Research - (2021) Volume 9, Issue 4
Control of Nosocomial Infections in a Hospital with Limited Means
Keerthy Sagar Reddy, Venkatagopalakrishnan R and Baskaran PK
Abstract
Background: Nosocomial infection is happening as post- admission infections in hospitals and requires the prolonging the stay at the wards. Usually, it happens after 48h of hospital admissions and play a significant role in morbidity, mortality and exhibit an elevated loss of patient’s economy. The aim of this study was to find the incidence, etiology and to discuss the control of nosocomial infections in the post-operative patients admitted at Sree Balaji Medical College and Hospital during September 2013 to August 2015.
Methods: 200 patients who were stayed in Surgical Post-Operative and general Wards at least for 48h in were included in the study after getting ethical permission and their informed consent. The microbiological analysis was carried out by strictly sterile conditions to void the false positive results. Data was gathered in a proforma and analysed using Epidemiological Information Package 2012 developed by Centre for Disease Control, Atlanta.
Results: 16 patients out of 200 (8%) developed Nosocomial infection during their hospital stay. The most common Nosocomial infection was urinary tract infection (5.5%) followed by respiratory infection (2%) and blood stream infection (0.5%) in studied patients. The most common organism causing Nosocomial infection was Klebsiella sp. (5%), E.Coli (2%) and Pseudomonas sp.1%).
Conclusion: Patients admitted in the Surgical post-operative ward are subjective to more risk at developing nosocomial infection than patients in general surgical wards. In our study, Urinary tract infection was the commonest followed by respiratory and blood stream infection. Gram-negative organisms were the most common organisms found in this study.
Keywords
Post-admission infections, Nosocomial infections, Common hospital pathogens, Risk factors, Infection in patients
Introduction
Nosocomial infection is the common term for infection occurring at hospitals due to the patients stay and characteristically defined as infections that develops 48 hours after admission into hospitals or within time of 48 hours after being discharged [1,2] and the infectious agent or toxin should not be incubating in the host (patient) at the time of admission. The patients have higher nosocomial risks (5 to 10-fold) at postoperative surgical wards than in general wards [3,4]. Nosocomial infection is more common in the elder patients, immunosuppressed, diabetic, renal failure, family members prone for Multi drug resistant organisms. After getting admitted, a patient's flora acquires the characteristics of surrounding bacterial pool. Most infections which become clinically evident after 48 hours of hospitalization are hospital acquired. Infections which occur after the discharge of the patient from the hospital is healthcare associated if the organisms were acquired during the postoperative ward stay. Hospital based programs of prevention, control and surveillance of hospital acquired infections are in place since the 1950s [5]. The Study on the Efficacy of Nosocomial Infection Control Project (SENIC) in 1970s shows that nosocomial rates can be reduced by 32% if infection surveillance were coupled with necessary infection control programs [6]. In 2005, the National Healthcare Safety Network (NHSN) was started in the U.S to integrate and succeed prior surveillance systems at the Center for Disease Control and Prevention (CDC). The National Nosocomial Infections Surveillance System for Healthcare Workers (NaSH) [7]. Developed as well as resource poor countries are faced with the excessive burden of healthcare associated infections. In a World Health Organization cooperative study (55 hospitals among 14 countries from four WHO regions), about 8.7% of hospitalized patients had hospital acquired infections [8]. A 6-year surveillance study from 2002 -2007 involving surgical postoperative wards & ICUs in Latin America, Asia, Africa, and Europe using CDC's NNIS definitions, showed higher rates of central line associated blood stream infections, ventilator associated pneumonias and catheter associated urinary tract infections than those of comparable United States surgical ICUs and post-operative wards [9].
Patients are treated better in hospitals than in other places. But the presence of many patients under the same roof, in the same room facilitate the spread of infection from a person to another. Infections in hospitals have existed even in ancient times. Hospital acquired infections in this era of powerful antibiotics still are an important consequence of hospitalization. A minimum of 4% of patients are discharged from the hospital after acquiring infections based on underlying disease of the patient, hospital size and numerous other factors. These kind of infections places a huge burden on the patient and the country. It prolongs hospital stay of the patient involved. So, it affects the economy of the patient’s family as the patient and his family could not go to work. Indirectly it even affects the production of the country. Nosocomial infection can be prevented by maintaining asepsis in the concerned ward. Washing hands of the health personnel is the most important factor. In addition to maintaining strict asepsis during urinary catheterization, changing post-operative dressings, changing colostomy bags, cleaning drainage tubes, during intubation, during insertion of vascular catheter is especially important. The present study aimed to analyse the nosocomial incidence in our hospital and formulate a practical method to avoid it.
To find incidence of Nosocomial infections in patients admitted in the Surgical post-operative ward premises of Sree Balaji Medical College & Hospital. To study the etiological agents in such infections. To discuss the management and control measures of nosocomial infections.
Materials and Methods
Inclusion criteria
All the patients admitted in surgical ward and who have undergone a surgery and then subsequently admitted in Surgical post-operative ward in Sree Balaji Medical College Hospital, during the period of September 2013– August 2015 were included in this study (N=200).
Exclusion criteria
All patients admitted in surgical post-operative ward for less than 48 hours.
Patients with evidence of sepsis at admission.
Patients with proven pre-existing infection.
Selection and study of this patients were done as mentioned in the proforma.
Study design
Prospective study.
Geographic distribution
Geographic distribution of the patients were predominantly from areas of Chromepet, rural Chennai and Kanchipuram districts.
Methodology
All the patients were asked a thorough and detailed history and general and systemic examination were done. Incidence, rate of infection, also known as cumulative incidence rate method, is to measure the frequency of new cases of nosocomial infections occurred in a given time. Since the measurement data and the method required are easy to be collected and calculated, it was widely used by many articles [10-12]. However, the weakness of this method is that it does not consider the time of hospitalization as well as some other risk factors that would influence the incidence rate. As indicated by research, the Nosocomial infection incidence rate was nearly zero in the first day of admission, significantly increased after 1 week’s stay, peaked during 4 to 7 weeks’ stay, then dropped as the time went on [13,14]. After careful clinical examination of the patients all were submitted to the following investigations.
Basic lab investigations
Complete blood count.
Blood-Sugar, Urea, Creatinine.
Liver function tests.
Urine analysis.
Culture.
Urine.
Sputum.
Blood.
Stool.
Chest X-Ray.
All the culture samples were delivered to laboratory in a sterile manner immediately.
Collection of samples
Urine
10 to 20 ml of mid-stream urine was collected in a sterile, dry, clean, wide necked bottle by explaining to the patient to avoid contamination i.e., a clean catch sample. The bottles were labeled with name, date and time of collection of samples. The sample was sent to the lab immediately and processed.From catheterized patients urine sample was collected by disinfecting the wall of catheter at its juncture with the drainage tube. Urine was aspirated from a sterile disposable syringe.
Blood
For blood culture the vein puncture site was washed with soap, rinsed with sterile water, cleaned with a swab of 70% alcohol and then dried. Blood was drawn with sterile 10 ml syringe and transferred to blood culture bottle containing thioglycate broth, tryptic soya broth and the bottles were gently rotated to ensure mixing of blood with the broth. The whole procedure is done in an aseptic manner to avoid contamination. Blood was drawn from two separate sites and two samples were sent. Catheter tip was also sent in a culture bottle.
Sputum
Sputum was collected after the patient rinses his mouth with sterile distilled water to remove excessive saliva and food debris. The patients were asked to cough deeply, and the expectorated sputum was collected in a sterilized screw capped open mouth containers. The suction material from the endotracheal tube was also collected in a sterile container and sent to the lab immediately. Endotracheal tube tip was also sent for culture.
