steps. Whereas, each sample has undergone centrifuge for 10000Xg for 5 minutes at 2-8°C [9]. Then supernatant of about 1-2ml. was recovered to Eppendroff tubes holding the same code prepared for storing at -20°C for later laboratory analysis, then ELISA analysis was performed for detection of the concerning biomarkers. Two ELISA kits for two biomarkers peptides were used: cathelicidin LL-37 kit and beta-defensin kit (Al- Shukairate establishment for medical supply, USA) and the procedure was performed according to manufacturer instructions.
Results
Demographics and clinical parameters: There were no significant differences regarding age and gender between the two groups. Control group had ICDAS (d0) and dmf=0, Whereas the study group showed different stages of caries severity (d3-d6) with highest percentage 63.64% having severe extensive caries (d6).
Salivary AMPs and their relation to dental caries experience: The levels of cathelicidin (LL-37) and beta-defensin (HBD) in saliva of both study and control groups are illustrated in Table 1. The present study found that the control group had higher level than the study group; however with non-significant difference (p>0.05), while the mean salivary level of beta-defensin was significantly higher in the control group than in the study group (P<0.05). Moreover, a highly significant negative association between the level of both cathelicidin LL-37 and betadefensin with caries experience (ds) in the study group, and significant negative association between LL-37 and beta-defensin with (dt) in the study group seen in Table 2. Even though; when comparing the level of cathelcidin LL-37 in the carious group measured by dmf with caries free group, it revealed a non-significant difference, while it revealed a significant decrease in the level of beta-defensin in the carious group than in the control group as shown in Table 3.
Table 1: Levels of salivary cathelicidin LL-37 and beta-defensin in study and control groups.
| LL-37 (ng/ml) | P-value | |||||||
| Group | Minimum | Maximum | Mean | ± SD | ± SE | T-test | df | |
| Control | 0.01 | 8.6 | 2.179 | 1.883 | 0.284 | 0.874 | 86 | 0.384 NS. |
| Study | 0.01 | 7.8 | 1.83 | 1.861 | 0.281 | |||
| Total | 0.01 | 8.6 | 2.004 | 1.869 | 0.199 | |||
| HBD (µg/ml) | P-value | |||||||
| Group | Minimum | Maximum | Mean | ± SD | ± SE | T-test | df | |
| control | 53.2 | 1308 | 275.352 | 256.56 | 38.678 | 2.108 | 86 | 0.038 S. |
| study | 46.3 | 664.8 | 176.495 | 175.932 | 26.523 | |||
| Total | 46.3 | 1308 | 225.924 | 224.283 | 23.909 | |||
Table 2: Correlation coefficient between caries experience and salivary cathelicidinLL-37and beta-defensin in the study and control groups.
| Study group | r | P value | |
|---|---|---|---|
| LL-37 | ds | -0.398 | 0.008 HS |
| dmfs | -0.198 | 0.197 NS | |
| dt | -0.323 | 0.032 S | |
| dmft | -0.2 | 0.193 NS | |
| HBD | ds | -0.463 | 0.002 HS |
| dmfs | -0.224 | 0.144 NS | |
| dt | -0.317 | 0.036 S | |
| dmft | -0.165 | 0.285 NS |
Table 3: Comparison of levels of salivary cathelicidin and beta-defensin in by caries experience measured by (dmf).
| Variables | Caries experience | T | P value | |||||
|---|---|---|---|---|---|---|---|---|
| caries free | caries dmf>=4 (high) | |||||||
| N | Mean | ±SD | N | Mean | ±SD | |||
| LL-37 | 44 | 2.179 | 1.883 | 44 | 1.83 | 1.861 | 0.874 | 0.384 NS |
| HBD | 44 | 275.352 | 256.56 | 44 | 176.495 | 175.932 | 2.108 | 0.038 S |
Salivary AMPs and their relation to dental caries severity: The results show a significant negative association between the level of cathelicidin LL-37 and caries severity, and highly significant negative association between betadefensin and caries severity as shown in Table 4. On the other hand, when comparing the stages of caries severity with each other and with the control group, it revealed a non-significant difference as shown in Table 5.