Statistical analysis
The information collected regarding all the selected cases were recorded in a Master Chart. Data analysis was done with the help of computer using Epidemiological Information Package (EPI 2010) developed by Centre for Disease Control, Atlanta. Using this software range, frequencies, percentages, means, standard deviations, chi square and 'p' values were calculated. Yate’s corrected chi square test was used to test the significance of difference between qualitative variables. A 'p' value less than 0.05 is taken to denote significant relationship.
Results
Relation between age and infection
The total number of patients included in the study was 200 during the period of 2013–2015 in Sree Balaji Medical College & Hospital.
The age and infection distributions were compiled in Table 1.
| Cases | ||
|---|---|---|
| No | % | |
| Up to 20 years | 19 | 9.5 |
| 21-30 years | 29 | 14.5 |
| 31-40 years | 26 | 13 |
| 41-50 years | 37 | 18.5 |
| 51-60 years | 45 | 22.5 |
| 61-70 years | 34 | 17 |
| >70 years | 10 | 5 |
| Total | 200 | 100 |
| Range | 13-80 years | |
| Mean | 46.0 years | |
| SD | 17.3 years | |
Table 1: Age distribution and prevalence of nosocomial infection.
Table 1 describes the number of patients admitted in relation with age of patient. The mean age of admission of patients in surgical post-operative ward was 46 years. 37% of patients were below 40 years of age and 63% of patients were above 40 years. 19 patients were below 20 years of age, 29 patients were between 21 to 30 years of age, 26 patients were between 31 to 40 years of age, 37 patients were between 41 to 50 years of age,45 patients were between 51 to 60 years age, 34 patients were between 61 to 70 years and 10 patients were more than 70 years of age. The greatest number of patients admitted were in the age group of 51 to 60 years of age-22.5%. As in all wards old age people are admitted more than younger people in Surgical post-operative ward. This is because old age people have decreased immunity, associated diseases like diabetes, hypertension, Coronary heart disease, Carcinoma. Also, they may be smokers and alcoholics. The rate of recovery is good in young patients compared to old age patients. Our study reveals that more patients were admitted to our hospital’ s surgical post-operative ward above 40 years of age than younger patients (Figure 1).
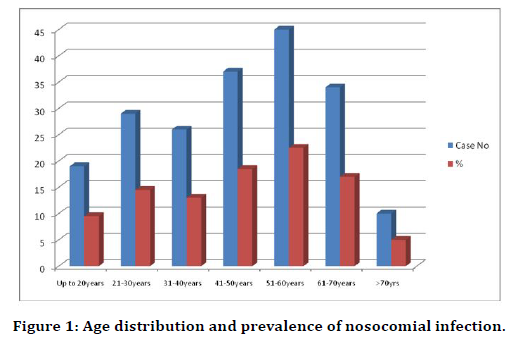
Figure 1. Age distribution and prevalence of nosocomial infection.
Sex distribution
The total number of patients included in the study was 200 during the period of 2013–2015 in Sree Balaji Medical College & Hospital, 114 were male and 86 were female patients. The Age, Sex distributions were compiled in Table 2. Total male patients were 114 and female patients were 86 in this study. 57% of the patients were male and 43% were female. More male patients are admitted in Post-operative ward than female patients (Figure 2).
| Sex | Cases | |
|---|---|---|
| No | % | |
| Male | 114 | 57 |
| Female | 86 | 43 |
| Total | 200 | 100 |
Table 2: Sex distribution and prevalence of nosocomial infections.
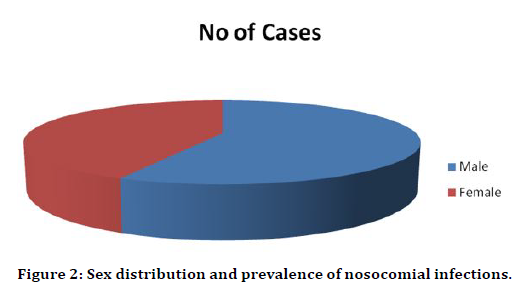
Figure 2. Sex distribution and prevalence of nosocomial infections.
Significance of Risk Factors
Diabetes mellitus and Nosocomial infection
Out of the 200 patients in our study 30 patients had previous history of hypertension and 42 patients had previous history of diabetes out of which 12 patients had both (Table 3 and Figure 3). Out of 42 diabetic patients 4 had nosocomial infection which is around 10% (Table 4). Table 5 and Figure 4 shows that out of 16 patients with nosocomial infection 4 were diabetic i.e., 25% were diabetic.
| Disease | No of Patients |
|---|---|
| Diabetes | 42 |
| Hypertension | 30 |
| Both | 12 |
| Total | 200 |
Table 3: Prevalence of diabetes and hypertension.
| Diabetes as a risk factor | Number of patients |
|---|---|
| DM with Nosocomial infection | 4 |
| DM without Nosocomial | 38 |
| Total DM patients | 42 |
Table 4: Nosocomial infection in DM and Non-DM patients.
| Risk Factors | Number of patients |
|---|---|
| DM | 4 |
| NON-DM | 12 |
| Total | 16 |
Table 5: DM as a risk factor in patients with nosocomial infections.
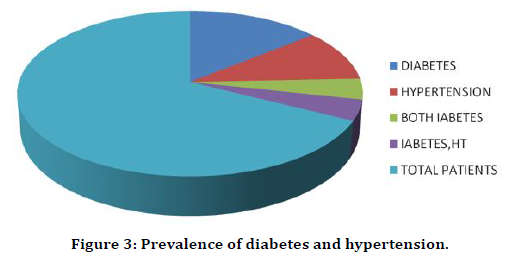
Figure 3. Prevalence of diabetes and hypertension.
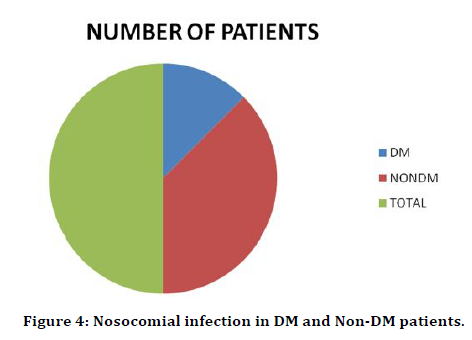
Figure 4. Nosocomial infection in DM and Non-DM patients.
Relation between UTI and diabetes mellitus
In our study there were 11 cases of Urinary tract nosocomial infection. Out of these only 2 patients were diabetic. This amounts to 18% of the patients with UTI (Table 6 and Figure 5). Out of 42 Diabetics only 2 patients had UTI. This is 5% of the patients with UTI (Table 7 and Figure 6).
| Risk factor | No of patients with UTI |
|---|---|
| DM | 2 |
| NON-DM | 9 |
| Total | 11 |
Table 6: Urinary tract infection with DM as a risk factor.
| Risk factor | Number of patients |
|---|---|
| DM | 2 |
| NON-DM | 40 |
| Total | 42 |
Table 7: Urinary tract infection with DM as a risk factor.
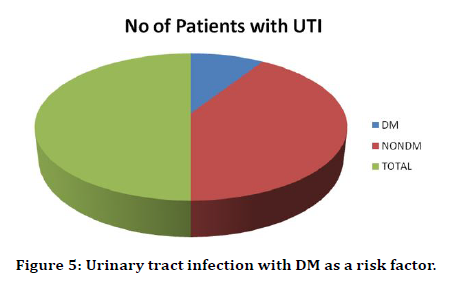
Figure 5. Urinary tract infection with DM as a risk factor.
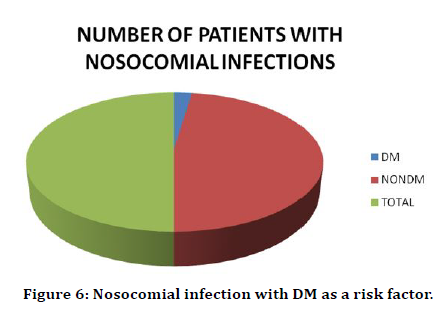
Figure 6. Nosocomial infection with DM as a risk factor.
Respiratory nosocomial infection and diabetes mellitus
There were 4 patients out of 200 who had respiratory tract infection. Out of these 4 patients only one patient had Diabetes mellitus. So, 25% of the patients with respiratory infection had Diabetes Mellitus (Table 8 and Figure 7).
| Risk Factors | Number of patients |
|---|---|
| DM | 1 |
| NON-DM | 3 |
| Total | 4 |
Table 8: Respiratory Nosocomial infection with DM as a risk factor.
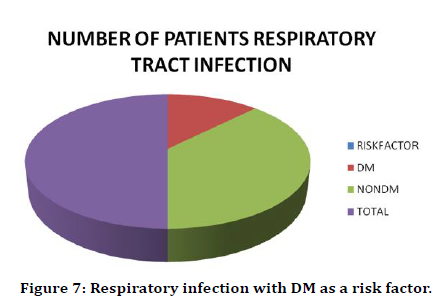
Figure 7. Respiratory infection with DM as a risk factor.
Respiratory infection and Diabetes mellitus
In this study 42 diabetic patients were admitted in post-operative ward. But only one had respiratory infection. This amounts to 2% (Table 9 and €Figure 8).
| Infection | Number of patients |
|---|---|
| Respiratory infection | 1 |
| NO Resp Infection | 41 |
| Total | 42 |
Table 9: Respiratory nosocomial infection and DM.
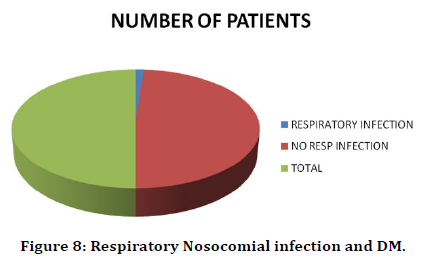
Figure 8. Respiratory Nosocomial infection and DM.
Blood stream infections and diabetes mellitus
Out of 42 diabetic patients only one had blood stream infection. This amounts to 2% of the infection (Table 10 and Figure 9). In this study only one patient had blood stream infection who was a diabetic (Table 11).
| Infection | Number of patients |
|---|---|
| Blood infection | 1 |
| No blood infection | 41 |
| Total | 42 |
Table 10: Blood stream infection with DM as a risk factor.
| Risk factor | Number |
|---|---|
| DM | 1 |
| Non-DM | 0 |
| Total | 1 |
Table 11: Blood stream infection and DM.
Figure 9. Blood stream infection and DM.
Incidence of nosocomial infections (Table 12 and Figure 10)
| Parameter | Value % |
|---|---|
| Total POW admissions during study period | 200 |
| Number of nosocomial infections | 16 |
| Incidence of nosocomial infection | 8 |
Table 12: Incidence of nosocomial infections.
Figure 10. Incidence of Nosocomial infections.
Nosocomical infection as per various cultures
Out of 20 patients 16 patients had nosocomial infection which amounts to incidence of 8%. 10 patients had urinary tract infection. 4 patients had hospital acquired pneumonia and only 1 patient had blood stream infection. 5.5% patients had urinary infection, 2% patients had sputum infection and 0.5% patients had blood infection (Table 13 and Figure 11).
| Culture | Positive cases | Negative cases | ||
|---|---|---|---|---|
| No | % | No | % | |
| Urine | 11 | 5.5 | 189 | 94.5 |
| Sputum | 4 | 2 | 196 | 98 |
| Blood | 1 | 0.5 | 199 | 99.5 |
| Total | 16 | 8 | 184 | 92 |
| ‘p’ value between | ||||
| Urine & sputum culture |
0.1143 Not significant | |||
| Urine & blood | 0.0083 Significant | |||
| Sputum & blood culture |
0.1859 Not significant | |||
Table 13: Nosocomial infections as per various cultures.
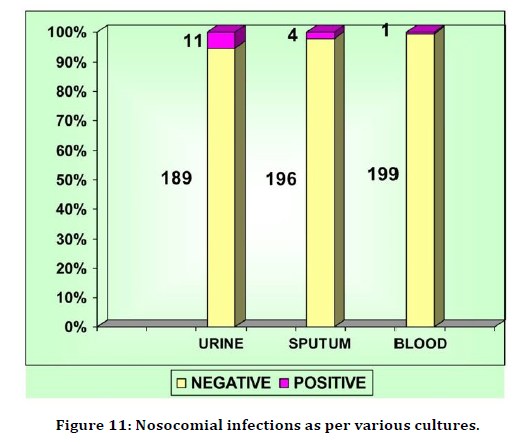
Figure 11. Nosocomial infections as per various cultures.
Organisms in cultures
Urinary tract infection was caused by Klebsiella in 6 patients, E. coli in 4 patients and pseudomonas in 1 patient. Hospital acquired pneumonia was seen in 4 patients out of which 3 were due to Klebsiella and 1 due to pseudomonas. Blood stream infection was seen in 1 patient only which was caused by Klebsiella. 5.5% patients had urinary tract infection, 2% patients had hospital acquired pneumonia and only 0.5% patient had hospital acquired blood stream infection. The most common organism causing nosocomial infection is Klebsiella followed by E. coli (Table 14).
| Organisms | Positive cases in | |||||||
|---|---|---|---|---|---|---|---|---|
| Urine culture | Sputum culture | Blood | Total | |||||
| No | % | No | % | No | % | No | % | |
| Klebsiella | 6 | 3 | 3 | 1.5 | 1 | 0.5 | 10 | 5 |
| Escherichia coli | 4 | 2 | - | - | - | - | 4 | 2 |
| Pseudomonas aeruginosa | 1 | 0.5 | 1 | 0.5 | - | 2 | 1 | |
| Total positive | 11 | 5.5 | 4 | 2 | 1 | 0.5 | 16 | 8 |
| Negative | 189 | 94.5 | 196 | 98 | 199 | 99.5 | 184 | 92 |
Table 14: Organisms in various cultures.
10 patients had infection with Klebsiella, 4 had E.Coli infection and only 2 had infection with Pseudomonas. Klebsiella was responsible for 62% of hospital acquired infection, Escherichia coli was responsible for 25% of infections and pseudomonas aeruginosa was responsible for 13% of infection (Table 15 and Figure 12).
| Positive | Negative cases | |||
|---|---|---|---|---|
| Organisms | No | % | No | % |
| Klebsiella | 10 | 5 | 190 | 95 |
| Escherichia coli | 4 | 2 | 196 | 98 |
| Pseudomonos aeruginosa | 2 | 1 | 198 | 99 |
| Total | 16 | 8 | 184 | 92 |
| ‘p’ value | ||||
| Klebsiella and E.Coli | 0.1737 Not significant | |||
| E.Coli and Pesudomonas | 0.0402 Significant | |||
Table 15: Nosocomial infections as per various organisms.
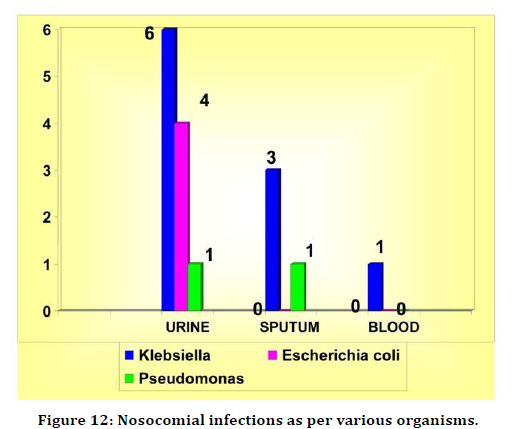
Figure 12. Nosocomial infections as per various organisms.