Table 4: Correlation coefficient between caries severity and salivary cathelicidin and beta-defensin in study and control groups.
| Group | r | P-value | ||
|---|---|---|---|---|
| Study | ICDAS | LL-37 | -0.301 | 0.047 S |
| HBD | -0.394 | 0.008 HS | ||
Table 5: Comparison of levels of salivary cathelicidin and beta-defensin among different stages of caries severity measured by (ICDAS).
| N | % | Mean | ±SD | ±SE | F | P value | ||
|---|---|---|---|---|---|---|---|---|
| LL-37 | d0 | 44 | 50 | 2.179 | 1.883 | 0.284 | 1.088 | 0.41 NS |
| d3 | 3 | 3.41 | 3.227 | 1.956 | 1.129 | |||
| d4 | 3 | 3.41 | 3.163 | 2.508 | 1.448 | |||
| d5 | 10 | 11.36 | 2.069 | 1.964 | 0.621 | |||
| d6 | 28 | 31.82 | 1.452 | 1.702 | 0.322 | |||
| HBD | d0 | 44 | 50 | 275.352 | 256.56 | 38.678 | 2.771 | 0.052 NS |
| d3 | 3 | 3.41 | 334.067 | 238.89 | 137.923 | |||
| d4 | 3 | 3.41 | 219.533 | 122.117 | 70.504 | |||
| d5 | 10 | 11.36 | 268.99 | 250.167 | 79.11 | |||
| d6 | 28 | 31.82 | 121.968 | 117.832 | 22.268 |
Salivary AMPs and correlation with each other: The present study results show a highly significant positive association between salivary levels of cathelicidin LL-37 and beta-defensin as demonstrated in Table 6.
Table 6: Correlation coeffeicient between levels of salivary cathelicidin LL-37 and beta-defensin.
| Group | r | P-value |
|---|---|---|
| Study | 0.902 | 0.000 HS |
| Control | 0.762 | 0.000 HS |
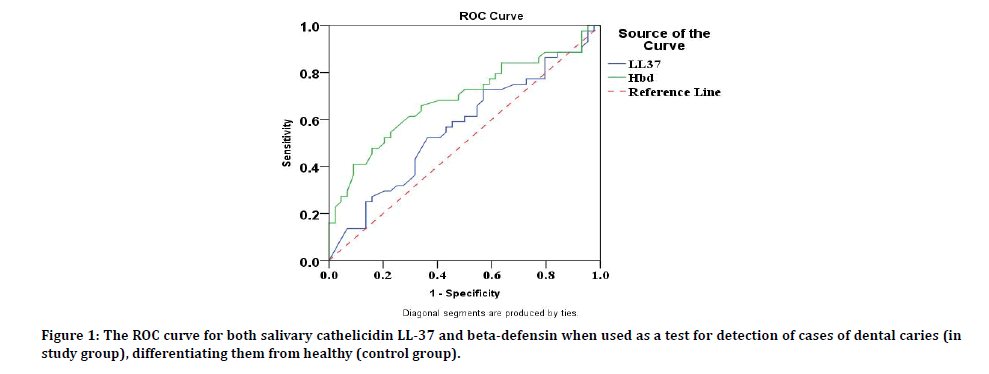
Specificity and sensitivity test of biomarkers cathelicidin LL-37 and beta-defensin: The use of the ROC curve to estimate the sensitivity (rate of true positive) and 1-specificity (rate of false positive) to distinguish the best salivary peptide biomarkers for disease detection. The area under the curve for salivary beta-defensin was significantly higher (0.686) from (0.5) value of an equivalent test, (p=0.003). The area under the ROC (AUR) results was “accepted” for the beta-defensin discrimination ability while “poor or no discrimination ability” of the cathelicidn LL-37 (p=0.293). Therefore, beta-defensin was considered as the best sensitivity detector than cathlicin LL-37 (1-specificity) as shown in Figure 1.