All 16 infections were seen in patients aged more than 40 years of age. None of the infection was seen in patients below 40 years of age. The mean age of the patient affected with Nosocomial infection was 59.3 years. Age was a significant factor in the incidence of Nosocomial infection as per this study. The p value was 0.0011 which was highly significant (Table 16 and Figure 13).
| Nosocomial infection | Age in years | ||
|---|---|---|---|
| Range | Mean | SD | |
| Positive | 41 –80 | 59.3 | 9.6 |
| Negative | 13 –79 | 44.9 | 17.3 |
| ‘p’ | 0.0011 Significant | ||
Table 16: Age and Nosocomial infection.
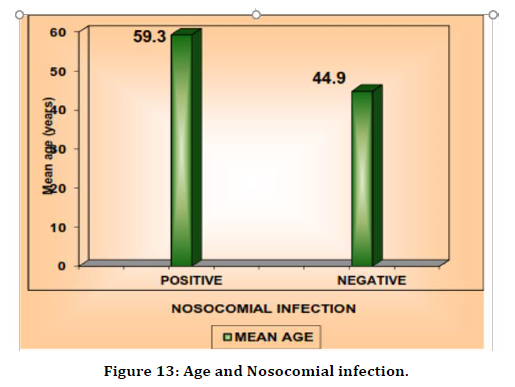
Figure 13. Age and Nosocomial infection.
Sex and nosocomial infection
There was equal distribution of infection between male and female patients. 8 male and 8 female patients had nosocomial infection. Male patients were affected in 7% cases and female patients in 9.3% cases. There was no statistically significant difference in infection betwe en both sex. UTI was present in 6 females and 5 males. So the UTI was seen in 55% of females and 45% of males. So there was no significant difference in UTI between male and female (Table 17 anf Figure 14).
| Nosocomial infection | ||||
|---|---|---|---|---|
| Positive | Negative | |||
| Sex | No | % | No | % |
| Male (114) | 8 | 7 | 106 | 93 |
| Female (86) | 8 | 9.3 | 78 | 90.7 |
| ‘p’ | 0.7441 Not significant | |||
Table 17: Sex and Nosocomial infection.
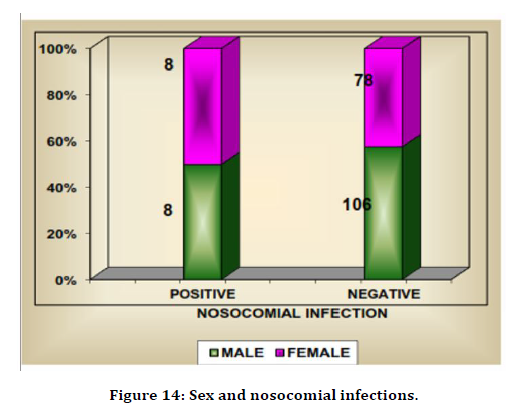
Figure 14. Sex and nosocomial infections.
Respiratory infection in both sexes
Respiratory infection was seen in 4 patients of which were male and only one female. But the significance of difference between both sex could not be ascertained as the total number of patients with respiratory infection is less (Figure 15).
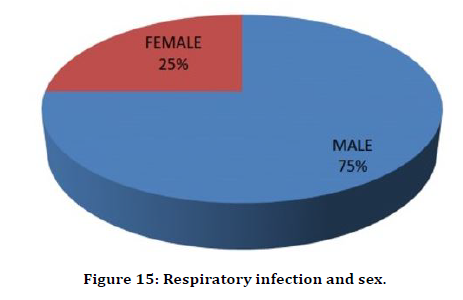
Figure 15. Respiratory infection and sex.
Relation between fever and nosocomial infection
All the 16 patients with nosocomial infection developed fever. So 100% of patients with nosocomial infection developed fever (Figure 16).
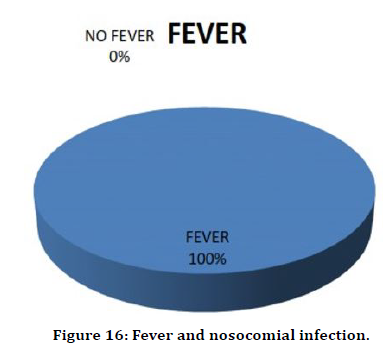
Figure 16. Fever and nosocomial infection.
Consolidation in chest x-ray and respiratory nosocomial infection
All 4 patients with Respiratory Nosocomial infection had consolidation in CXR (Figure 17).
Figure 17. Consolidation in Chest X Ray and respiratory nosocomial infection.
Relation between leucocytosis and nosocomial infection
All 16 patients with Nosocomial infection had Leucocytosis (Figure 18).
Figure 18. Leucocytosis and nosocomial infection.
Discussion
The incidence of Nosocomial infection in this study is 8%. The commonest infection was Urinary tract infection followed by respiratory tract infection and blood stream infection. Klebsiella was the most common organism followed by Escherichia coli and pseudomonas aeuroginosa. Urinary tract infection was caused mostly by Klebsiella and Escherichia coli and pseudomonas. Respiratory infection was caused by Klebsiella and pseudomonas. Blood stream infection was caused by Klebsiella. The age of patients admitted in Post-operative ward was between 13 years and 85 years. Most patients were above 40 years of age i.e 63% of patients. The most common age group was between 40 to 50 years i.e 22.5%. All nosocomial infections were in patients above 40 years old and it is statistically significant. In this study age had a significant relation to Nosocomial infection. In this study nosocomial infection was equally distributed between male and female. There was no statistically significant correlation between sex and Nosocomial infection. In this study out of the 16 patients with nosocomial infection 4 had diabetes mellitus i.e. 25% of the patients with nosocomial infection had diabetes mellitus. Urinary tract infection was seen in 2 diabetics and 9 non- diabetics i.e 18% were diabetic and the remaining 82% were non-diabetic. Out of the 4 patients with respiratory infection one was a diabetic i.e 25% were diabetic and 75% nondiabetic. Blood stream infection was seen in only one patient who was diabetic.
Relation with the aim of the study
The result in this study falls within the average range of infection in India. As per this study Urinary tract infection is the commonest nosocomial infection in the post-operative ward. Diabetes mellitus is seen in 25% of patients with Nosocomial infection But out of 42 patients with diabetes only 4 had Nosocomial infection which was 10% only.
Limitations and Strengths
This study has some limitations. All patients admitted in Post-operative ward could not be included in the study because most of the patients were shifted out before 48 hours of admission. Sample could be obtained only one time as most of the patients were shifted to the surgical ward from Post-operative ward. Empirical antibiotics were given to all patients admitted in our Postoperative ward in combination with the postoperative antibiotics. This is the reason for low incidence of Nosocomial infection in our study. The relation between duration of stay in the post-operative ward and the incidence could not be calculated. The sample size in this study is 200 which is relatively a large sample. The study was conducted thought the year. So, there is less chance for seasonal variation in this study.
Prevention and Control
Prevention of nosocomial infection should be a priority in all hospitals. To prevent nosocomial infection, it is important to understand the mechanisms behind its development and the different strategies employed to prevent the transmission of microorganisms. Role of surveillance systems: National surveilance was initiated in the USA in 1970. Since that time CDCs National Nosocomial Infections Surveilance (NNIS) system has provided hospitals with standardized methods to collect and compare data on nosocomial infections. A study was done that showed the efficacy of infection-control programs and surveillance. The incidence of nosocomial infection was decreased with 32% in comparison to hospitals without strategies where it increased by 18%. 12 There are also recommendations for local surveillance systems, especially in wards, such as ICU, which experience high frequency of infections. Local surveillance includes continuous collection, recording of data and feedback on the incidence of nosocomial infections. Furthermore, should all hospitals have an integrated and monitored infection control-program. This program should include components that are aimed at decreasing the frequency of infections. These programs have proven to be amazingly effective. They inform, educate and give feedback. Further they provide documentation and data, which can be compared between and within hospitals. The key components in the program may vary between hospitals, but generally includes limiting the transmission of organisms through adherence to the basic precautions (hand hygiene, glove use and aseptic procedures), isolation strategies and proper handling of waste. Other factors such as minimizing of invasive procedures and identification and control of outbreaks are also included. Types of Preventive Strategies in Hospitals: There are various strategies, ranging from obvious to high-tech, that may be employed to minimize the occurrence of nosocomial infections and the emergence and spread of multi-drug resistant pathogens. These preventive measures can be divided into three categories. The first category concerns the hospital environment, 15 the second targets the prevention of the most frequent nosocomial infection and the last is aimed at the prevention of the development of multi-drug resistant pathogens.
General preventive measures:
Hand-washing and alcohol disinfection.
Barrier methods.
Cleaning environment. Isolation.
Aseptic techniques.
Appropriate use of prophylactic antibiotics
Prevention of specific nosocomial infections
Reduction in multi-drug resistant pathogens Antibiotic Policy Infection-control program
General preventive measures: There are some preventive measures that are known as universal or basic precautions. These basic procedures apply when handling all patients, regardless of suspected or confirmed diagnosis or infection status. They are important to prevent the transmission of microorganisms and to protect the patient. The most frequent route of transmission is by direct or indirect contact. Direct contact involves body surface to body surface, and it may happen in all forms of daily patient care. Indirect transmission involves contact between the patient and a contaminated object. In general, for transmission to occur the host must be susceptible and the other person infected or colonised with bacteria (hands, nose, throat). These universal procedures are based on the principle that all body fluids, non-intact skin and mucous membranes are potentially infective. Implementation of these to all patients will reduce the risk of infection from both known and unknown sources and is a prerequisite for an effective control of infection.
The basic procedures include
Hand hygiene is considered a key element in the prevention of direct transmission of pathogens. This was the first international request supported by WHO and the 16 World Alliance for Patient Safety.18 Washing hands thoroughly, especially between contact with different patients and after contact with blood or other body fluids, should be routine. Also, after handling equipment or articles that may have been contaminated. Hand washing might seem like something simple, but it is often done incorrectly. Thus, it is important to educate staff and remind them about the importance of hand hygiene. Barrier precautions: Gloves have an important role in reducing the transmission of microorganisms. Hospital staff wear gloves for different reasons; most importantly they provide a protective barrier, and they prevent contamination of hands when handling material such as blood, body fluids, secretions and excretions. Their protection of contamination with blood-borne organisms is extremely important when handling patient with HIV, Hepatitis B or C. Gloves also protect the patient from those organisms colonising hands of hospital personnel. Thus gloves should be used during the application of invasive device and aseptic procedures, and during patient-care, especially if it involves the patient's mucous membranes or non-intact skin. Gloves must always be changed between patients and hands should be washed after gloves are removed. Wearing gloves does not replace the need for hand washing, because gloves may have small, nonapparent defects or may be torn during use, and hands can become contaminated during removal of gloves. There was done a study to assess the protective effect of gloves during surgical procedures. The study concluded that depending on duration of wear, surgical gloves developed micro-perforations not immediately recognized by staff and this created a route for transmission of microorganisms. Other strategies used to create a barrier for transmission are: Mask, eye protection and face shield. They are used during procedures that are likely to generate splashes or sprays of blood or other body fluids and secretions. The usage of masks also protects the patient from droplet or airborne transmission of bacteria during close contact between hospital staff and patient. Gown is used in procedures or patient-care where there is a likelihood of contamination of health care worker and during surgical procedures to protect the patient. Environment: All equipment used in patientcare must be cleaned and reprocessed properly before it comes in contact with other persons. Needles and other sharp objects must be handled with care to prevent injuries and transmission of bloodborne infections. There are established routines and guidelines in hospitals for handling of waste and contaminated materials. Every hospital should also have adequate procedures for routine care, cleaning and disinfection of environmental surfaces. Isolation. There are two types of isolation. The first is “protective” and includes the isolation of immunocompromised patients to reduce risk for opportunistic infections. The reasons for decreased immunity can be many. It can be physiological, due to extreme of age and malnourishment, or it can be due to diseases (DM, cancer, liver cirrhosis, AIDS.) Also, the increasing use of immunosuppressant drugs (corticosteroids, chemotherapy) may contribute to a le ss effective immune system. The second type of isolation is “source isolation”. Here the infected or colonized patient is isolated to prevent the transmission to staff or other patients. It is considered gold standard to isolate the infected patient in a single room. This can be problematic sometimes due to limited space aAnd crowded conditions and the necessity to transfer patients between units can make this difficult to manage. Asepsis describes a condition in which living pathogenic organisms are absent, creating a state of sterility.31 Antisepsis is the use of chemical solutions or disinfectants to reduce the number of microbes present on skin. To reach a state of asepsis varies techniques are being used, including surgical hand washing, scrubs, sterile clothes/gowns, clean room, and sterile instruments. The proper preparation of patient with shaving, cleaning and applying of disinfectant is also important.3 The application of invasive devices, such as endotracheal tube (mechanical ventilation), catheters (urinary and central venous) and surgical drains are increasing. Devices can easily become colonized and due to their disruption of host’s normal protective barriers they allow microorganisms to pass into tissue where they don’t belong and with the potential to cause disease. This has made the proper use of aseptic precautions even more important. First, something simple as cleaning will be mentioned. Where the aim is to remove visible dirt. The use of soaps or detergent diluted in water remove microorganisms from the object due to the mechanical action (because neither possess antibacterial action). Thorough cleaning can remove up to 90% of microorganisms and increase the effect of disinfection and sterilization carried out afterwards. It must be done in a standardized manner to be effective. Sterilization can be achieved by physical and chemical means and it means that the object is 18 free of microorganisms. Physical methods are based on the action of heat (autoclaving, dry thermal or wet thermal) or radiation with gamma rays. Chemical methods include gas sterilisation with for example ethylene oxide. Disinfection is the application of a solution to an animate or inanimate surface.
Three distinctions can be made based on the guidelines from CDC
High level disinfection can be expected to destroy all microorganisms, except for large quantity of spores.
Intermediate disinfection inactivates mycobacterium, vegetative bacteria, most viruses, and fungi. Do not necessarily kill bacterial spores. Low level: Kill most bacteria, some viruses and some fungi. Cannot be relied on to kill resistant bacteria and spores. Often the more active the disinfectant, the more toxic it is. Toxic solutions can be applied to inanimate surfaces, but for human skin the less toxic alternatives should be used. Thus there is no ideal disinfectant. It must be chosen according to the individual situation. Solutions include alcohol, iodine, chlorhexidine, hydrogen peroxide and others.
Antibiotic prophylaxis
Chemoprophylaxis is used only when it has been documented that benefits are greater than risks. Some of the indications are selected surgical procedures, persons at risk of infective endocarditis (patient with mechanical heart valves) and in severely immunocompromised patient where the chance for infection is higher.28 There are conflicting result when it comes to VAP and administration of antibiotic prophylaxis. There exist some clinical evidence that aerosolised antibiotics could prevent VAP, but they are weak. The concerns about high cost, resistant bacteria and other potentially risk factors of aerosolised antibiotics led several evidence-based groups to recommend against routine use of antibiotic prophylaxis. Surgical operations are classified according to the purpose of antibiotic prophylactics. The indications for antibiotic prophylactics can vary between different hospitals, but in general this classification can be followed. Clean: includes procedures that are performed in normally sterile tissue. Examples are orthopaedics, neurosurgery, and cardiovascular procedures. The incidence of SIS is <2% and antibiotic prophylactics is not indicated. The exception is insertion of prosthetic devices (knee or hip arthroplasty), where antimicrobial agents are given because an infection here would be disastrous. Contaminated: included gastrointestinal, respiratory and genitourinary tracts. Here antibiotics are given for 48 hours. Infected: here it will be appropriate with antibiotic treatment instead of prophylactics, because an infection already exist.8 The prophylactics should be given IV and within one hour prior to the intervention. Administration of antibiotics for longer time before a procedure is counterproductive, as there will be an increased risk for infection by resistant bacteria. Prevention of specific nosocomial infections: Urinary tract infection. This is the most common hospital acquired infection. It is related to the use of urinary catheter in 80%. Fortunately, this infection is associated with less morbidity than the other infections. In average, it prolongs the hospital stay with 3,8 days. 20 The aetiology is ascending gram-negative bacteria (E.coli, Kleibsiella and Proteus). They cause infection by colonising the catheter, which bypasses the normal barriers of the patient. It creates a direct route for the bacteria into the bladder. (Candida may also cause infection, more commonly in immunocompromised hosts.) Risk factors for development of UTI are prolonged catheterization, diabetes, female sex and age above 50 years.