Figure 1: The ROC curve for both salivary cathelicidin LL-37 and beta-defensin when used as a test for detection of cases of dental caries (in study group), differentiating them from healthy (control group).
Discussion and Conclusion
Challenges still exist in recognizing risk factors and predictors of oral diseases [12]. By analyzing a set of components (as proteins and peptides) found in saliva, it could be possible to estimate and monitor risk of occurrence and progression of an oral disease [13,14]. To the best of our knowledge, this is the first study in Iraq to investigate the association between caries and biomarkers of innate immunity (cathelicidin and beta-defensins).
Cathelicidin LL-37 and beta-defensin with caries experience among children
There are only few previous studies available that, have addressed the subject of caries and AMPs directly, specifically the cathelicidin. Nevertheless, the findings of the present study agree with the previous observations that reported similarly non-significant difference between children with caries and children without caries in cathelicidin level [15, 16], with a trend of being lower in children with high caries activity than children who are free from caries or with low caries activity [15]. While one study found it to be significantly lower in subjects with caries [9]. A possible explanation for nonsignificant difference could lie partly in previous observations that, cathelicidin levels were not correlated with SM (the main bacteria involved in caries process) [15,16]. It may be also related to high variability of cathelicidin concentration in children’s saliva.
Regarding the defensins, the present study found a significant difference in the level of betadefensins in saliva between the two groups being significantly lower in the children with caries (moderate and severe) than children who are caries free. Furthermore, a significant negative correlation between beta-defensin level and caries experience measured by (ds/dt) score. This result joins controversial results from other previous studies; while it agrees with former observation where defensins (alpha-defensin) level was lower in children with caries [17] and a negative association between defensins (both alpha- and beta- defensins) with dental caries in children [18]. Inversely, it disagrees with other outcomes [15,16,19] that found nonsignificant difference among older childrens and toddlers with caries (and severe ECC) and those free from caries in concentration levels of betadefensin and alpha-defensin. Likewise, it was different from findings [11,20] that showed a significant difference where beta-defensin level was higher in those with caries than those were free from caries, also another researcher [21] failed to demonstrate correlation between caries experience and AMPs levels.
This discrepancy of results could reflect a variation in the number of gene copy and variation in responses to bacterial stimulation. Another reason a way from methodological differences is sample age and developmental stage of caries in the studies. For example, in older children, caries may become more established and presence of species other than SM, LB have impact in caries progression. The notion that maturation of AMPs responses continues with age and differences in responding ability to bacterial colonization over time could cause elevated susceptibility to oral diseases [22]. Beta-defensin expression has been hypothesized to be genetically controlled with involvement of variations in copy number of DEFB4, DEFB103, DEFB104 or regulatory untranslated-5 region (UTR) polymorphism in DEFB1 genes. Encoding genes for peptides play a critical role in caries susceptibility [23].
AMPs also have versatile antimicrobial activity, in addition, intrinsic variations in susceptibility or resistance profiles of Streptococus mutans strains to the salivary AMPs produced by their host have been previously reported [21,24,25]. Specific amount of AMPs may affect colonization of plaque by particular strain. The susceptibility of strains isolated from high caries individuals exhibited higher resistance to AMPs than caries free individuals21. Nevertheless, bactericidal activities of AMPs in saliva against SM (restricting its growth), which is the main etiological factor of dental caries is evident. With important synergistic or additive interaction of AMPs was found towards SM collectively [21,25-27].