Prevention
Catheterise only when necessary. The insertion should be atraumatic and done under sterile conditions. The drainage of urine should be closed (standard). Maintain good patient hygiene to prevent the contamination and colonisation of bacteria. The catheter should be changed with appropriate intervals. Intervals depend on the development of symptoms or not. If no symptoms it can be kept for ca 3 weeks. If symptoms, such as dysuria, change of colour or blood in urine, it should be changed at once. In some patients it can be convenient to screen the urine for bacteria (in some ICU settings) at appropriate intervals.
HAP
It is ranged third among nosocomial infections (15%) in USA and the prolonged stay is calculated to be on average 5,9 days. 20 The aetiology is often endogenous, and includes microorganisms colonising stomach and upper airways and mouth. The resultant infection is thus often mixed with bacteria such as Pneumococcus, Hemophilus, S.aureus, Enterobacteria and Pseudomonas.7,38 Other microorganisms affecting the lungs are viruses, especially Respiratory syncitial virus (RSV) causing bronchiolitis in children. Where Influenza virus is more frequent in elderly patient. This viral infection possesses a risk for secondary bacterial infection (superinfection) of the lungs. With severely immunocompromised hosts, organisms such as Legionella and Aspergillus may be the etiological agent. Prevention: different breathing exercises and cough techniques, correct position of patient in bed (semi- recumbent, 30 degrees) and early mobilisation are recommended. In some cases, the need to increase analgesics may be needed to keep the patient motivated and pain free when mobilised. The patient should be educated and explained why these activities are important to get a better compliance Catheter related bloodstream infection: Catheter-related bloodstream infections (CRBSIs) are an important cause of nosocomial infection, especially in the ICU, where patients frequently have invasive device. It was estimated by the CDC a rate of 1,8 -5,2 bloodstream infections per 1000 catheter days. It is likely that they cause a substantial number of deaths in hospitalized patients.18 The average of prolonged stay varies from 7 -24 days.20 The source of infection is most frequently due to contamination of the catheter, either during its 22 insertion or it can be contaminated later from the patient’s skin. Thus, bacteria constituting the cutaneous flora are most encountered. These are coagulasenegative staphylococci (S. epidermidis 60 -90%), S.aureus and Candida.8 Enterococcus and gram negatives can also be found in some situations. The main risk factors are the length of catheterization, improper insertion, and poor post insertion care. Prevention: There exist a sixstep strategy that can be followed to reduce the occurrence of these infections. It includes antiseptic hand washing, including disinfection, full aseptic precautions and use of chlorhexidine during insertion. Further should the femoral route be avoided if possible and keep days of catheter placement to a minimum. Other preventive strategies are the impregnation of the catheters with antibiotics, either minocyclinerifampicin or silver sulfadiazine. This is recommended in high-risk patients and patient with long-term cannulation. The major concern here is the potential to induce multi-drug antimicrobial resistance. The use of ultrasound during insertion is good in difficult cases and in inexperienced doctors. It will reduce the formation of hematoma and strictly aseptic, and the time it is open should be minimised. Care at the insertion site with the maintenance of adequate hygiene and dressings and the frequent inspection to identify any change in the area around the catheter are of importance. The catheters should be replaced with regular intervals to reduce chance of colonisation and infection. Peripheral catheters can be changed every 48 - hour and central catheter every seventh day. If any changes occur before, the catheter is removed, and tip of catheter is send to microbiology for culture. Changes to be aware of include local signs of inflammation and systemic signs s uch as fever and rigors. In a patient with fever of unknown origin (FUO) and with CVC, hospital staff should always suspect catheter-related sepsis. 7, 8 Patient receiving total parenteral nutrition (TPN) through CVC lumen has increased risk for CRBSI. If the TPN is with high glucose concentration, the doctor should also keep in mind the possibility of Candida infection. Surgical site infections (SSI): SSI is infection in the site of surgery (wound infection). Incidence varies from 0,5 - 22 % depending on type of operation and general state of patient. The same is true when it comes to the aetiology agent. But in general are S.aureus, coagulasenegative Staphylococci, Enterococci, E.coli, Pseudomonas and Enterobacter most frequently encountered. The development of SSI prolongs the stay by ca 7, 4 days.20 Factors influencing SSI are the patient’s general health, especially the existence of underlying diseases (diabetes, malignancy, malnutrition and obesity). Others are the operation, surgical teams, postoperative care and antibiotic prophylaxis. The presence of foreign bodies or drains and the intrinsic virulence of the microorganisms are additional factors influencing the occurrence of SSI. Prevention can be divided into 3 phases. Preoperative: Minimize the stay of patient in hospital before the procedure. Be sure that the area being incised is shaved and thoroughly cleaned. Antibiotic prophylaxis is given when indicated. The surgical team and the operation room are prepared after aseptic procedures. Intraoperative: Use correct techniques, ensure bleeding control, excise devitalised tissue and remove foreign bodies. Keep surgical staff to a minimum and follow aseptic procedure. Postoperative: Aseptic care of the site, insert drain if necessary. The type of drain used depends on the site drained. If it is potentially infected, open drain will be applied. If it is draining a sterile place, a closed drain is often used. The wound should then be covered with sterile dressings. Preventive strategies to limit the development of MDR pathogens: Bacteria can develop resistance towards antibiotics. This is a problem in both the community and in health care facilities. The ways a pathogen may become resistant to antibiotics can be divided into intrinsic or acquired. Intrinsic resistance may be due to the lack of the molecular target for an antibiotic or that the membrane is impermeable to the agent. Acquired resistance is principally due to one of four mechanisms: drug inactivation, reduced permeability, drug-efflux or target modification. Many of the bacteria causing diseases in hospitals have a high level of resistance to antimicrobial therapy. This is due to the fact that many patients receive antibiotics, especially broad-spectrum, when hospitalised. They are administered both for therapy and as prophylactics. An example is in ICU, where 60% of patients receives antibiotics at any given time. This makes ICU a natural environment for the development of resistant pathogenic strains. Of concern are Staphylococci resistant to Methicillin and Vancomycin (MRSA, VRSA), Vancomycinresistant Enterococci (VRE), ESBL-producing Klebsiella, Enterobacter, Pseudomonas and Acinetobacter. There were an estimated 94,360 invasive MRSA infections in 2005, with 18,650 deaths. MRSA now kills more people annually in the USA than AIDS and breast cancer combined. Thus the prevention of resistance in bacteria is becoming more and more important due to the growing problem. There is a wide national difference of multi-drug resistance. In Europe, microbial resistance data is collected by the EARSS programme (European Antimicrobial Resistance Surveillance System) funded by the European Commission. This system tests antimicrobial susceptibility in different countries. The collected data provide national results for comparison. This documentation keeps hospitals informed about the prevalence of resistant strains in their region. The Antimicrobial Use Committee is a committee that provides information about the appropriate use of antibiotics. They recommend, guide and educate hospital administrations by developing antibiotic-control policies. Further, they receive documentation from hospitals about antimicrobial administration and trends during a given period. All these measures contribute to an overview of how and when antibiotics are administrated. Hospitals often develop their own antibiotic policy and program. Here they inform about the appropriate use, according to dose, choice and duration. Other general recommendations in antibiotic policies, include the limitation of topical antimicrobials, administration of narrow spectrum when possible, avoidance of combination of antibiotics if not necessary and the obtainment of appropriate specimen for microbiological examination before any treatment is initiated. These programs and policies are believed to decrease the inappropriate administration of antibiotics, thus reducing the development of resistant bacteria. Another problem arising is the interchange of patients between hospitals and community, and the widespread use of antibiotics means that many multi-drug resistant organisms are now found in non-hospital environments. Lastly, and not to forget, are viruses and fungi. They may also develop resistance to treatment, and this may be of concern due to the lack of other treatment options for these organisms. An example is Herpes virus, which has been reported to be resistant to Acyclovir and Ganciclovir. HIV positive patients are most affected by this. Vaccinations. As antibiotics only are effective against bacteria, vaccines were developed to target viruses, including smallpox, measles, mumps, typhoid fever, rubella, diphtheria, tetanus, yellow fever, pertussis, and poliomyelitis. Advancements in antiviral drugs were made starting in the 1970s with the introduction of Acyclovir to protect against herpes and cold sores In Norway we have a vaccination program which was started in 1952 and it is recommend today that all children should be vaccinated against 10 different diseases: - DTP combination vaccine against diphtheria, tetanus and whooping cough -Hib vaccine against Haemophilus influenza type b infection-Vaccination against pneumococcal disease -Vaccination against poliomyelitis -MMR combination vaccine against measles, mumps and rubella -Vaccination against human papillomavirus (HPV, from 2009) Vaccine against tuberculosis (BCG vaccine) is no longer in the vaccination program. This was started from the school year 2009/2010. The vaccine is now only 27 offered to children with increased risk for TBC infection. The same holds for Hepatitis B vaccination. A special indication is for all health care workers to be vaccinated against Hepatitis B and Hepatitis A. This is due to the increased risk for infection when exposed or handling patient’s body fluids, secretions and excretions. In addition to vaccines that are part of the childhood vaccination program, it is offered a variety of other vaccines such as influenza vaccine, and the different vaccines for travelling. Early recognition of nosocomial infections: Depending of type of disease, there are some common manifestations of infection that often are present. The diagnosis and recognition of the disease mentioned in the text (UTI, HAP, SSI, CRBSI, gastroenteritis) can be quite easy to recognize if they present with their classical symptoms and signs, and if in patients who are not of extreme age or sedated. However, this is not always the case and sometimes the only reliable and obvious sign is fever. Thus, a change in temperature (increased/decreased) should always alert a physician that something is wrong. The occurrence of fever is a frequent finding in hospitalised patients, and especially in ICU patients. In the critically ill it should be taken even more seriously and call for an urgent response. This is du e to the increased risk of progression into a more severe clinical picture, such as sepsis. Further, are patients in ICU often deeply sedated and the presence of medical devices (mechanical ventilation, urinary catheter, invasive device) makes it difficult for patient to report symptoms and for the physician to see or find them. Recognition of sepsis: Sepsis is an acute clinical syndrome that needs prompt recognition and resuscitation. The syndrome reflects the response of the host and not the cause. Thus, the immediate threat for the patient is not only the uncontrolled growth of bacteria, but also the consequences of the systemic inflammatory response. The biochemical process of sepsis is complex and includes the activation and interaction of different mediators of the immune system, coagulation cascade, vasoactive mediators and acute phase reactions. The result is vasodilatation, loss of endothelial barrier, occlusion of capillaries and impaired myocardial contractility, which all leads to insufficient perfusion and reduced oxygen delivery to tissues, producing the clinical picture. The criteria of SIRS are often 28 present and should alert a physician to look for the cause and primary site of infection by history, physical examination and tests, including urinalysis and urine culture (particularly in patients who have indwelling catheters), serial blood cultures, and cultures of other suspect body fluids. As sepsis progress, signs of changed mental status (confusion or deceased alertness) develop, blood pressure starts to fall, and oliguria can often be noticed. The increase of BUN and serum creatinine indicates the failing of kidneys and the progression of renal insufficiency the patient often hyperventilates to compensate for the raised level of lactic acid, which occur in shock due to the anaerobic metabolism. This cause a low PaCo2 and manifestations of respiratory alkalosis. As shock progresses metabolic acidosis develop and pH decreases. The progression to shock indicates that body’s compensatory measures have failed [15-51].
Conclusion
The incidence of nosocomial infection in patients admitted in Surgical Post-operative ward was 8% in the study population. The commonest infection was Urinary tract infection followed by respiratory tract infection and blood stream infection. Gram negative bacteria especially Klebsiella was the predominant organism. Age was a significant factor in this study. All patients who had Nosocomial infection were above 40 years old. Old age patients were more susceptible than young patients. There was no significant difference in incidence between male and female patients. This study was conducted in a single hospital, hence needs further evaluation, hence more studies in multiple hospitals are required to arrive at a better conclusion.
Funding
No funding sources.
Ethical Approval
The study was approved by the Institutional Ethics Committee.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgments
The encouragement and support from Bharath University, Chennai is gratefully acknowledged. For provided the laboratory facilities to carry out the research work.
References
- Mahieu LM, De Muynck AO, Ieven MM, et al. Risk factors for central vascular catheter-associated Blood stream infections among patients in a neonatal intensive care unit. J Hosp Infect 2001; 48:108-116.
- Ferrer M, Valencia M, Torres A. Management of Ventilatorassociated pneumonia. In: Vincent JL. Book of intensive careand emergency medicine. Verlag Berlin Heidelberg: Springer, 2008; 353–364.
- Wenzel RP, Edmond MB. The impact of hospital- acquired bloodstream infections. Emerg Infect Dis 2001; 7:174-177.
- Vincent JL, Bihari DJ, Suter PM, et al. The prevalence of nosocomial infection in intensive care units in Europe. Results of the European prevalence of infection in intensive care (EPIC) study. EPIC international advisory committee. JAMA 1995; 274:639–644.
- https://www.cdc.gov/mmwr/preview/mmwrhtml/00017800.htm
- Hughes JM. Study on the efficacy of nosocomial infection control (SENIC Project): Results and implications for the future. Chemotherapy 1988; 34:553-561.
- Edwards JR, Peterson KD, Andrus ML, et al. National healthcare safety network (NHSN) report, data summary for 2006 through 2007, issued November 2008. Am J Infect Control 2008; 36:609-626.
- Tikhomirov E. WHO programme for the control ofhospital infections. Chemioterapia 1987; 6:148-151.
- Rosenthal VD, Maki DG, Mehta A, et al. International nosocomial infection control consortium report, data summary for 2002-2007, issued January 2008. Am J Infect Control 2008; 36:627-637.
- Tam AY, Yeung CY. The changing pattern of severe neonatal staphylococcal infection: A 10 -yearstudy. AustPaediatr J 1988; 24:275-279.
- Tseng YC, Chiu YC, Wang JH, et al. Nosocomial bloodstream infection in a neonatal intensive care unit of a medical center: A three-year review. J MicrobiolImmunol Infect 2002; 35:168-172.
- Lachassinne E, Letamendia-Richard E, Gaudelus J. Epidemiology of nosocomial infections in neonates. Archives Pediatr 2004; 11:229-233.
- Freeman J, McGowan JE. Differential risk of nosocomialinfection. Am J Med 1981; 70:915-918.
- Freeman J, McGowan JE. Methodologic issues inhospital epidemiology. III. Investigating the modifying effects of time and severity of underlying illness on estimates of cost of nosocomial infection. Reviews Infect Dis 1984; 6:285-300.