This is also supported by another study findings [18], as an association was found between salivary amino acids in parotid secretion and caries experience, as a trend of amino acids (lysine and Arginine mainly) being significantly higher in caries free individual. Defensins are known as antimicrobial peptides that are rich with these amino acids in their structure. And salivary free arginine could be derived from defensins and other arginine-rich peptide cleavage. Arginine and its related form shown to have a protective role against dental caries. Suggesting that both beta-defensin and alpha-defensin reduce possibilities for caries experience and are connected to absence of caries.
Cathelicidin LL-37 and beta-defensin with caries severity among children
This study also investigated the relation of salivary AMPs and caries severity measured according to ICDAS criteria that is concerned with stage of caries progression (advancement) in the decayed teeth. The results of the present study found non-significant difference in the level of both peptides when compared among different stages of caries severity (d3-d6score); however, the study revealed a significant negative correlation between both cathelicidin and beta-defensins levels with caries severity. Unfortunately, there is no previous study available in literature to compare results with except one study [11] which investigated relation of the caries severity measured by (ICDAS) in early childhood caries with beta-defensin and histatin, and found positive correlation being higher in the more severe or cavitated caries (score ≥ d3) caries compared to milder caries (score Ë?d3). In general, findings of the decreased level of salivary AMPs among children with caries and negative correlation with caries experience and severity could imply (AMPs as a biological/ immune factor) that increases susceptibility to caries. Whereas increased levels among caries free children could possibly reflect a protection (resistance to caries) offered by these AMPs to teeth and oral cavity mainly by their well-known synergistic antimicrobial activity.
Correlation of Salivary Cathelicidin LL-37 and Betadefensin
The present study found a highly significant positive correlation in the levels of the two AMPs; cathelicidin and beta-defensins. This could be supported by available data in literature that exist coexpression of cathlicidins and beta-defensins and they generally act in synergism with each other and with other AMPs (alpha-defensins, histatins, calprotectin), their coexpression could provide a natural antimicrobial barrier [28]. It also agrees with previous findings [21,16] where levels of beta-defensins and cathelicidins found to be positively correlated within same subjects.
References
- Al-Qahtani SM, Razak PA, Khan SD. Knowledge and practice of preventive measures for oral health care among male intermediate schoolchildren in Abha, Saudi Arabia. Int J Environment Res Public Health 2020; 17:703.
- Duangthip D, Gao SS, Chen KJ, et al. Oral health-related quality of life and caries experience of Hong Kong preschool children. Int Dent J 2020; 70:100-107.
- Sengupta K, Ersbøll AK, Christensen LB, et al. Inequality, Familial aggregation, and risk prediction of caries in siblings. JDR Clin Translational Res 2020; 20:2380084420951147.
- Fábián TK, Hermann P, Beck A, et al. Salivary defense proteins: their network and role in innate and acquired oral immunity. Int J Mol Sci 2012; 13:4295-320.
- Krisanaprakornkit S, Khongkhunthian S. The role of antimicrobial peptides in periodontal disease (Part I): An overview of human defensins and cathelicidin. Thai J Periodontol 2010; 1:33-44.
- Grant M, Kilsgård O, Åkerman S, et al. The human salivary antimicrobial peptide profile according to the oral microbiota in health, periodontitis and smoking. J Innate Immunity 2019; 11:432-444.
- Yu G, Baeder DY, Regoes RR, et al. Combination effects of antimicrobial peptides. Antimicro Agents Chemother 2016; 60:1717-24.
- Di Somma A, Moretta A, Canè C, et al. Antimicrobial and antibiofilm peptides. Biomolecules 2020; 10:652.
- Davidopoulou S, Diza E, Menexes G, et al. Salivary concentration of the antimicrobial peptide LL-37 in children. Archives Oral Biol 2012; 57:865-869.
- Jin G, Weinberg A. Human antimicrobial peptides and cancer. In Seminars in cell and developmental biology. Academic Press 2019.
- Jurczak A, KoÅ?cielniak D, Papież M, et al. A study on β-defensin-2 and histatin-5 as a diagnostic marker of early childhood caries progression. Bio Res 2015; 48:1-9.
- Masood M, Yusof N, Hassan MI, et al. Assessment of dental caries predictors in 6-year-old school children-results from 5-year retrospective cohort study. BMC Public Health 2012; 12:1-7.
- Güncü GN, Yilmaz D, Könönen E, et al. Salivary antimicrobial peptides in early detection of periodontitis. Frontiers Cellular Infection Microbiol 2015; 5:99.
- Laputková G, Schwartzová V, BánovÄin J, et al. Salivary protein roles in oral health and as predictors of caries risk. Open Life Sci 2018; 13:174-200.
- Tao R, Jurevic RJ, Coulton KK, et al. Salivary antimicrobial peptide expression and dental caries experience in children. Antimicrob Agents Chemotherapy 2005; 49:3883-38888.
- Colombo NH, Ribas LF, Pereira JA, et al. Antimicrobial peptides in saliva of children with severe early childhood caries. Archives Oral Bio 2016; 69:40-46.
- El-Kwatehy WM, Youssef A. Salivary alpha-defensin 1-3, total protein and total antioxidant in children with gingivitis. Int J Health Sci Res 2016; 7:174-180.
- Ribeiro TR, Dria KJ, de Carvalho CB, et al. Salivary peptide profile and its association with early childhood caries. Int J Paediatr Dent 2013; 23:225-234.
- Toomarian L, Sattari M, Hashemi N, et al. Comparison of Neutrophil apoptosis α-defensins and calprotectin in children with and without severe early childhood caries. Iranian J Immunol 2011; 8:11-19.
- Barrera J, Tortolero S, Rivas A, et al. Increased expression and levels of human β defensins (hBD2 and hBD4) in adults with dental caries. J Health Sci 2013; 3:88-97.
- Phattarataratip E. The role of salivary antimicrobial peptides in shaping Streptococcus mutans ecology.
- Malcolm J, Sherriff A, Lappin DF, et al. Salivary antimicrobial proteins associate with ageâ?related changes in streptococcal composition in dental plaque. Mol Oral Microbiol 2014; 29:284-293.
- Navarra CO, Robino A, Pirastu N, et al. Caries and innate immunity: DEFB1 gene polymorphisms and caries susceptibility in genetic isolates from North-Eastern Italy. Caries Res 2016; 50:589-594.
- Komatsuzawa H, Ouhara K, Kawai T, et al. Susceptibility of periodontopathogenic and cariogenic bacteria to defensins and potential therapeutic use of defensins in oral diseases. Current Pharm Design 2007; 13:3084-3095.
- Komatsuzawa H, Ouhara K, Kawai T, et al. Susceptibility of periodontopathogenic and cariogenic bacteria to defensins and potential therapeutic use of defensins in oral diseases. Current Pharm Design 2007; 13:3084-3095.
- Maisetta G, Batoni G, Esin S, et al. Activity of human β-defensin 3 alone or combined with other antimicrobial agents against oral bacteria. Antimicrob Agents Chemother 2003; 47:3349-3351.
- Nishimura E, Eto A, Kato M, et al. Oral streptococci exhibit diverse susceptibility to human β-defensin-2: Antimicrobial effects of hBD-2 on oral streptococci. Current Microbiol 2004; 48:85-87.
- Dale BA, Tao R, Kimball JR, et al. Oral antimicrobial peptides and biological control of caries. BMC Oral Health 2006; 6:1-7.
Author Info
Ghufran Mohammed SS Al-Ali1*, Zainab Juma Jafar1 and Batool Hasan AL-Ghurab2
1Department of Pediatric and Preventive Dentistry, College of Dentistry, University of Baghdad, Iraq2Department of Basic Science, College of Dentistry, University of Baghdad, Iraq
Received: 07-Mar-2021 Accepted: 26-Mar-2021 Published: 02-Apr-2021