- Aiken AM, Mturi N, Njuguna P, et al. Risk and causes of paediatric hospital-acquired bacteraemia in Kilifi district hospital, Kenya: A prospective cohort study. Lancet 2011; 378:2021-2027.
- Gastmeier P, Geffers C, Brandt C, et al. Effectiveness of a nationwide nosocomial infection surveillance system for reducing nosocomial infections. J Hosp Infect 2006; 64:16-22.
- Klevens RM, Edwards JR, Richards CL, et al. Estimating healthcare- associated infections in US hospitals, 2002. Public Health Rep 2007; 122:160-166.
- https://www.cdc.gov/ncidod/dhqp/%20pdf/Scott_CostPaper.%20pdf.%20Accessed7/1/2009
- Richards MJ, Edwards JR, Culver DH, et al. Nosocomial infections in pediatric intensive care units in the United States. National Nosocomial Infections Surveillance System. Pediatrics. Apr 1999;103(4):e39.
- Grohskopf LA, Sinkowitz-Cochran RL, Garrett DO, et al. Anational point-prevalence survey of pediatric intensive care unit-acquired infections in the United States. J Pediatr 2002; 140:432-438.
- Sohn AH, Garrett DO, Sinkowitz-Cochran RL, et al. Prevalence of nosocomial infections in neonatal intensive care unit patients: Results from the first national point- prevalence survey. J Pediatr 2001; 139:821-827.
- Edward AM, Warren DK, Fraser VJ. Ventilator-associated pneumonia in pediatric ntensive care unit patients: Risk factors and outcomes. Pediatrics 2002; 109:758-764.
- Fayon MJ, Tucci M, Lacroix J, et al. Nosocomial pneumonia and tracheitis in a pediatric intensive care unit: A prospective study. Am J Respir Crit Care Med 1997; 155:162-169.
- Zaoutis TE, Coffin SE. Clinical syndromes of device associated infections. In: Long SS, Pickering LK, Prober CG. Principles and practice of pediatric infectious diseases. 3rd Edn. Churchill Livingstone 2008.
- Chastre J, Wolff M, Fagon JY, et al. Comparison of 8 vs 15 days of antibiotic therapy for ventilator-associated pneumonia in adults: A randomized trial. JAMA 2003; 290:2588-2598.
- Collee JG, Fraser AG, Marmion BP, et al. Culture M2A7. National of bacteria. In: Mackie McCartney practical medical microbiology. 14th Edn, (Churchill Livingstone, London) 1996; 113-129.
- National Committee for Clinical Laboratory Standards: Performance standards for antimicrobial susceptibility testing. 8th Information Supplement M2A7. National Committee for Clinical Laboratory Standards, Villanova, PA.
- Sengupta A, Lehmann C, Diener-West M, et al. Catheter duration and risk of CLA-BSI in neonates with PICCs. Pediatrics 2010; 125:648-653.
- Butler-O'Hara M, D'Angio CT, Hoey H, et al. An evidence-based catheter bundle alters central venous catheter strategy in newborn infants. J Pediatr 2012; 160:972 -977.
- Hei MY, Zhang XC, Gao XY, et al. Catheter-related infection and pathogens of umbilical venous catheterization in a neonatal intensive care unit inChina. Am J Perinatol 2012; 29:107-114.
- van derZwet WC, Kaiser AM, van Elburg RM, et al. Nosocomial infections in a Dutch neonatal intensive care unit: surveillance study with definitions for infection specifically adapted for neonates. J Hosp Infect 2005; 61:300-311.
- Couto RC, Pedrosa TM, TofaniCde P, et al. Risk factors for nosocomial infection in a neonatal intensive care unit. Infect Control Hospital Epidemiol 2006; 27:571-575.
- Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care–associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 2008; 36:309 -332.
- Harley RW, Culver DH, White JW, et al. The efficacy ofinfection surveillance and control programmes inpreventing Nosocomial infections in US hospitals. Am J Epidemiol 1985; 12:182.
- Cook DJ, Walter SD, Cook RJ, et al. Incidence of and risk factors for ventilator-associated pneumonia in critically ill patients. Ann Intern Med 1998; 129:440.
- Chastre J, Fagon JY. Ventilator-associated pneumonia. Am J Respir Crit Care Med 2002; 165:867–903.
- Celis R, Torres A, Gatell JM, et al. Nosocomial pneumonia: A multivariate analysis of risk and prognosis. Chest 1988; 93:318–324.
- Craven DE, Kunches LM, Lichtenberg DA, et al. Nosocomial infection and fatality in medical and surgical intensive care unit patients. Arch Intern Med 1988; 148:1161–1168.
- Shannon SC. Chronic critical illness. In Jesse BH, Gregory AS, Lawrence DH, Eds. Principles of critical care. 3rd Edn, McGraw Hill, 2005; 207–215.
- Tablan OC, Anderson LJ, Besser R, et al. Guidelines for preventing health-care-associated pneumonia, 2003: Recommendations of the CDC and the healthcare infection control practice advisory committee. MMWR Recomm Rep 2004; 53:1–36.
- Richards MJ, Edwards JR, Culver DH, et al. Nosocomial infections in combined medical-surgical intensive care units in the United States. Infect Control Hosp Epidemiol 2000; 21:510–511.
- Laupland KB, Zygun DA, Davies HD, et al. Incidence and risk factors for acquiring nosocomial urinary tract infection in the critically ill. J Crit Care 2002; 17:50–57.
- Erbay H, Yalcin AN, Serin S, et al. Nosocomial infections in intensive care unit in a Turkish university hospital: A 2 -year survey. Intensive Care Med 2003; 29:1482–1488.
- O'Grady NP, Alexander M, Dellinger EP, et al. Guidelines for the prevention of intravascular catheter related infections. Infect Control Hosp Epidemiol 2002; 23:759–769.
- Finkelstein R, Rabino G, Kassis I, et al. Device associated, device-day infection rates in an Israeli adult general intensivecare unit. J Hosp Infect 2000; 44:200–205.
- Rosser CJ, Bare RL, Meredith JW. Urinary tract infections in the critically ill patient with a urinary catheter. Am J Surg 1999; 177:287 – 290.
- Jean YF, Jean C. Nosocomial pneumonia. In Mitchell PF, Edward A, Vincent JL, et al. Edn. Text book of critical care. 5th Edn. Elsevier 2005; 663–677.
- American Thoracic Society and Infectious Diseases Society of America. Guidelines for the management of adults with hospital acquired, ventilator-associated and healthcare associated pneumonia. Am J Respir Crit Care Med 2005; 171:388–416.
- Rello J, Ollendorf DA, Oster G, et al. Epidemiology and outcomes of ventilator-associated pneumonia in a large US database. Chest 2002; 122:2115–21121.
- Marra AR, Wey SB, Castelo A, et al. Nosocomial bloodstream infections caused by Klebsiella pneumoniae: Impact of extended spectrum ß-lactamase (ESBL) production on clinical outcome in a hospital with high ESBL prevalence. BMC Infect Dis 2006; 14:24.
- Pfaller MA, Jones RN, Doern GV, et al. Bacterial pathogens isolated from patients with bloodstream infection: Frequencies of occurrence and antimicrobial susceptibility patterns from the ENTRY antimicrobial surveillance program (United States and Canada, 1997). Antimicrobial Agents Chemotherapy 1998; 42:1762–1770.
Author Info
Keerthy Sagar Reddy, Venkatagopalakrishnan R and Baskaran PK
Department of General Surgery, Sree Balaji Medical College & Hospital, Bharath Institute of Higher Education and Research, Chennai, Tamil Nadu, IndiaCitation: Keerthy Sagar Reddy, R Venkatagopalakrishnan, PK Baskaran, Control of Nosocomial Infections in a Hospital with Limited Means, J Res Med Dent Sci, 2021, 9 (4): 365-380.
Received: 20-Mar-2021 Accepted: 16-Apr-2021
